Abstract
The physiological function of interleukin-6 within the central nervous system (CNS) is complex; interleukin-6 exerts neurotrophic and neuroprotective effects and yet can also function as a mediator of inflammation, demyelination, and astrogliosis depending on the cellular context. However, the roles of interleukin-6 in astrocytes are poorly understood. In the present study, we investigated the effect of the pro-inflammatory cytokine interleukin-6 on the production of the inflammatory mediator prostaglandin E2 in mouse astrocytes. Interleukin-6 stimulated prostaglandin E2 production in a time-dependent fashion via a rapid and transient induction of cyclooxygenase-2 messneger RNA, followed by cyclooxygenase-2 protein synthesis. Interleukin-6 may act on the nervous system by interacting with its specific soluble interleukin-6 receptor and the signal transducer 130-kDa glycoprotein. Simultaneous treatment of astrocytes with interleukin-6 and soluble interleukin-6 receptor caused marked induction of prostaglandin E2 synthesis, and this effect was suppressed by adding a neutralizing antibody against soluble interleukin-6 receptor. Furthermore, the mouse 130-kDa glycoprotein antibody suppressed prostaglandin E2 formation induced by interleukin-6, as well as interleukin-6/soluble interleukin-6 receptor complexes, in a dose-dependent manner. These results indicate that interleukin-6/soluble interleukin-6 receptor complexes and the signal transducer 130-kDa glycoprotein play an important role in the regulation of cyclooxygenase-2 expression and subsequent prostaglandin E2 formation in mouse astrocytes and that interleukin-6 is an important regulator of immune and inflammatory processes in the CNS.
Similar content being viewed by others
Introduction
Prostaglandins (PGs) are major lipid mediators produced by cyclooxygenase (COX) activity. They play important modulatory roles in various cells throughout the body. Prostaglandin E2 (PGE2) is the most abundant prostaglandin in the brain (Bishai and Coceani 1992) and is considered to play an essential role in several pathological states. In the central nervous system (CNS), PGE2 is responsible for fever induction (Ushikubi et al. 1998) and participates in eliciting pain hypersensitivity (Samad et al. 2001). In addition, elevated PGE2 levels have been detected in the cerebrospinal fluid of patients with Alzheimer’s disease (AD; Montine et al. 1999), stroke (Jacobs et al. 1987), ischemia (Nogawa et al. 1997), and amyotrophic lateral sclerosis (ALS; Almer et al. 2001). When cells and tissue are exposed to various pro-inflammatory stimuli, arachidonic acid is liberated from membrane phospholipids and converted to PGs by the action of COX enzymes.
COX is the key enzyme in the metabolic pathway leading to PG and thromboxane A2 formation from arachidonic acid (Dubois et al. 1998). Two isoforms have been identified, COX-1 and COX-2 (Smith et al. 1996). COX-1 is constitutively expressed in nearly all normal mammalian tissues and mediates the synthesis of PGs required for physiological tissue homeostasis. By contrast, COX-2 is constitutively expressed in the brain in specific neuronal populations where it seems to be regulated by synaptic activity (Hayaishi 1991; Yamagata et al. 1993; Marcheselli and Bazan 1996). However, in inflammatory pathological conditions, the immune-competent cells of the CNS, astrocytes and microglia, are likely to be the major sources of PGs following induction of COX-2 (Minghetti and Levi 1995; O’Banion et al. 1996; Bauer et al. 1997; Vane et al. 1998; Minghetti et al. 1999; Molina-Holgado et al. 2000).
Interleukin-6(IL-6) was initially described as a central mediator of the immune system, hemopoiesis, and acute phase reaction. IL-6 is a member of the neurocytokine family, which also includes ciliary neurotrophic factor, leukemia inhibitory factor, oncostatin M, and cardiotrophin-1. These molecules have overlapping biological activities: they possess similar secondary structures and exert their activities through related multisubunit receptors (Rose-John and Heinrich 1994; Heinrich et al. 1998). The IL-6 receptor (IL-6R), gp80, can exist either as a membrane-bound protein or a soluble protein (sIL-6R; Kishimoto et al. 1994). Binding of IL-6 to either the membrane-bound or soluble form of IL-6R promotes homodimerization of a 130-kDa glycoprotein (gp130) molecule. Both neurons and glia can produce IL-6 (Gadient and Otten 1994; Sawada et al. 1995; Marz et al. 1998; Their et al. 1999).
Astrocytes are the major cell type in the CNS. These cells not only supply nutrients to neurons but also mediate inflammatory responses in the CNS. Following CNS injury or an immune/inflammatory challenge, astrocytes undergo a phenotypic alteration—a response known as activation. The activated astrocytes then release cytokines and other pro-inflammatory mediators, including PGs. These released substances communicate with such neighboring cells as neurons and microvascular cells. Astrocytes are a major source of PGs in the CNS: in culture, these cells synthesize up to 20 times more PGs than do neurons (Seregi et al. 1984). Their ability to produce PGE2 upon stimulation with interleukin-1β (IL-1β), ATP, platelet-activating factor, transforming growth factor-β (TGF-β), oncostatin M, tumor necrosis factor-α (TNF-α), or lipopolysaccharide (LPS) has been documented (Fontana et al. 1982; O’Banion et al. 1996; Ishimoto et al. 1997; Luo et al. 1998; Mollace et al. 1998; Pistritto et al. 1998, 1999; Molina-Holgado et al. 2000; Teather et al. 2002; Repovic et al. 2003; Xu et al. 2003). However, PGE2 formation upon stimulation of astrocytes with IL-6 has not been investigated. In the present study, we used purified mouse astrocytes to investigate the effects of IL-6 on PGE2 synthesis and the expression of COX-2, which catalyzes PG formation. In addition, possible roles of sIL-6R and gp130 in PGE2 formation were also explored.
Materials and Methods
Materials
PGE2, recombinant mouse IL-6, d-glucose, and bovine pancreas insulin were purchased from Sigma Chemical Co. (St. Louis, MO, USA). DNase I and RNase inhibitor were obtained from Takara-bio Co. (Tokyo, Japan). Oligo(dt) primer and ribonuclease H were purchased from Invitrogen (Carlsbad, CA, USA). Biotin-conjugated goat anti-rabbit IgG and streptavidin–horseradish peroxidase (HRP) were obtained from Dako Cytomation (Kyoto, Japan). Anti-human sIL-6R goat antibody and anti-mouse gp130 goat antibody were purchased from R&D Systems (Minneapolis, MN, USA). Recombinant human sIL-6R and anti-mouse COX-2 rabbit antibody were purchased from PreproTec EC Ltd. (London, UK) and Cayman Chemical Co. (Ann Arbor, MI, USA), respectively. All other reagents were purchased from commercial sources and generally were the highest purity available.
Cell Culture
Astrocytes were prepared from mixed primary glial cell cultures obtained from newborn C57BL/B6 mouse brain as previously described (Sawada et al. 1993). The purity of the astrocytes thus obtained was greater than 95%, as estimated by indirect immunofluorescence using anti-glial fibrillary acidic protein antibody. These astrocyte-enriched cultures did not contain neurofilament-positive neurons and the contamination of microglia was negligible. The cells were cultured in Eagle’s minimum essential medium (Eagle’s MEM; Sigma) containing 10% heat-inactivated fetal bovine serum (FBS; JRH Biosciences, Lenexa, KS, USA), 2 mg/ml glucose, 5 μg/ml bovine insulin, 100 IU/ml penicillin, and 100 μg/ml streptomycin (Gibco BRL, Rockville, MD, USA) at 37°C in an atmosphere of 5% CO2 and 95% air and then subcultured every 3 days before the experiments were performed. For total RNA extraction, cells were plated in 10-cm diameter dishes (Falcon: 5 × 105 cells/dish in 10 ml of culture medium); for the PGE2 enzyme immunoassay (EIA), cells were plated in 24-well plates (Iwaki: 4 × 104 cells/well in 400 μl of culture medium) and then cultured for 2 days until they were nearly confluent. The medium was then replaced with fresh medium, and cells were activated with or without various concentrations of IL-6 or other reagents followed by further culturing for the indicated periods.
Measurement of PGE2
PGE2 was assayed using a specific EIA system (Amersham, UK). Briefly, supernatants of cultured astrocytes or standard PGE2 were incubated for 1 h with specific anti-PGE2 reagent and peroxidase-labeled PGE2 in pre-packed 96-well plates containing a goat anti-mouse solid phase. Unlabeled PGE2 (standard or unknown) and PGE2–peroxidase complex competed for a limited number of binding sites on PGE2-specific antibodies, and the amount of peroxidase-labeled ligand was inversely proportional to the concentration of added standard or unknown. After washing four times with washing buffer, tetramethylbenzidine/hydrogen peroxide single pot substrate was added and incubated for 30 min. Addition of acid solution stopped the reaction, and the resultant color was read at 450 nm in a microtiter plate photometer. The concentration of unlabeled PGE2 in a sample was determined by interpolation from a standard curve, and the data are expressed as pg PGE2 in each well.
Western Blot Analysis
Cells were exposed to 10 ng/ml IL-6 for the indicated periods of time. Cells were washed three times with ice-cold Ca2+- and Mg2+-free phosphate-buffered saline (PBS) and lysed in 100 mM phosphate buffer (pH 7.4) containing 1 mM EDTA and 0.05% Triton X-100 (v/v). The cell lysates were centrifuged at 10,000×g for 10 min at 4°C and the resultant supernatants were used for analysis. Protein concentration was determined by the Bradford method (Bradford 1976) using bovine serum albumin as a standard protein. For Western blotting, 10 μg of cell protein from each sample for COX-2 detection was subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis on an 8% separating gel under reducing conditions. Proteins were then transferred onto poly(vinylidene difluoride) membranes (Millipore, Bedford, MA, USA) by semi-dry blotting. The electroblotted membranes were blocked for 1 h in 2% Block Ace solution (Dainippon Pharmaceutical, Osaka, Japan) and then incubated with anti-mouse COX-2 rabbit antibody (1:32,000) for 1.5 h at room temperature. Subsequently, the membranes were washed three times in PBS containing 0.05% Tween 20 (TPBS) and incubated for 45 min with biotin-conjugated goat anti-rabbit IgG (1:50,000). After washing membranes three times with TPBS, they were incubated with streptavidin-HRP (1:50,000) for 45 min at room temperature and the resulting protein bands were visualized using ECL Plus Western blotting detection reagents (GE Healthcare Bio-Sciences, Piscataway, NJ, USA) according to the manufacturer’s instructions. Immunoblots were digitized using ImageMaster ID Elite (GE Healthcare Bio-Sciences) and expressed as fold induction compared with control cultures.
RNA Extraction and Reverse Transcription PCR Analysis
Total RNA was extracted from astrocytes using ISOGEN (Nippongene Co., Tokyo, Japan) according to the manufacturer’s instructions. The purity of the RNA preparations was checked by measuring the absorbance ratio at 260/280 nm. Each RNA sample was subjected to first-strand complementary DNA (cDNA) synthesis using SuperScript II reverse transcriptase (Invitrogen) following the protocol suggested by the manufacturer. Reactions were performed at 42°C for 50 min. cDNA samples (0.5 μl) were amplified for 30 (β-actin) or 35 cycles (COX-2) in an Astec PC-800 thermal cycler using Taq DNA polymerase and polymerase chain reaction (PCR) reagents (Takara-bio Co.), with primers specific for β-actin and COX-2, in a total volume of 25 μl. The PCR amplimer pairs for the analysis of COX-2 cDNA were 5′-CATGGGTGTGAAGGGAAATAAG-3′ (sense) and 5′-TGAGATAGTGACCGTGGGGG-3′ (antisense), which were complementary to the conserved regions of the cDNA for mouse COX-2 (O’Banion et al. 1992). The β-actin cDNA amplimers were 5′-GTGGGCCGGTCTAGGCACCA-3′ (sense) and 5′-GGTTGGCCTTAGGGTTCAGG-3′ (antisense; Kaartinen et al. 1995). β-actin messenger RNA (mRNA) was assessed to control for the amount and the integrity of RNA in each sample. Each PCR cycle consisted of a denaturation step (94°C for 45 s), an annealing step (60°C for 45 s), and a primer extension step (72°C for 2 min). PCR products were separated by 10% polyacrylamide gel electrophoresis and visualized by ethidium bromide staining. The densities of the bands corresponding to COX-2 and β-actin were analyzed by densitometry (NIH Image J software).
Statistical Analysis
One-way analysis of variance was carried out to determine the levels of significance in experiments. Multiple group comparisons were performed using the Student–Newman–Keuls test. Data are given as means ± SD values.
Results
IL-6 Stimulates PGE2 Release in Cultured Mouse Astrocytes
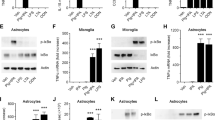
Release of PGE2 into the supernatants of cultured astrocytes was assayed by EIA. The dose-dependent effects of IL-6 were examined by 20-h culturing at concentrations of 0.01 to 10 ng/ml. As shown in Fig. 1a, IL-6 produced a dose-dependent increase in PGE2 release from astrocytes; the plateau was reached at 1 ng/ml. The increase in PGE2 release at plateau, which was about 2.5-fold over the control level, was observed at concentrations of 1 and 10 ng/ml. Based on this observation, IL-6 was used at a concentration of 10 ng/ml in further experiments unless stated otherwise. The time-dependent changes in PGE2 release at a concentration of 10 ng/ml of IL-6 are shown in Fig. 1b. PGE2 release increased significantly from as early as 8 h in culture and thereafter reached a maximum at 20 h in culture. These results indicate that IL-6 induces PGE2 release in astrocytes.
IL-6 induces PGE2 release in astrocytes. Cells were treated with vehicle as a control or the indicated concentrations of IL-6 for 20 h (a). Cells were treated with vehicle as a control (empty circle) and 10 ng/ml IL-6 for various times as indicated (filed circle) (b). After incubation, the culture media were collected and the levels of PGE2 were measured by EIA. Data are mean ± SD (bars) values from five independent experiments. Significant increases in PGE2 release compared with the control are indicated by asterisks: *p < 0.05, **p < 0.01
IL-6 Induces COX-2 mRNA Expression in Cultured Mouse Astrocytes
To determine the mechanism by which IL-6 increases PGE2 release into the supernatants of cultured astrocytes, we isolated RNA from astrocytes and subjected it to reverse transcriptase PCR (RT-PCR) analysis using primers for COX-2. Expression of the β-actin gene was used as a control for normalization. The RT-PCR analysis of RNA isolated from IL-6-treated astrocytes confirmed that COX-2 mRNA was induced in mouse astrocytes by 10 ng/ml IL-6 (Fig. 2). As shown in Fig. 2, treatment with IL-6 resulted in a time-dependent expression of COX-2 mRNA. COX-2 mRNA levels increased significantly from as early as 1 h in culture and thereafter seemed to be produced at a similar level, namely 2.4-fold above the control level, until 5 h in culture; COX-2 mRNA levels returned to the control level after 6 h in culture. These data indicate that COX-2 mRNA levels are increased in IL-6-stimulated astrocytes.
Time course of COX-2 mRNA induction by IL-6 in astrocytes. Cells were treated with 10 ng/ml IL-6 for the indicated times. Total RNA was isolated and the levels of mRNAs for COX-2 and β-actin were determined by RT-PCR (a). Band intensities were analyzed using Image J 1.32 software, and for each sample, COX-2 mRNA levels were normalized to the mRNA levels of the housekeeping β-actin gene. Data are mean ± SD (bars) values from five independent experiments. Values are expressed as fold induction to control cultures defined as 1.0. Significant increases in COX-2 mRNA induction compared with the control are indicated by asterisks: *p < 0.05 (b)
IL-6 Induces COX-2 Protein Synthesis in Cultured Mouse Astrocytes
A recent study demonstrated that IL-6 mediates COX-2 induction in prostatic intraepithelial neoplasia cells (Liu et al. 2002). Therefore, we examined the effect of IL-6 on COX-2 expression by astrocytes. As shown in Fig. 3, astrocytes expressed basal levels of COX-2, and this expression was increased in the presence of 10 ng/ml IL-6. The IL-6 induction of COX-2 expression was time-dependent, with a peak increase noted 2 h after IL-6 treatment, and COX-2 expression remained elevated up to 6 h after treatment. COX-2 migrates in this electrophoretic system as a doublet (owing to glycosylation) approximately 70–72 kDa in size (Morham et al. 1995).
Time course of COX-2 protein induction by IL-6 in astrocytes. Cells were treated with 10 ng/ml IL-6 for the indicated times. Then, cell lysates were prepared and Western blot analysis was performed as described in the “Materials and Methods”. A typical Western blot pattern of COX-2 is shown in a. The approximate sizes of glycosylated and unglycosylated COX-2 proteins are indicated on the right. A quantitative assay of Western blotting was performed using ImageMaster ID Elite. Data are mean ± SD (bars) values from four independent experiments. Values are expressed as fold induction to control cultures defined as 1.0. Significant increases in COX-2 protein induction compared with the control are indicated by asterisks: *p < 0.05
Effect of IL-6 in the Absence and Presence of sIL-6R on PGE2 Release in Cultured Mouse Astrocytes
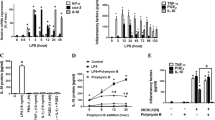
The receptor mediating the biological activities of IL-6 is composed of two subunits: IL-6R and gp130. Additionally, sIL-6R, with a molecular weight of approximately 50 kDa, was identified as a central element in the mediation of IL-6 signaling (Peters et al. 1998). To determine whether IL-6 could complex with sIL-6R and affect PGE2 release, astrocytes were incubated with increasing concentrations of sIL-6R and sIL-6R antibodies. Figure 4a shows the effects of IL-6 on PGE2 production when added in combination with sIL-6R to the culture. When astrocytes were treated with 10 ng/ml IL-6 in the presence of various concentrations of sIL-6R for 24 h, the release of PGE2 was dependent on the concentration of sIL-6R (0.01–100 ng/ml): the maximum increase in PGE2 release, which was about 5.3-fold over the control level (17.83 ± 5.52 ng/ml), was observed at a concentration of 100 ng/ml sIL-6R. Figure 4b shows the effects of a neutralizing antibody against human sIL-6R on PGE2 formation induced by 10 ng/ml IL-6 in a 24-h culture. The human sIL-6R antibody suppressed the PGE2 formation induced by IL-6 in a dose-dependent manner (0.01–10 μg/ml). The effective suppression of PGE2 release induced by sIL-6R antibody was observed at concentrations of 1 and 10 μg/ml. These results indicate that IL-6/sIL-6R complexes stimulate PGE2 release in cultured astrocytes.
PGE2 induction by IL-6 in the presence of sIL-6R in astrocytes. Cells were treated with vehicle as a control or 10 ng/ml IL-6 in the presence of various concentrations of sIL-6R (a) or sIL-6R antibody (b) for 24 h. After incubation, the culture media were collected and the levels of PGE2 were measured by EIA. Data are mean ± SD (bars) values from five independent experiments. Significant increases in PGE2 release compared with the control are indicated by asterisks (a); significant decreases in PGE2 release compared with unstimulated cells are also indicated by asterisks (b): *p < 0.05, **p < 0.01
Effect of a Neutralizing Antibody against gp130 on PGE2 Release in Cultured Mouse Astrocytes
The first step in the IL-6 transducing mechanisms is the binding of the cytokine to its specific receptor subunit followed by the association of a second subunit, the signal transducer gp130 (Hibi et al. 1990). To determine whether IL-6-induced PGE2 release is coupled with gp130, astrocytes were incubated with increasing concentrations of an anti-mouse gp130 antibody. Figure 5 shows the effect of this neutralizing antibody against mouse gp130 on PGE2 formation induced by IL-6 and sIL-6R in a 24-h culture. As the maximum effect on PGE2 release induced by IL-6 was observed at a concentration of 1 ng/ml (Fig. 1a), cells were treated with 1 ng/ml IL-6 in the presence of various concentrations of gp130 antibody, with or without 100 ng/ml sIL-6R. The mouse gp130 antibody suppressed the PGE2 formation induced by IL-6 as well as IL-6/sIL-6R complexes in a dose-dependent manner (0.01–10 μg/ml): the maximum suppressions on PGE2 release induced by IL-6 and IL-6/sIL-6R complexes, which were about 77.7% and 68.3% compared with the control levels, respectively, were observed in the presence of the gp130 antibody at a concentration of 10 μg/ml. As shown in Fig. 5, treatment with gp130 antibody resulted in a decrease in PGE2 release at concentrations of 1 and 10 μg/ml in non-treated cells (control). These results may be the effect of the gp130 antibody on other stimulants.
Participation of gp130 in the induction of PGE2 expression in astrocytes. Cells were treated with 1 ng/ml IL-6 in the presence of various concentrations of gp130 antibody, with or without 100 ng /ml sIL-6R for 24 h. After incubation, the culture media were collected and the levels of PGE2 were measured by EIA. Data are mean ± SD (bars) values from four independent experiments. Significant increases in PGE2 release compared with the control are indicated by asterisks: *p < 0.05, **p < 0.01, ***p < 0.001
Discussion
Astrocyte activation occurs during immune reactions (Saas et al. 2000), and these activated cells synthesize and release such inflammatory compounds as IL-1β and PGs (Dayton and Major 1996). Inflammation-related events appear to have a significant role in the progression and propagation of the neurodegenerative processes in AD (McGeer et al. 1990) and ALS (Almer et al. 2001). Interestingly, the levels of PGs and/or the enzymes involved in PG production are increased in ALS (Almer et al. 2001) and AD (Stephenson et al. 1996). A role for astrocyte-derived PGs in neuronal cell death has been demonstrated. For example, PGE2 stimulates astrocytic glutamate release and prevents astrocytes from taking up glutamate (Bezzi et al. 1998; Pasti et al. 2001); the consequent increase in extracellular glutamate is neurotoxic (Drachman and Rothstein 2000). These results suggest that astrocytes (and the substances they release) may be a critical component in the mechanisms underlying neurodegeneration. Astrocytes are a major source of PGs in the CNS. Their ability to produce PGE2 upon stimulation with several stimulants has been reported. For example, LPS and TGF-β caused a concentration-dependent increase in PGE2 production in primary cultures of rat astrocytes (Luo et al. 1998; Pistritto et al. 1999). Increased production of PGE2 after stimulation with IL-1β has also been shown in murine astrocytes (O’Banion et al. 1996). Repovic et al. (2003) demonstrated that oncostatin M synergistically upregulates PGE2 production induced by the pro-inflammatory mediators IL-1 β, TNF-α, and LPS in astrocytes and astroglioma cells. In this report, we demonstrate that IL-6 increases PGE2 levels at concentrations of 0.1–10 ng/ml and in time-dependent manners in cultured mouse astrocytes (Fig. 1a, b). Since PGE2 is freely permeable and cannot be stored intracellularly, the observed rise in PGE2 level is likely to be due to its de novo synthesis (Smith et al. 2000).
Intracellular PGE2 synthesis requires three classes of enzymes: phospholipase A2, COX, and PGE2 synthase. Each of these enzymes has at least two different isoforms, which may be differently expressed in various cell types. Two COX enzymes have been identified thus far. COX-1 is constitutively expressed and largely responsible for baseline prostaglandin production (DeWitt and Smith 1988). COX-2 is the major inducible form of COX and is associated with delayed prostaglandin production (Xie et al. 1991; Kudo and Murakami 1999). In mouse astrocytes treated with IL-6, COX-2 mRNA and protein levels are highly upregulated in medium supplemented with 10% FBS (Figs. 2 and 3). Thus, IL-6-enhanced PGE2 production is mediated by the stimulatory effect of IL-6 on the expression of COX-2. The present report confirms the results of other studies (Luo et al. 1998; Repovic et al. 2003; Xu et al. 2003) by showing that cultured astrocytes express the inducible COX and secrete PGE2. On the other hand, O’Banion et al. (1996) demonstrated that IL-1β and phorbol 12-myristate 13-acetate, as well as LPS, TNF-α, and basic fibroblast growth factor (bFGF), but not IL-6, caused a significant accumulation of COX-2 mRNA in astrocytes cultured in serum-free medium. A possible explanation for this discrepancy is as follows. The production of PGs is affected by the serum content of the medium. For example, in osteoblasts and vascular smooth myocytes, serum induces a rapid increase in the expression level of COX-2, but has little effect on COX-1 expression (Pilbeam et al. 1993; Pritchard et al. 1994). FBS contains various mitogenic growth factors, such as bFGF, epidermal growth factor, insulin-like growth factor, and platelet-derived growth factor (PDGF). PDGF can upregulate the mRNA for COX in fibroblasts (Lin et al. 1989). Thus, it is conceivable that these mitogenic growth factors mediate the increase in the expression level of COX-2.
In this report, by 5-h culture, the time needed for maximal expression of COX-2, there was no PGE2 synthesis. On the other hand, at the time point where PGE2 is detected (8–24 h culture), there is no COX-2 expression (Figs. 1 and 2). A possible explanation for this discrepancy is as follows. In mammalian cells, prostaglandin-biosynthetic pathways utilizing endogenous arachidonic acid are subdivided into three distinct phases which show different kinetics and possible recruitment of different sets of biosynthetic enzymes. The constitutive immediate response, which occurs within several minutes after stimuli causing a rapid and transient increase in cytoplasmic Ca2+, is regulated by posttranslational activation of constitutively expressed enzymes. The delayed response, which proceeds gradually for several hours after pro-inflammatory stimuli, requires de novo synthesis of particular biosynthetic enzymes. The induced immediate response, which is elicited by Ca2+-mobilizing stimuli after priming by pro-inflammatory stimuli, reflects the combination of the above two responses and involves both constitutive and inducible enzymes. For example, rat peritoneal macrophages treated with LPS predominantly produced PGE2 during culture for 6–24 h by the delayed response. Delayed PGE2 generation was accompanied by increasing expression of cytosolic phospholipase A2 (cPLA2) and de novo induction of COX-2. In the delayed response, cPLA2 and type IIA secretory phospholipase A2 function cooperatively with inducible COX-2, which was in turn coupled with PGE2 synthase. COX-1 was non-functional in the delayed response, even though it was constitutively expressed in the cell (Kudo and Murakami 1999). In mouse astrocytes treated with IL-6, PGE2 synthesis may be regulated by the delayed response.
Cells responsive to IL-6 express on their surface a low affinity receptor that does not have transducing activity. The IL-6/IL-6R complexes induces the homodimerization of a signal-transducing component, gp130, leading to cytoplasmic signaling cascades that activate components of the Janus kinase (JAK)/signal transducers and activators of transcription (STAT) pathway, particularly the activation of the transcription factor STAT-3 (Taga and Kishimoto 1997). sIL-6R can be generated by shedding of the membrane-bound receptor (Mullberg et al. 1993, 1994) or by alternative splicing of mRNA (Lust et al. 1992). Since the transmembrane and cytoplasmic regions of the IL-6R are not essential for signal transduction, sIL-6R can form a complex with IL-6 in solution and associate with gp130, thereby activating signal transduction. Previously, Oh et al. (1998) showed that human astrocytes express low levels of IL-6R and require the addition of sIL-6R for IL-6-mediated responses. Herein, we have shown that treatment of murine astrocytes with IL-6 and sIL-6R leads to marked induction of PGE2 synthesis, and this effect was suppressed by adding a neutralizing antibody against sIL-6R (Fig. 4). These results suggest that sIL-6R plays a pivotal role in determining the level of PGE2 synthesis by astrocytes in the CNS and furthermore influences IL-6 function in the CNS. This is supported by several reports that CNS cells that are normally slightly responsive or unresponsive to IL-6 become responsive on addition of sIL-6R. In human astrocytes, IL-6 alone has no effect on α1-antichymotrypsin expression, whereas addition of sIL-6R leads to its expression (Kordula et al. 1998). In the presence of sIL-6R, IL-6 inhibits TNF-α-induced VCAM-1 expression in astrocytes (Oh et al. 1998). These findings suggest that sIL-6R is a functionally relevant CNS molecule. It is not clear if addition of a neutralizing antibody against sIL-6R to sIL-6R will have any effect on IL-6-induced stimulation of PGE2 synthesis where exogenous sIL-6R is not added. However, Fig. 4a, b indicates that IL-6/sIL-6R complexes stimulate PGE2 release in cultured mouse astrocytes.
Gp130, a protein associated with the IL-6/sIL-6R complexes, is a signal-transducing receptor shared by members of the IL-6 cytokine family comprising IL-6, interleukin-11, leukemia inhibitory factor, oncostatin M, ciliary neurotrophic factor, and cardiotrophin-1 (Taga and Kishimoto 1997). The JAK/STAT pathway, an important signal pathway downstream of gp130, has been implicated in diverse pathophysiological processes such as ischemia (Justicia et al. 2000; Choi et al. 2003a, b), inflammation associated with astroglial activation (Gautron et al. 2002), and seizures (Choi et al. 2003a, b; Rosell et al. 2003). Whether increases in gp130-associated STAT3 expression after such a variety of perturbations contribute to subsequent cell death or survival is not fully understood. Numerous studies demonstrate that IL-6 exerts multiple effects, both beneficial and destructive, on CNS cells. In glial cells, IL-6 promotes astrocyte proliferation and is believed to be involved in astrogliosis (Van Wagoner and Benveniste 1999). The present study provided evidence that the mouse gp130 antibody suppressed the PGE2 formation induced by IL-6 as well as IL-6/sIL-6R complexes in a dose-dependent manner (Fig. 5), suggesting that the IL-6/sIL-6R/gp130 system can modulate the function of PGE2 synthesis in astrocytes.
Although the precise mechanism underlying induction of PGE2 synthesis by IL-6 has not yet been elucidated, IL-6/sIL-6R and the signal transducer gp130 play important roles in the regulation of COX-2 expression and subsequent PGE2 formation in mouse astrocytes, and IL-6 is an important regulator of immune and inflammatory processes in the CNS.
Abbreviations
- AD:
-
Alzheimer’s disease
- ALS:
-
Amyotrophic lateral sclerosis
- bFGF:
-
Basic fibroblast growth factor
- COX:
-
Cyclooxygenase
- cPLA2 :
-
Cytosolic phospholipase A2
- MEM:
-
Minimum essential medium
- EIA:
-
Enzyme immunoassay
- FBS:
-
Fetal bovine serum
- gp130:
-
130-kDa glycoprotein
- IL-1 β:
-
Interleukin-1β
- IL-6:
-
Interleukin-6
- IL-6R:
-
Interleukin-6 receptor
- JAK:
-
Janus kinase
- LPS:
-
Lipopolysaccharide
- PBS:
-
Ca2+- and Mg2+-free phosphate-buffered saline
- PDGF:
-
Platelet-derived growth factor
- PGE2 :
-
Prostaglandin E2
- PGs:
-
Prostaglandins
- sIL-6R:
-
Soluble interleukin-6 receptor
- STAT:
-
Signal transducers and activators of transcription
- TGF-β:
-
Transforming growth factor-β
- TNF-α:
-
Tumor necrosis factor-α
- TPBS:
-
PBS containing 0.05% Tween 20
References
Almer, G., Guegan, C., Teismann, P., et al. (2001). Increased expression of the pro-inflammatory enzyme cyclooxygenase-2 in amyotrophic lateral sclerosis. Annals of Neurology, 49, 176–185. doi:10.1002/1531-8249(20010201)49:2<176::AID-ANA37>3.0.CO;2-X.
Bauer, M. K. A., Lieb, K., Schulze-Osthoff, K., et al. (1997). Expression and regulation of cyclooxygenase-2 in rat microglia. European Journal of Biochemistry, 243, 726–731. doi:10.1111/j.1432-1033.1997.00726.x.
Bezzi, P., Carmignoto, G., Pasti, L., et al. (1998). Prostaglandins stimulate calcium-dependent glutamate release in astrocytes. Nature, 391, 281–285. doi:10.1038/34651.
Bishai, I., & Coceani, F. (1992). Eicosanoid formation in the rat cerebral cortex. Molecular and Chemical Neuropathology, 17, 219–238.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254. doi:10.1016/0003-2697(76)90527-3.
Choi, J. -S., Kim, S. Y., Cha, J. -H., et al. (2003a). Upregulation of gp130 and STAT3 activation in the rat hippocampus following transient forebrain ischemia. Glia, 41, 237–246. doi:10.1002/glia.10186.
Choi, J. -S., Kim, S. Y., Park, H. -J., et al. (2003b). Upregulation of gp130 and differential activation of STAT and p42/44 MAPK in the rat hippocampus following kainic acid-induced seizures. Brain Research. Molecular Brain Research, 119, 10–18. doi:10.1016/j.molbrainres.2003.08.010.
Dayton, E. T., & Major, E. O. (1996). Recombinant human interleukin 1β induces production of prostaglandins in primary human fetal astrocytes and immortalized human fetal astrocyte cultures. Journal of Neuroimmunology, 71, 11–18. doi:10.1016/S0165-5728(96)00111-7.
DeWitt, D. L., & Smith, W. L. (1988). Primary structure of prostaglandin G/H synthase from sheep vesicular gland determined from the complementary DNA sequence. Proceedings of the National Academy of Sciences of the United States of America, 85, 1412–1416. doi:10.1073/pnas.85.5.1412.
Drachman, D. B., & Rothstein, J. D. (2000). Inhibition of cyclooxygenase-2 protects motor neurons in an organotypic model of amyotrophic lateral sclerosis. Annals of Neurology, 48, 792–795. doi:10.1002/1531-8249(200011)48:5<792::AID-ANA14>3.0.CO;2-5.
Dubois, R. N., Abramson, S. B., Crofford, L., et al. (1998). Cyclooxygenase in biology and disease. The FASEB Journal, 12, 1063–1073.
Fontana, A., Kristensen, F., Dubs, R., Gemsa, D., & Weber, E. (1982). Production of prostaglandin E and an interleukin-1 like factor by cultured astrocytes and C6 glioma cells. Journal of Immunology (Baltimore, MD.: 1950), 129, 2413–2419.
Gadient, R. A., & Otten, U. (1994). Identification of interleukin-6(IL-6)-expressing neurons in the cerebellum and hippocampus of normal adult rats. Neuroscience Letters, 182, 243–246. doi:10.1016/0304-3940(94)90807-9.
Gautron, L., Lafon, P., Chaigniau, M., Tramu, G., & Laye, S. (2002). Spatiotemporal analysis of signal transducer and activator of transcription 3 activation in rat brain astrocytes and pituitary following peripheral immune challenge. Neuroscience, 112, 717–729. doi:10.1016/S0306-4522(02)00115-X.
Hayaishi, O. (1991). Molecular mechanisms of sleep-wake regulation: Roles of prostaglandins D2 and E2. The FASEB Journal, 5, 2575–2581.
Heinrich, P. C., Behrmann, I., Muller-Newen, G., Schaper, F., & Graeve, L. (1998). Interleukin-6-type cytokine signalling through the gp130/Jak/STAT pathway. The Biochemical Journal, 334, 297–314.
Hibi, M., Murakami, M., Saito, M., Hirano, T., Taga, T., & Kishimoto, T. (1990). Molecular cloning and expression of an IL-6 signal transducer, gp130. Cell, 63, 1149–1157. doi:10.1016/0092-8674(90)90411-7.
Ishimoto, H., Nakahata, N., Matsuoka, I., & Nakanishi, H. (1997). Effects of ATP on phosphoinositide hydrolysis and prostaglandin E2 generation in rabbit astrocytes. The Journal of Pharmacy and Pharmacology, 49, 520–524.
Jacobs, T. P., Shohami, E., Baze, W., et al. (1987). Deteriorating stroke model: Histopathology, edema, and eicosanoid changes following spinal cord ischemia in rabbits. Stroke, 18, 741–750.
Justicia, C., Gabriel, C., & Planas, A. M. (2000). Activation of the JAK/STAT pathway following transient focal cerebral ischemia: Signaling through Jak1 and Stat3 in astrocytes. Glia, 30, 253–270. doi:10.1002/(SICI)1098-1136(200005)30:3<253::AID-GLIA5>3.0.CO;2-O.
Kaartinen, V., Voncken, J. W., Shuler, C., et al. (1995). Abnormal lung development and cleft palate in mice lacking TGF-β3 indicates defects of epithelial-mesenchymal interaction. Nature Genetics, 11, 415–421. doi:10.1038/ng1295-415.
Kishimoto, T., Taga, T., & Akira, S. (1994). Cytokine signal transduction. Cell, 76, 253–262. doi:10.1016/0092-8674(94)90333-6.
Kordula, T., Rydel, R. E., Brigham, E. F., Horn, F., Heinrich, P. C., & Travis, J. (1998). Oncostatin M and the interleukin-6 and soluble interleukin-6 receptor complex regulate α1-antichymotrypsin expression in human cortical astrocytes. The Journal of Biological Chemistry, 273, 4112–4118. doi:10.1074/jbc.273.7.4112.
Kudo, I., & Murakami, M. (1999). Diverse functional coupling of prostanoid biosynthetic enzymes in various cell types. Advances in Experimental Medicine and Biology, 469, 29–35.
Lin, A. H., Bienkowski, M. J., & Gorman, R. R. (1989). Regulation of prostaglandin H synthase mRNA levels and prostaglandin biosynthesis by platelet-derived growth factor. The Journal of Biological Chemistry, 264, 17379–17383.
Liu, X. -H., Kirschenbaum, A., Lu, M., et al. (2002). Prostaglandin E2 stimulates prostatic intraepithelial neoplasia cell growth through activation of the interleukin-6/gp130/Stat-3 signaling pathway. Biochemical and Biophysical Research Communications, 290, 249–255. doi:10.1006/bbrc.2001.6188.
Luo, J., Lang, J. A., & Miller, M. W. (1998). Transforming growth factor β1 regulates the expression of cyclooxygenase in cultured cortical astrocytes and neurons. Journal of Neurochemistry, 71, 526–534.
Lust, J. A., Donovan, K. A., Kline, M. P., Greipp, P. R., Kyle, R. A., & Maihle, N. J. (1992). Isolation of an mRNA encoding a soluble form of the human interleukin-6 receptor. Cytokine, 4, 96–100. doi:10.1016/1043-4666(92)90043-Q.
Marcheselli, V. L., & Bazan, N. G. (1996). Sustained induction of prostaglandin endoperoxide synthetase-2 by seizures in hippocampus. The Journal of Biological Chemistry, 271, 24794–24799. doi:10.1074/jbc.271.40.24794.
Marz, P., Cheng, J. G., Gadient, R. A., et al. (1998). Sympathetic neurons can produce and respond to interleukin 6. Proceedings of the National Academy of Sciences of the United States of America, 95, 3251–3256. doi:10.1073/pnas.95.6.3251.
McGeer, P. L., McGeer, E., Rogers, J., & Sibley, J. (1990). Anti-inflammatory drugs and Alzheimer disease. Lancet, 335, 1037. doi:10.1016/0140-6736(90)91101-F.
Minghetti, L., & Levi, G. (1995). Induction of prostanoid biosynthesis by bacterial lipopolysaccharide and isoproterenol in rat microglial cultures. Journal of Neurochemistry, 65, 2690–2698.
Minghetti, L., Walsh, D. T., Levi, G., & Perry, V. H. (1999). In vivo expression of cyclooxygenase-2 in rat brain following intraparenchymal injection of bacterial endotoxin and inflammatory cytokines. Journal of Neuropathology and Experimental Neurology, 58, 1184–1191. doi:10.1097/00005072-199911000-00008.
Molina-Holgado, E., Ortiz, S., Molina-Holgado, F., & Guaza, C. (2000). Induction of COX-2 and PGE2 biosynthesis by IL-1β is mediated by PKC and mitogen-activated protein kinases in murine astrocytes. British Journal of Pharmacology, 131, 152–159. doi:10.1038/sj.bjp.0703557.
Mollace, V., Colasanti, M., Muscoli, C., et al. (1998). The effect of nitric oxide on cytokine-induced release of PGE2 by human cultured astroglial cells. British Journal of Pharmacology, 124, 742–746. doi:10.1038/sj.bjp.0701852.
Montine, T. J., Sidell, K. R., Crews, B. C., et al. (1999). Elevated CSF prostaglandin E2 levels in patients with probable AD. Neurology, 53, 1495–1498.
Morham, S. G., Langenbach, R., Loftin, C. D., et al. (1995). Prostaglandin synthase 2 gene disruption causes severe renal pathology in the mouse. Cell, 83, 473–482. doi:10.1016/0092-8674(95)90125-6.
Mullberg, J., Schooltink, H., Stoyan, T., et al. (1993). The soluble interleukin-6 receptor is generated by shedding. European Journal of Immunology, 23, 473–480. doi:10.1002/eji.1830230226.
Mullberg, J., Oberthur, W., Lottspeich, F., et al. (1994). The soluble human IL-6 receptor: Mutational characterization of the proteolytic cleavage site. Journal of Immunology (Baltimore, MD.: 1950), 152, 4958–4968.
Nogawa, S., Zhang, F., Ross, M. E., & Iadecola, C. (1997). Cyclo-oxygenase-2 gene expression in neurons contributes to ischemic brain damage. The Journal of Neuroscience, 17, 2746–2755.
O’Banion, M. K., Winn, V. D., & Young, D. A. (1992). cDNA cloning and functional activity of a glucocorticoid-regulated inflammatory cyclooxygenase. Proceedings of the National Academy of Sciences of the United States of America, 89, 4888–4892. doi:10.1073/pnas.89.11.4888.
O’Banion, M. K., Miller, J. C., Chang, J. W., Kaplan, M. D., & Coleman, P. D. (1996). Interleukin-1β induces prostaglandin G/H synthase-2 (cyclooxygenase-2) in primary murine astrocyte cultures. Journal of Neurochemistry, 66, 2532–2540.
Oh, J. -W., Wagoner, N. J. V., Rose-John, S., & Benveniste, E. N. (1998). Role of IL-6 and the soluble IL-6 receptor in inhibition of VCAM-1 gene expression. Journal of Immunology (Baltimore, MD.: 1950), 161, 4992–4999.
Pasti, L., Zonta, M., Pozzan, T., Vicini, S., & Carmignoto, G. (2001). Cytosolic calcium oscillations in astrocytes may regulate exocytotic release of glutamate. The Journal of Neuroscience, 21, 477–484.
Peters, M., Muller, A. M., & Rose-John, S. (1998). Interleukin-6 and soluble interleukin-6 receptor: direct stimulation of gp130 and hematopoiesis. Blood, 92, 3495–3504.
Pilbeam, C. C., Kawaguchi, H., Hakeda, Y., Voznesensky, O., Alander, C. B., & Raisz, L. G. (1993). Differential regulation of inducible and constitutive prostaglandin endoperoxide synthase in osteoblastic MC3T3-E1 cells. The Journal of Biological Chemistry, 268, 25643–25649.
Pistritto, G., Mancuso, C., Tringali, G., Perretti, M., Preziosi, P., & Navarra, P. (1998). The relative contribution of constitutive and inducible cyclooxygenase activity to lipopolysaccharide-induced prostaglandin production by primary cultures of rat hypothalamic astrocytes. Neuroscience Letters, 246, 45–48. doi:10.1016/S0304-3940(98)00226-2.
Pistritto, G., Franzese, O., Pozzoli, G., et al. (1999). Bacterial lipopolysaccharide increases prostaglandin production by rat astrocytes via inducible cyclo-oxygenase: Evidence for the involvement of nuclear factor κB. Biochemical and Biophysical Research Communications, 263, 570–574. doi:10.1006/bbrc.1999.1413.
Pritchard, K. A., O’Banion, M. K., Miano, J. M., et al. (1994). Induction of cyclooxygenase-2 in rat vascular smooth muscle cells in vitro and in vivo. The Journal of Biological Chemistry, 269, 8504–8509.
Repovic, P., Mi, K., & Benveniste, E. N. (2003). Oncostatin M enhances the expression of prostaglandin E2 and cyclooxygenase-2 in astrocytes: Synergy with interleukin-1β, tumor necrosis factor-α, and bacterial lipopolysaccharide. Glia, 42, 433–446. doi:10.1002/glia.10182.
Rose-John, S., & Heinrich, P. C. (1994). Soluble receptors for cytokines and growth factors: generation and biological function. The Biochemical Journal, 300, 281–290.
Rosell, D. R., Akama, K. T., Nacher, J., & McEwen, B. S. (2003). Differential expression of suppressors of cytokine signaling-1, -2, and -3 in the rat hippocampus after seizure: Implications for neuromodulation by gp130 cytokines. Neuroscience, 122, 349–358. doi:10.1016/S0306-4522(03)00594-3.
Saas, P., Boucraut, J., Walker, P. R., et al. (2000). TWEAK stimulation of astrocytes and the proinflammatory consequences. Glia, 32, 102–107. doi:10.1002/1098-1136(200010)32:1<102::AID-GLIA100>3.0.CO;2-U.
Samad, T. A., Moore, K. A., Sapirstein, A., et al. (2001). Interleukin 1β-mediated induction of cox-2 in the CNS contributes to inflammatory pain hypersensitivity. Nature, 410, 471–475. doi:10.1038/35068566.
Sawada, M., Suzumura, A., Ohno, K., & Marunouchi, T. (1993). Regulation of astrocyte proliferation by prostaglandin E2 and the α subtype of protein kinase C. Brain Research, 613, 67–73. doi:10.1016/0006-8993(93)90455-V.
Sawada, M., Suzumura, A., & Marunouchi, T. (1995). Cytokine network in the central nervous system and its roles in growth and differentiation of glial and neuronal cells. International Journal of Developmental Neuroscience, 13, 253–264. doi:10.1016/0736-5748(94)00076-F.
Seregi, A., Keller, M., Jackisch, R., & Hertting, G. (1984). Comparison of the prostanoid synthesizing capacity in homogenates from primary neuronal and astroglial cell cultures. Biochemical Pharmacology, 33, 3315–3318. doi:10.1016/0006-2952(84)90099-6.
Smith, W. L., Garavito, R. M., & DeWitt, D. L. (1996). Prostaglandin endoperoxide H synthases(cyclooxygenases)-1 and -2. The Journal of Biological Chemistry, 271, 33157–33160. doi:10.1074/jbc.271.52.33157.
Smith, W. L., DeWitt, D. L., & Garavito, R. M. (2000). Cyclooxygenases: Structural, cellular, and molecular biology. Annual Review of Biochemistry, 69, 145–182. doi:10.1146/annurev.biochem.69.1.145.
Stephenson, D. T., Lemere, C. A., Selkoe, D. J., & Clemens, J. A. (1996). Cytosolic phospholipase A2(cPLA2) immunoreactivity is elevated in Alzheimer’s disease brain. Neurobiology of Disease, 3, 51–63. doi:10.1006/nbdi.1996.0005.
Taga, T., & Kishimoto, T. (1997). Gp130 and the interleukin-6 family of cytokines. Annual Review of Immunology, 15, 797–819. doi:10.1146/annurev.immunol.15.1.797.
Teather, L. A., Lee, R. K. K., & Wurtman, R. J. (2002). Platelet-activating factor increases prostaglandin E2 release from astrocyte-enriched cortical cell cultures. Brain Research, 946, 87–95. doi:10.1016/S0006-8993(02)02866-4.
Their, M., Marz, P., Otten, U., Weis, J., & Rose-John, S. (1999). Interleukin-6(IL-6) and its soluble receptor support survival of sensory neurons. Journal of Neuroscience Research, 55, 411–422. doi:10.1002/(SICI)1097-4547(19990215)55:4<411::AID-JNR2>3.0.CO;2-D.
Ushikubi, F., Segi, E., Sugimoto, Y., et al. (1998). Impaired febrile response in mice lacking the prostaglandin E receptor subtype EP3. Nature, 395, 281–284. doi:10.1038/26233.
Van Wagoner, N. J., & Benveniste, E. N. (1999). Interleukin-6 expression and regulation in astrocytes. Journal of Neuroimmunology, 100, 124–139. doi:10.1016/S0165-5728(99)00187-3.
Vane, J. R., Bakhle, Y. S., & Botting, R. M. (1998). Cyclooxygenases 1 and 2. Annual Review of Pharmacology and Toxicology, 38, 97–120. doi:10.1146/annurev.pharmtox.38.1.97.
Xie, W., Chipman, J. G., Robertson, D. L., Erikson, R. L., & Simmons, D. L. (1991). Expression of a mitogen-responsive gene encoding prostaglandin synthase is regulated by mRNA splicing. Proceedings of the National Academy of Sciences of the United States of America, 88, 2692–2696. doi:10.1073/pnas.88.7.2692.
Xu, J., Chalimoniuk, M., Shu, Y., et al. (2003). Prostaglandin E2 production in astrocytes: Regulation by cytokines, extracellular ATP, and oxidative agents. Prostaglandins, Leukotrienes, and Essential Fatty Acids, 69, 437–448. doi:10.1016/j.plefa.2003.08.016.
Yamagata, K., Andreasson, K. I., Kaufmann, W. E., Barnes, C. A., & Worley, P. F. (1993). Expression of a mitogen-inducible cyclooxygenase in brain neurons: Regulation by synaptic activity and glucocorticoids. Neuron, 11, 371–386.
Acknowledgments
We are very grateful to Dr. Akira Tanaka for his valuable and helpful advice on the experiments and manuscript preparation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chikuma, T., Yoshimoto, T., Ohba, M. et al. Interleukin-6 Induces Prostaglandin E2 Synthesis in Mouse Astrocytes. J Mol Neurosci 39, 175–184 (2009). https://doi.org/10.1007/s12031-009-9187-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-009-9187-6