Abstract
Background
Following aneurysmal subarachnoid hemorrhage (SAH), patients are monitored closely for vasospasm in the intensive care unit. Conditional vasospasm-free survival describes the risk of future vasospasm as a function of time elapsed without vasospasm. Conditional survival has not been applied to this clinical scenario but could improve patient counseling and intensive care unit use. The objective of this study was to characterize conditional vasospasm-free survival following SAH.
Methods
This was a single institution, retrospective cohort study of patients treated for aneurysmal SAH between 1/1/2000–6/1/2020. The primary outcome was the development of vasospasm defined by the first instance of either radiographic vasospasm on computed tomography angiography, Lindegaard Index > 3.0 by transcranial doppler ultrasonography, or vasospasm-specific intraarterial therapy. Multivariable Cox regression was performed, and conditional vasospasm-free survival curves were constructed.
Results
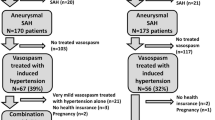
A total of 528 patients were treated for aneurysmal SAH and 309 (58.5%) developed vasospasm. Conditional survival curves suggest patients who survive to postbleed day 10 without vasospasm have a nearly 90% chance of being discharged without vasospasm. The median onset of vasospasm was postbleed day 6. Age more than 50 years was associated with a lower risk (hazard ratio [HR] = .76; 95% confidence interval [CI] 0.64–0.91; p < 0.001). Higher initial systolic blood pressure (HR = 1.18; 95% CI 1.046–1.350; p = .008), Hunt-Hess grades 4 or 5 (HR = 1.304; 95% CI 1.014–1.676), and modified Fisher scale score of 4 (HR = 1.808; 95% CI 1.198–2.728) were associated with higher vasospasm than the respective lower grades.
Conclusion
Conditional survival provides a useful framework for counseling patients and making decisions around vasospasm risk for patients with aneurysmal SAH, while risk factor-stratified plots facilitate a patient-centric, evidence-based approach to these conversations and decisions.



Similar content being viewed by others
References
Kassell NF, Sasaki T, Colohan AR, Nazar G. Cerebral vasospasm following aneurysmal subarachnoid hemorrhage. Stroke. 1985;16:562–72.
Weir B, Grace M, Hansen J, Rothberg C. Time course of vasospasm in man. J Neurosurg. 1978;48:173–8.
Kassell NF, Torner JC, Haley EC Jr, Jane JA, Adams HP, Kongable GL. The International cooperative study on the timing of aneurysm surgery. Part 1: overall management results. J Neurosurg. 1990;73:18–36.
Kassell NF, Torner JC, Jane JA, Haley EC Jr, Adams HP. The international cooperative study on the timing of aneurysm surgery. Part 2: surgical results. J Neurosurg. 1990;73:37–47.
Hieke S, Kleber M, König C, Engelhardt M, Schumacher M. Conditional survival: a useful concept to provide information on how prognosis evolves over time. Clin Cancer Res. 2015;21:1530–6.
Roden DM, Pulley JM, Basford MA, Bernard GR, Clayton EW, Balser JR, et al. Development of a large-scale de-identified DNA biobank to enable personalized medicine. Clin Pharmacol Ther. 2008;84:362–9.
Hunt WE, Hess RM. Surgical risk as related to time of intervention in the repair of intracranial aneurysms. J Neurosurg. 1968;28:14–20.
Lindegaard KF, Nornes H, Bakke SJ, Sorteberg W, Nakstad P. Cerebral vasospasm after subarachnoid haemorrhage investigated by means of transcranial Doppler ultrasound. Acta Neurochir Suppl. 1988;42:81–4.
Hosmann A, Klenk S, Wang WT, Koren J, Sljivic S, Reinprecht A. Endogenous arterial blood pressure increase after aneurysmal subarachnoid hemorrhage. Clin Neurol Neurosurg. 2020;190:105639.
Inagawa T. Risk factors for cerebral vasospasm following aneurysmal subarachnoid hemorrhage: a review of the literature. World Neurosurg. 2016;85:56–76.
Frontera JA, Claassen J, Schmidt JM, Wartenberg KE, Temes R, Connolly ES Jr, et al. Prediction of symptomatic vasospasm after subarachnoid hemorrhage: the modified fisher scale. Neurosurgery. 2006;59:21–7.
Rumalla K, Lin M, Ding L, Gaddis M, Giannotta SL, Attenello FJ, et al. Risk factors for cerebral vasospasm in aneurysmal subarachnoid hemorrhage: a population-based study of 8346 patients. World Neurosurg. 2021;145:e233–41.
Fisher CM, Kistler JP, Davis JM. Relation of cerebral vasospasm to subarachnoid hemorrhage visualized by computerized tomographic scanning. Neurosurgery. 1980;6:1–9.
Frontera JA, Fernandez A, Schmidt JM, Claassen J, Wartenberg KE, Badjatia N, et al. Defining vasospasm after subarachnoid hemorrhage: what is the most clinically relevant definition? Stroke. 2009;40:1963–8.
Charpentier C, Audibert G, Guillemin F, Civit T, Ducrocq X, Bracard S, et al. Multivariate analysis of predictors of cerebral vasospasm occurrence after aneurysmal subarachnoid hemorrhage. Stroke. 1999;30:1402–8.
Mertens AC, Yong J, Dietz AC, Kreiter E, Yasui Y, Bleyer A, et al. Conditional survival in pediatric malignancies: analysis of data from the childhood cancer survivor study and the surveillance, epidemiology, and end results program. Cancer. 2015;121:1108–17.
Zamboni BA, Yothers G, Choi M, Fuller CD, Dignam JJ, Raich PC, et al. Conditional survival and the choice of conditioning set for patients with colon cancer: an analysis of NSABP trials C-03 through C-07. J Clin Oncol. 2010;28:2544–8.
Zabor EC, Gonen M, Chapman PB, Panageas KS. Dynamic prognostication using conditional survival estimates. Cancer. 2013;119:3589–92.
Haydu LE, Scolyer RA, Lo S, Quinn MJ, Saw RPM, Shannon KF, et al. Conditional survival: an assessment of the prognosis of patients at time points after initial diagnosis and treatment of locoregional melanoma metastasis. J Clin Oncol. 2017;35:1721–9.
Porter KR, McCarthy BJ, Berbaum ML, Davis FG. Conditional survival of all primary brain tumor patients by age, behavior, and histology. Neuroepidemiology. 2011;36:230–9.
Davis FG, McCarthy BJ, Freels S, Kupelian V, Bondy ML. The conditional probability of survival of patients with primary malignant brain tumors: surveillance, epidemiology, and end results (SEER) data. Cancer. 1999;85:485–91.
Farah P, Blanda R, Kromer C, Ostrom QT, Kruchko C, Barnholtz-Sloan JS. Conditional survival after diagnosis with malignant brain and central nervous system tumor in the United States, 1995–2012. J Neurooncol. 2016;128:419–29.
Marshall DC, Hatch RA, Gerry S, Young JD, Watkinson P. Conditional survival with increasing duration of ICU admission: an observational study of three intensive care databases. Crit Care Med. 2020;48:91–7.
Macdonald RL, Pluta RM, Zhang JH. Cerebral vasospasm after subarachnoid hemorrhage: the emerging revolution. Nat Clin Pract Neurol. 2007;3:256–63.
Diringer MN, Bleck TP, Claude Hemphill J, Menon D, Shutter L, Vespa P, et al. Critical care management of patients following aneurysmal subarachnoid hemorrhage: recommendations from the Neurocritical Care Society’s multidisciplinary consensus conference. Neurocritic Care. 2011;15:211–40.
Chartrain AG, Awad AJ, Sarkiss CA, Feng R, Liu Y, Mocco J, et al. A step-down unit transfer protocol for low-risk aneurysmal subarachnoid hemorrhage. Neurosurg Focus. 2017;43:E15.
Collins CI, Hasan TF, Mooney LH, Talbot JL, Fouraker AL, Nelson KF, et al. Subarachnoid hemorrhage “fast track”: a health economics and health care redesign approach for early selected hospital discharge. Mayo Clin Proc Innov Qual Outcomes. 2020;4:238–48.
Inouye SK. Delirium in older persons. N Engl J Med. 2006;354:1157–65.
Foreman PM, Chua MH, Harrigan MR, Fisher WS 3rd, Tubbs RS, Shoja MM, et al. External validation of the Practical Risk Chart for the prediction of delayed cerebral ischemia following aneurysmal subarachnoid hemorrhage. J Neurosurg. 2017;126:1530–6.
Lee H, Perry JJ, English SW, Alkherayf F, Joseph J, Nobile S, et al. Clinical prediction of delayed cerebral ischemia in aneurysmal subarachnoid hemorrhage. J Neurosurg. 2018;2018:1–8.
Darkwah Oppong M, Iannaccone A, Gembruch O, Pierscianek D, Chihi M, Dammann P, et al. Vasospasm-related complications after subarachnoid hemorrhage: the role of patients’ age and sex. Acta Neurochir. 2018;160:1393–400.
Dennis J, Yengo-Kahn AM, Kirby P, Solomon GS, Cox NJ, Zuckerman SL. Diagnostic algorithms to study post-concussion syndrome using electronic health records: validating a method to capture an important patient population. J Neurotrauma. 2019;36:2167–77.
Abdellaoui A, Sanchez-Roige S, Sealock J, Treur JL, Dennis J, Fontanillas P, et al. Phenome-wide investigation of health outcomes associated with genetic predisposition to loneliness. Hum Mol Genet. 2019;28:3853–65.
Wise ES, Ladner TR, Song J, Eagle SS, Mocco J, Wergin JE, et al. Race as a predictor of delay from diagnosis to endarterectomy in clinically significant carotid stenosis. J Vasc Surg. 2015;62:49–56.
Yang BQ, Assad TR, O’Leary JM, Xu M, Halliday SJ, D’Amico RW, et al. Racial differences in patients referred for right heart catheterization and risk of pulmonary hypertension. Pulm Circ. 2018;8:2045894018764273.
Venkatraman A, Khawaja AM, Gupta S, Hardas S, Deveikis JP, Harrigan MR, et al. Intra-arterial vasodilators for vasospasm following aneurysmal subarachnoid hemorrhage: a meta-analysis. J Neurointerv Surg. 2018;10:380–7.
Li K, Barras CD, Chandra RV, Kok HK, Maingard JT, Carter NS, et al. A review of the management of cerebral vasospasm after aneurysmal subarachnoid hemorrhage. World Neurosurg. 2019;126:513–27.
Vergouwen MDI, Vermeulen M, van Gijn J, Rinkel GJE, Wijdicks EF, Muizelaar JP, et al. Definition of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage as an outcome event in clinical trials and observational studies: proposal of a multidisciplinary research group. Stroke. 2010;41:2391–5.
Dankbaar JW, Rijsdijk M, van der Schaaf IC, Velthuis BK, Wermer MJH, Rinkel GJE. Relationship between vasospasm, cerebral perfusion, and delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage. Neuroradiology. 2009;51:813–9.
Funding
REDCap, used for study data management, is supported by NCATS/National Institutes of Health grant UL1 TR000445. One author (PDK) is supported by a training grant from the National Cancer Institute of the National Institutes of Health under award number T32CA106183.
Author information
Authors and Affiliations
Contributions
PDK: design of the work, analysis and interpretation of data, and drafting the work. AMYK: design of the work, analysis and interpretation of data, and drafting the work. ART: acquisition of data and critically revising the intellectual content. SVJ: acquisition of data and critically revising the intellectual content. RAR: acquisition and interpretation of data and critically revising the intellectual content. FY: analysis and interpretation of data and critically revising the intellectual content. ZZ: analysis and interpretation of data and critically revising the intellectual content. MTF: conception and design of the work, interpretation of the data, and critically revising the content. MRF: conception and design of the work, interpretation of the data, and critically revising the content. All authors approve of the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no conflicts of interest to disclose.
Ethical Approval
Researchers adhered to ethical guidelines; this study was approved by the local institutional review board.
Informed Consent
Requirement for informed consent was waived as data are anonymous and did not contain any Health Insurance Portability and Accountability Act identifiers.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kelly, P.D., Yengo-Kahn, A.M., Tang, A.R. et al. Conditional Vasospasm-Free Survival Following Aneurysmal Subarachnoid Hemorrhage. Neurocrit Care 37, 81–90 (2022). https://doi.org/10.1007/s12028-022-01444-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-022-01444-z