Abstract
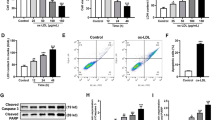
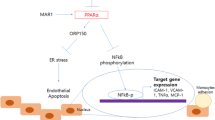
Atherosclerotic cerebral infarction (ACI) is characterized by extremely high fatality and disability rate. Recent studies indicate that co-stimulatory signal of tumor necrosis factor superfamily OX40/OX40L contributes to the atherosclerosis effect in ACI patients. However, it remains unclear the mechanism underlying the anti-atherosclerosis process. So this study aims to investigate the effects of rosuvastatin on the expression of OX40L, peroxisome proliferator-activated receptors gamma (PPAR-γ) in human umbilical vein endothelial cells (HUVEC), and human peripheral blood lymphocytes. Different concentration of rosuvastatin and oxidized low-density lipoprotein (OX-LDL) co-intervene HUVEC to observe the expression of OX40L and PPAR-γ using real-time quantitative RT-PCR (Q-RTPCR) and Western-blot. Furthermore, we examined the level changes of plasmic sOX40L and hs-CRP in acute atherosclerotic cerebral infarction patients. The results demonstrated that concentration-dependent and time-dependent OX-LDL remarkably stimulate the expression of OX40L and inhibit the expression of PPAR-γ in vitro. But concentration-dependent rosuvastatin can reverse the impact of OX-LDL, suggesting that rosuvastatin can prevent the expression of OX40L, and the process may be associated with mevalonate pathway. In vivo, acute atherosclerotic cerebral infarction patients taking 20 mg rosuvastatin exhibited significantly reduced expression of OX40L in peripheral blood lymphocyte, sOX40L in blood plasma, and hs-CRP compared with before treatment. Our studies identified rosuvastatin as an effective medicine in controlling atherosclerosis process in ACI by inhibiting OX40L and stimulating PPAR-γ expression.





Similar content being viewed by others
References
Alexander RW (1995) Hypertension and the pathogenesis of atherosclerosis. Hypertension 25(1):155–161
Andarini S, Kikuchi T, Nukiwa M, Pradono P, Suzuki K, Ohkouchi S, Inoue A, Maemondo M, Ishii N, Saijo Y, Sugamura K, Nukiwa T (2004) Adenovirus vector-mediated in vivo gene transfer of OX40 ligand to tumor cells enhances antitumor immunity of tumor-bearing hosts. Cancer Res 64(1):3281–3287
Bhakdi S, Dorweiler B, Kirchmann R, Torzewski J, Weise E, Tranum-Jensen J, Walev I, Wieland E (1995) On the pathogenesis of atherosclerosis: enzymatic transformation of human low-density lipoprotein to an atherogenic moiety. J Exp Med 182(6):1959–1971
Collins AR, Meehan WP, Kintscher U, Jackson S, Wakino S, Noh G, Palinski W, Hsueh WA, Law RE (2001) Troglitazone inhibits formation of early atherosclerotic lesions in diabetic and nondiabetic low-density lipoprotein receptor-deficient mice. Arterioscler Thromb Vasc Biol 21(3):365–371
Collot-Teixeira S, Martin J, McDermott-Roe C, PostonR MGJL (2007) CD36 and macrophages in atherosclerosis. Cardiovasc Res 75:468–477
Do KH, Yeo SS, Lee J, Jang SH (2013) Injury of the corticoreticular pathway in patients with proximal weakness of cerebral infarct: diffusion tensor tractography study. Neurosci Lett 546(1):21–25
Dutta P, Courties G, Wei Y, Leuschner F, Gorbatov R, Robbins CS, Iwamoto Y, Thompson B, Carlson AL, Heidt T, Majmudar MD, Lasitschka F, Etzrodt M, Waterman P, Waring MT, Chicoine AT, van der Laan AM, Niessen HW, Piek JJ, Rubin BB, Butany J, Stone JR, Katus HA, Murphy SA, Morrow DA, Sabatine MS, Vinegoni C, Moskowitz MA, Pittet MJ, Libby P, Lin CP, Swirski FK, Weissleder R, Nahrendorf M (2012) Myocardial infarction accelerates atherosclerosis. Nature 487(7404):325–329
Feig JE, Shang Y, Rotllan N, Vengrenyuk Y, Wu C, Shamir R (2011) Statins promote the regression of atherosclerosis via activation of the CCR7-dependent emigration pathway in macrophages. PLoS One 6(12):e28534
Flanagan K, Fitzgerald K, Baker J, Regnstrom K, Gardai S, Bard F, Mocci S, Seto P, You M, Larochelle C, Prat A, Chow S, Li L, Vandevert C, Zago W, Lorenzana C, Nishioka C, Hoffman J, Botelho R, Willits C, Tanaka K, Johnston J, Yednock (2012) Laminin-411 Is a vascular ligand for MCAM and facilitates TH17 cell entry into the CNS. PLoS One 7(7):e40443
Han L, Li MG, Liu YX, Han CG, Ye P (2012) Atorvastatin may delay cardiac aging by upregulating peroxisome proliferator-activated receptors in rats. Pharmacology 89(1–2):74–82
Henson P (2003) Suppression of macrophage inflammatory responses by PPARs. Proc Natl Acad Sci U S A 100(11):6295–6296
Gold JA, Karulf M, Kelly A, Weinberg AD, Jeffrey A (2010) OX40 ligand regulates inflammation and mortality in the innate immune response to sepsis. J Immunol 185(8):4856–4862
Lin R, Liu J, Gan W, Yang G (2004) C-reactive protein-induced expression of CD40-CD40L and the effect of lovastatin and fenofibrate on it in human vascular endothelial cells. Biol Pharm Bull 27(10):1537–1543
Liu P, Yan J, Gong J, Wang C, Chen G (2011) Positive correlation between pregnancy-associated plasma protein-A level and OX40 ligand expression in patients with acute coronary syndromes. Biomed Pharmacother 65(3):193–197
Martín-Ventura JL, Blanco-Colio LM, Gómez-Hernández A, Munoz-Garcia B, Vega M, Serrano J, Ortega L, Hernandez G, Tunon J, Egido J (2005) Intensive treatment with atorvastatin reduces inflammation in mononuclear cells and human atherosclerotic lesions in 1 month. Stroke 36(8):1796–1800
Marx N, Bourcier T, Sukhova GK, Libby P, Plutzky J (1999) PPAR-gamma activation in human endothelial cells increases plasminogen activator inhibitor type-1 expression: PPAR-gamma as a potential mediator in vascular disease. Arterioscler Thromb Vasc Biol 19(3):546–551
Mendel I, Shevach EM (2006) Activated T cells express the 0X40 ligand: requirements for induction and co-stimulatory function. Immunology 117(2):196–204
Nakano M, Fukumoto Y, Satoh K, Tto Y, Kagaya Y, Ishii N, Sugamura K, Shimokawa H (2010) OX40 ligand plays an important role in the development of atherosclerosis through vasa vasorum neovascularization. Cardiovasc Res 88(3):539–546
Ohshima Y, Yang LP, Uchiyama T, Tanaka Y, Baum P, Sergerie M, Hermann P, Delespesse G (1998) OX40 co-stimulation enhances interleukin-4 (IL-4) expression at priming and promotes the differentiation of naive human CD4(+) T cells into high IL-4-producing effectors. Blood 92(9):3338–3345
Ortego M, Bustos C, Hernández-Presa MA, Tuñón J, Diaz C, Hernandez G, Egido J (1999) Atorvastatin reduces NF-kappaB activation and chemokine expression in vascular smooth muscle cells and mononuclear cells. Atherosclerosis 147(2):253–261
Peng DQ, Huang S, Yuan SG, Zhao SP (2010) Increased soluble OX40L is associated with carotid intima-media thickness. Clin Lab 56(9–10):449–457
Rajamannan NM, Subramaniam M, Stock SR, Stone NJ, Springett M, Ignatiev KI, McConnell JP, Singh RJ, Bonow RO, Spelsberg TC (2005) Atorvastatin inhibits calcification and enhances nitric oxide synthase production in the hypercholesterolemic aortic valve. Heart 91(6):806–810
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3(6):1101–1108
Shao Q, Shen LH, Hu LH, Pu J, Jing Q, He B (2012) Atorvastatin suppresses inflammatory response induced by OX-LDL through inhibition of ERK phosphorylation, IκBα degradation, and COX-2 expression in murine macrophages. J Cell Biochem 113(2):611–618
Srivastava AK, Kalita J, Dohare P (2009) Studies of free radical generation by neurons in a rat model of cerebral venous sinus thrombosis. Neurosci Lett 450:127–131
Sugawara A, Takeuchi K, Uruno A, Ikeda Y, Arima S, Kudo M, Sato K, Taniyama Y, Ito S (2001) Transcriptional suppression of type 1 angiotensin II receptor gene expression by peroxisome proliferator-activated receptor-gamma in vascular smooth muscle cells. Endocrinology 142(7):3125–3134
Valzasina B, Guiducci C, Dislich H, Killeen N, Weinberg AD (2005) Triggering of OX40 (CD134) on CD4(+)CD25+ T cells blocks their inhibitory activity: a novel regulatory role for OX40 and its comparison with GITR. Blood 105(7):2845–2851
Voqiatzi G, Tousoulis D, Stefanadis C (2009) The role of oxidative stress in atherosclerosis. Hellenic J Cardiol 50:402–409
Wang XS, Ishimori N, Korstanje R, Rollins J, Paigen B (2005) Identifying novel genes for atherosclerosis through mouse–human comparative genetics. Am J Hum Genet 77(1):1–16
Wang YH, Liu YJ (2007) OX40-OX40L interactions: a promising therapeutic target for allergic diseases? J Clin Invest 117(12):3655–3657
Weber C, Noels H (2011) Atherosclerosis: current pathogenesis and therapeutic options. Nat Med 2 17(11):1410–1422
Xu Z, Zhao S, Zhou H, Ye H, Li J (2004) Atorvastatin lowers plasma matrix metalloproteinase-9 in patients with acute coronary syndrome. Clin Chem 50(4):750–753
Zingoni A, Sornasse T, Cocks BG, Tanaka Y, Santoni A, Lewis L (2004) Cross-talk between activated human NK cells and CD4+ T cells via OX40–OX40 ligand interactions. J Immunol 173(1):3716–3724
Acknowledgments
This project is supported by the Natural Science Foundation of Heilongjiang Province, China (grant no. D201167 ) and Science and Technology Project of Heilongjiang Province, China (grant no. GC10C305-2).
Conflicts of Interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jing-Yu Zhang and Feng-Jun Wang contributed equally to the present study.
Rights and permissions
About this article
Cite this article
Zhang, JY., Liu, B., Wang, YN. et al. Effect of Rosuvastatin on OX40L and PPAR-γ Expression in Human Umbilical Vein Endothelial Cells and Atherosclerotic Cerebral Infarction Patients. J Mol Neurosci 52, 261–268 (2014). https://doi.org/10.1007/s12031-013-0134-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-013-0134-1