Abstract
Background
Cortical spreading depolarization (SD) is a propagating depolarization wave of neurons and glial cells in the cerebral gray matter. SD occurs in all forms of severe acute brain injury, as documented by using invasive detection methods. Based on many experimental studies of mechanical brain deformation and concussion, the occurrence of SDs in human concussion has often been hypothesized. However, this hypothesis cannot be confirmed in humans, as SDs can only be detected with invasive detection methods that would require either a craniotomy or a burr hole to be performed on athletes. Typical electroencephalography electrodes, placed on the scalp, can help detect the possible presence of SD but have not been able to accurately and reliably identify SDs.
Methods
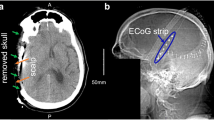
To explore the possibility of a noninvasive method to resolve this hurdle, we developed a finite element numerical model that simulates scalp voltage changes that are induced by a brain surface SD. We then compared our simulation results with retrospectively evaluated data in patients with aneurysmal subarachnoid hemorrhage from Drenckhahn et al. (Brain 135:853, 2012).
Results
The ratio of peak scalp to simulated peak cortical voltage, Vscalp/Vcortex, was 0.0735, whereas the ratio from the retrospectively evaluated data was 0.0316 (0.0221, 0.0527) (median [1st quartile, 3rd quartile], n = 161, p < 0.001, one sample Wilcoxon signed-rank test). These differing values provide validation because their differences can be attributed to differences in shape between concussive SDs and aneurysmal subarachnoid hemorrhage SDs, as well as the inherent limitations in human study voltage measurements. This simulated scalp surface potential was used to design a virtual scalp detection array. Error analysis and visual reconstruction showed that 1 cm is the optimal electrode spacing to visually identify the propagating scalp voltage from a cortical SD. Electrode spacings of 2 cm and above produce distorted images and high errors in the reconstructed image.
Conclusions
Our analysis suggests that concussive (and other) SDs can be detected from the scalp, which could confirm SD occurrence in human concussion, provide concussion diagnosis on the basis of an underlying physiological mechanism, and lead to noninvasive SD detection in the setting of severe acute brain injury






Similar content being viewed by others
References
Langlois JA, Rutland-Brown W, Wald MM. The epidemiology and impact of traumatic brain injury: a brief overview. J Head Trauma Rehabil. 2006;21:375–8.
Coronado VG, McGuire LC, Sarmiento K, et al. Trends in Traumatic Brain Injury in the U.S. and the public health response: 1995–2009. J Safety Res. 2012;43:299–307.
Lefevre-Dognin C, Cogné M, Perdrieau V, Granger A, Heslot C, Azouvi P. Definition and epidemiology of mild traumatic brain injury. Neurochirurgie. 2020;67:218–21.
Mansell JL, Tierney RT, Higgins M, McDevitt J, Toone N, Glutting J. Concussive signs and symptoms following head impacts in collegiate athletes. Brain Inj. 2010;24:1070–4.
Costello DM, Kaye AH, O’Brien TJ, Shultz SR. Sport related concussion: potential for biomarkers to improve acute management. J Clin Neurosci. 2018;56:1–6.
Davis GA, Ellenbogen RG, Bailes J, et al. The Berlin International Consensus meeting on concussion in sport. Neurosurgery. 2018;82:232–6.
Master CL, Mayer AR, Quinn D, Grady MF. Concussion. Ann Intern Med. 2018;169:ITC1-ITC16.
Provencher MT, Frank RM, Shubert DJ, Sanchez A, Murphy CP, Zafonte RD. Concussions in sports. Orthopedics. 2019;42:12–21.
Hobbs JG, Young JS, Bailes JE. Sports-related concussions: diagnosis, complications, and current management strategies. Neurosurg Focus. 2016;40:E5: https://doi.org/10.3171/2016.1.FOCUS15617.
Shively SB, Perl DP. Traumatic brain injury, shell shock, and posttraumatic stress disorder in the military–past, present, and future. J Head Trauma Rehabil. 2012;27:234–9.
Wells TS, Miller SC, Adler AB, Engel CC, Smith TC, Fairbank JA. Mental health impact of the Iraq and Afghanistan conflicts: a review of US research, service provision, and programmatic responses. Int Rev Psychiatry. 2011;23:144–52.
Guskiewicz KM, Marshall SW, Bailes J, et al. Association between recurrent concussion and late-life cognitive impairment in retired professional football players. Neurosurgery. 2005;57:719–26.
Harmon KG, Drezner JA, Gammons M, et al. American Medical Society for sports medicine position statement: concussion in sport. Br J Sports Med. 2013;47:15–26.
Bruce J, Echemendia R, Meeuwisse W, Comper P, Sisco A. 1 year test-retest reliability of ImPACT in professional ice hockey players. Clin Neurophysiol. 2013;28:14–25.
Lichtenstein JD, Moser RS, Schatz P. Age and test setting affect the prevalence of invalid baseline scores on neurocognitive tests. Am J Sports Med. 2013;42:479–84.
Resch J, Driscoll A, McCaffrey N, et al. ImPact test-retest reliability: reliably unreliable? J Athl Train. 2013;48:506–11.
Barlow M, Schlabach D, Peiffer J, Cook C. Differences in change scores and the predictive validity of three commonly used measures following concussion in the middle school and high school aged population. Inter J Sports Phys Ther. 2011;6:150–7.
McClure DJ, Zuckerman SL, Kutscher SJ, Gregory AJ, Solomon GS. Baseline neurocognitive testing in sports-related concussions: the importance of a Prior Night’s sleep. Am J Sports Med. 2014;42:472–8.
Cole WR, Arrieux JP, Schwab K, Ivins BJ, Qashu FM, Lewis SC. Test-retest reliability of four computerized neurocognitive assessment tools in an active duty military population. Arch Clin Neuropsychol. 2013;28:732–42.
Thiagarajan P, Ciuffreda KJ, Ludlam DP. Vergence dysfunction in mild traumatic brain injury (mTBI): a review. Ophthalmic Physiol Opt. 2011;31:456–68.
Echemendia RJ, Putukian M, Mackin RS, Julian L, Shoss N. Neuropsychological test performance prior to and following sports-related mild traumatic brain injury. Clin J Sport Med. 2001;11:23–31.
Echemendia RJ, Meeuwisse W, McCrory P, et al. The Sport Concussion Assessment Tool 5th Edition (SCAT5): Background and rationale. Br J Sports Med. 2017;51:848–850.
Ochiai H, Abe T. Clinical features and early detection of sport-related concussion. Acute Med Surg. 2019;6:49–53.
Broglio SP, Cantu RC, Gioia GA, et al. National Athletic Trainers’ Association position statement: management of sport concussion. J Athl Train. 2014;49:245–65.
Somjen GG. Mechanisms of spreading depression and hypoxic spreading depression-like depolarization. Physiol Rev. 2001;81:1065–96.
Pacheco JM, Hines-Lanham A, Stratton C, et al. Spreading depolarizations occur in mild traumatic brain injuries and are associated with postinjury behavior. eNeuro. 2019;6:1–17.
Bouley J, Chung DY, Ayata CY, Brown RH Jr, Henninger N. Cortical spreading depression denotes concussion injury. J Neurotrauma. 2019;36:1008–17.
Smith DH, Stewart W. “Concussion” is not a true diagnosis. Nat Rev Neurol. 2020;16:457–8.
McCuddy WT, España LY, Nelson LD, Birn RM, Mayer AR, Meier TB. Association of acute depressive symptoms and functional connectivity of emotional processing regions following sport-related concussion. Neuroimage Clin. 2018;19:434–42.
O’Jile JR, Ryan LM, Betz B, et al. Information processing following mild head injury. Arch Clin Neuropsychol. 2006;21:293–6.
Leao AAP. Spreading depression of activity in the cerebral cortex. J Neurophysiol. 1944;7:359–90.
Reid KH, Marrannes R, Wauquier A. Spreading depression and central nervous system pharmacology. J Pharmacol Methods. 1988;19:1–21.
Leao AA. The slow voltage variation of cortical spreading depression of activity. Electroencephalogr Clin Neurophysiol. 1951;3:315–21.
Drenckhahn C, Winkler MK, Major S, et al. Correlates of spreading depolarization in human scalp electroencephalography. Brain. 2012;135:853–68.
Lauritzen M, Dreier JP, Fabricius M, Hartings JA, Graf R, Strong AJ. Clinical relevance of cortical spreading depression in neurological disorders: migraine, malignant stroke, subarachnoid and intracranial hemorrhage, and traumatic brain injury. J Cereb Blood Flow Metab. 2011;31:17–35.
Strong AJ, Fabricius M, Boutelle MG, et al. Spreading and synchronous depressions of cortical activity in acutely injured human brain. Stroke. 2002;33:2738–43.
Leao AAP. Further observations on the speading depression of activity in the cerebral cortex. J Neurophysiol. 1947;10:409–14.
Kraig RP, Nicholson C. Extracellular ionic variations during spreading depression. Neuroscience. 1978;3:1045–59.
Dreier JP. The role of spreading depression, spreading depolarization and spreading ischemia in neurological disease. Nat Med. 2011;17:439–47.
Enger R, Tang W, Vindedal GF, et al. Dynamics of ionic shifts in cortical spreading depression. Cereb Cortex. 2015;25:4469–76.
Woitzik J, Hecht N, Pinczolits A, et al. Propagation of cortical spreading depolarization in the human cortex after malignant stroke. Neurology. 2013;80:1095–102.
Grafstein B. Locus of propagation of spreading cortical depression. J Neurophysiol. 1956;19:308–16.
Mayevsky A, Doron A, Manor T, Meilin S, Zarchin N, Ouaknine GE. Cortical spreading depression recorded from the human brain using a multiparametric monitoring system. Brain Res. 1996;740:268–74.
Dohmen C, Sakowitz OW, Fabricius M, et al. Delayed secondary phase of peri-infarct depolarizations after focal cerebral ischemia: relation to infarct growth and neuroprotection. Ann Neurol. 2008;63:720–8.
Dreier JP, Woitzik J, Fabricius M, et al. Delayed ischaemic neurological deficits after subarachnoid haemorrhage are associated with clusters of spreading depolarizations. Brain. 2006;129:3224–37.
Dreier JP, Major S, Manning A, et al. Cortical spreading ischaemia is a novel process involved in ischaemic damage in patients with aneurysmal subarachnoid haemorrhage. Brain. 2009;132:1866–81.
Fabricius M, Fuhr S, Bhatia R, et al. Cortical spreading depression and peri-infarct depolarization in acutely injured human cerebral cortex. Brain. 2006;129:778–90.
Hartings JA, Bullock MR, Okonkwo DO, et al. Spreading depolarisations and outcome after traumatic brain injury: a prospective observational study. Lancet Neurol. 2011;10:1058–64.
Helbok R, Schiefecker AJ, Friberg C, et al. Spreading depolarizations in patients with spontaneous intracerebral hemorrhage: association with perihematomal edema progression. J Cereb Blood Flow Metab. 2017;37:1871–82.
Dreier JP, Reiffurth C. The stroke-migraine depolarization continuum. Neuron. 2015;86:902–22.
Hartings JA, Shuttleworth CW, Kirov SA, et al. The continuum of spreading depolarizations in acute cortical lesion development: examining Leao’s legacy. J Cereb Blood Flow Metab. 2017;37:1571–94.
Oliveira-Ferreira AI, Milakara D, Alam M, et al. Experimental and preliminary clinical evidence of an ischemic zone with prolonged negative DC shifts surrounded by a normally perfused tissue belt with persistent electrocorticographic depression. J Cereb Blood Flow Metab. 2010;30:1504–19.
Hartings JA, York J, Carroll CP, et al. Subarachnoid blood acutely induces spreading depolarizations and early cortical infarction. Brain. 2017;140:2673–90.
Luckl J, Lemale CL, Kola V, et al. The negative ultraslow potential, electrophysiological correlate of infarction in the human cortex. Brain. 2018;141:1734–52.
Dreier JP, Major S, Foreman B, et al. Terminal spreading depolarization and electrical silence in death of human cerebral cortex. Ann Neurol. 2018;83:295–310.
Dreier JP, Major S, Lemale CL, et al. Correlates of spreading depolarization, spreading depression, and negative ultraslow potential in epidural versus subdural electrocorticography. Front Neurosci. 2019;13:373.
Carlson AP, Shuttleworth CW, Major S, Lemale CL, Dreier JP, Hartings JA. Terminal spreading depolarizations causing electrocortical silencing prior to clinical brain death: case report. J Neurosurg. 2018;131:1773–9.
Menyhárt Á, Frank R, Farkas AE, et al. Malignant astrocyte swelling and impaired glutamate clearance drive the expansion of injurious spreading depolarization foci. J Cereb Blood Flow Metab. 2021;Online ahead eprint.
Hadjikhani N, Sanchez Del Rio M, Wu O, et al. Mechanisms of migraine aura revealed by functional MRI in human visual cortex. Proc Natl Acad Sci USA. 2001;98:4687–92.
Major S, Huo S, Lemale CL, et al. Direct electrophysiological evidence that spreading depolarization-induced spreading depression is the pathophysiological correlate of the migraine aura and a review of the spreading depolarization continuum of acute neuronal mass injury. Geroscience. 2020;42:57–80.
Kumagai T, Walberer M, Nakamura H, et al. Distinct spatiotemporal patterns of spreading depolarizations during early infarct evolution: evidence from real-time imaging. J Cereb Blood Flow Metab. 2011;31:580–92.
Zachar J. Zacharova D [Mechanical energy as causative agent of spreading depression]. Cesk Fysiol. 1958;7:189–90.
Zachar J, Zacharova D. [Mechanism of the origin of spreading cortical depression] (in Slovak). Lek Pr. 1963;3:3–110.
Zachar J, Zacharova D. Subthreshold changes at the site of initiation of spreading cortical depression by mechanical stimuli. Electroencephalogr Clin Neurophysiol. 1961;13:896–904.
Bures J, Buresova O, Krivanek J. Initiation of Spreading Depression: Mechanical Stimuli. The Mechanism and Application of Leao's Spreading Depression of Electroencephalographic Activity. Prague: Academia; 1974:26–30.
Shah KR, West M. The effect of concussion on cerebral uptake of 2-deoxy-D-glucose in rat. Neurosci Lett. 1983;40:287–91.
Takahashi H, Manaka S, Sano K. Changes in extracellular potassium concentration in cortex and brain stem during the acute phase of experimental closed head injury. J Neurosurg. 1981;55:708–17.
West M, Parkinson D, Havlicek V. Spectral analysis of the electroencephalographic response in experimental concussion in the rat. Electroencephalogr Clin Neurophysiol. 1982;53:192–200.
Meyer JS, Denny-Brown D. Studies of cerebral circulation in brain injury. II Cerebral concussion. Electroencephalogr Clin Neurophysiol. 1955;7:529–44.
Watanabe N, Noriaki W. The mechanism and pathophysiology of rat cerebral concussion: changes in rCBF, ECoG, SPC following skull impact. J Med Soc Toho Univ. 2002;49:23–30.
Oka H, Kako M, Matsushima M, Ando K. Specific type of head injury in children. Report of 5 cases. Brain. 1977;100:287–98.
Takahashi H, Nakazawa S. Specific type of head injury in children. Report of 5 cases. Childs Brain. 1980;7:124–31.
Barkhoudarian G, Hovda DA, Giza CC. The molecular pathophysiology of concussive brain injury. Clin Sports Med. 2011;30:33–48.
Sakas DE, Whitwell HL. Neurological episodes after minor head injury and trigeminovascular activation. Med Hypotheses. 1997;48:431–5.
The editor. Vasodilatation and migraine. Lancet. 1990;335:822–823.
Pietrobon D, Moskowitz MA. Chaos and commotion in the wake of cortical spreading depression and spreading depolarizations. Nat Rev Neurosci. 2014;15:379–93.
Marshall WH. Spreading cortical depression of Leao. Physiol Rev. 1959;39:239–79.
Geddes-Klein DM, Schiffman KB, Meaney DF. Mechanisms and consequences of neuronal stretch injury in vitro differ with the model of trauma. J Neurotrauma. 2006;23:193–204.
Keating CE, Cullen DK. Mechanosensation in traumatic brain injury. Neurobiol Dis. 2021;148:105210, Online ahead of print: https://doi.org/10.1016/j.nbd.2020.105210.
Tavalin SJ, Ellis EF, Satin LS. Mechanical perturbation of cultured cortical neurons reveals a stretch-induced delayed depolarization. J Neurophysiol. 1995;74:2767–73.
Hibino M, Itoh H, Kinosita K Jr. Time courses of cell electroporation as revealed by submicrosecond imaging of transmembrane potential. Biophys J. 1993;64:1789–800.
Fischer P, Sugimoto K, Chung DY, et al. Rapid hematoma growth triggers spreading depolarizations in experimental intracortical hemorrhage. J Cereb Blood Flow Metab. 2021;41:1264–76.
Hallez H, Vanrumste B, Grech R, et al. Review on solving the forward problem in EEG source analysis. J Neuroeng Rehabil. 2007;4: https://doi.org/10.1186/1743-0003-4-46.
Ramon C, Schimpf PH, Haueisen J. Influence of head models on EEG simulations and inverse source localizations. BioMed Eng OnLine. 2006; 5:1–13: https://doi.org/10.1186/1475-925X-5-10.
Miller JW, Kim W, Holmes MD, Vanhatalo S. Ictal localization by source analysis of infraslow activity in DC-coupled scalp EEG recordings. Neuroimage. 2007;35:583–97.
Chamanzar A, Behrmann M, Grover P. Neural silences can be localized rapidly using noninvasive scalp EEG. Commun Biol. 2021;4:429.
Cuffin BN. Effects of local variations in skull and scalp thickness on EEG’s and MEG’s. IEEE Trans Biomed Eng. 1993;40:42–8.
Cuffin BN. Effects of head shape on EEG’s and MEG’s. IEEE Trans Biomed Eng. 1990;37:44–52.
Stavtsev AI, Ushakov VL, Verkhliutov VM. [Modeling the effect of the layer thickness and tissue conductivities of the head and the brain on the EEG potentials using finite element method][Article in Russian]. Zh Vyssh Nerv Deiat Im I P Pavlova. 2007;57:742–752.
Baillet S, Mosher JC, Leahy RM. Electromagnetic brain mapping. IEEE Signal Process Mag. 2001;18:14–30.
Leahy RM, Mosher JC, Spencer ME, Huang MX, Lewine JD. A study of dipole localization accuracy for MEG and EEG using a human skull phantom. Electroencephalogr Clin Neurophysiol. 1998;107:159–73.
Wen P. The impact of inhomogeneous tissue anisotropy on potential distribution within head model. Australas Phys Eng Sci Med. 2003;26:115–8.
Plis SM, George JS, Jun SC, Ranken DM, Volegov PL, Schmidt DM. Probabilistic forward model for electroencephalography source analysis. Phys Med Biol. 2007;52:5309–27.
Seery GE. Surgical anatomy of the scalp. Dermatol Surg. 2002;28:581–7.
Penn JW, Bell EL. Electrical Parameter Values of Some Human Tissues in the Radiofrequency Radiation Range. 1978; Defense Technical Information Center, Report # SAM-TR-78–38: https://apps.dtic.mil/sti/pdfs/ADA064202.pdf
Gabriel C, Peyman A, Grant EH. Electrical conductivity of tissue at frequencies below 1 MHz. Phys Med Biol. 2009;54:4863–78.
Pellman EJ, Viano DC, Tucker AM, Casson IR. Concussion in professional football: location and direction of helmet impacts-Part 2. Neurosurgery. 2003;53:1328–40.
Dannhauer M, Lanfer B, Wolters CH, Knosche TR. Modeling of the human skull in EEG source analysis. Hum Brain Mapp. 2011;32:1383–99.
Petrov Y. Anisotropic spherical head model and its application to imaging electric activity of the brain. Phys Rev E Stat Nonlin Soft Matter Phys. 2012;86:011917–011917. https://doi.org/10.1103/PhysRevE.86.011917.
Bruno P, Vatta F, Mininel S, Inchingolo P. Referenced EEG and head volume conductor model: geometry and parametrical setting. Conf Proc IEEE Eng Med Biol Soc. 2004;2:833–6.
Johnson G, Massoudi M, Rajagopal KR. Flow of a fluid-solid mixture between flat plates. Chem Eng Sci. 1991;46:1713–23.
Tallgren P, Vanhatalo S, Kaila K, Voipio J. Evaluation of commercially available electrodes and gels for recording of slow EEG potentials. Clin Neurophysiol. 2005;116:799–806.
Voipio J, Tallgren P, Heinonen E, Vanhatalo S, Kaila K. Millivolt-scale DC shifts in the human scalp EEG: evidence for a nonneuronal generator. J Neurophysiol. 2003;89:2208–14.
Oostenveld R, Fries P, Maris E, Schoffelen JM. FieldTrip: Open source software for advanced analysis of MEG, EEG, and invasive electrophysiological data. Comput Intell Neurosci. 2011;2011:156869.
Vorwerk J, Oostenveld R, Piastra MC, Magyari L, Wolters CH. The FieldTrip-SimBio pipeline for EEG forward solutions. Biomed Eng Online. 2018;17:37.
Roache PJ. Verification and validation in computational science and engineering. Albuquerque, New Mexico: Hermosa Publishers; 1998.
Press WH, Teukolsky SA, Vetterling WT, Flannery BP. 3.6 Interpolation on a Grid in Multidimensions. Numerical Recipes 3rd Edition: The Art of Scientific Computing. Cambridge University Press, Cambridge; 2007:132–134.
Dreier JP, Fabricius M, Ayata C, et al. Recording, analysis, and interpretation of spreading depolarizations in neurointensive care: review and recommendations of the COSBID research group. J Cereb Blood Flow Metab. 2017;37:1595–625.
Voorhies JM, Cohen-Gadol A. Techniques for placement of grid and strip electrodes for intracranial epilepsy surgery monitoring: pearls and pitfalls. Surg Neurol Int. 2013;4:98: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3740610/.
Hartings JA, Wilson JA, Hinzman JM, et al. Spreading depression in continuous electroencephalography of brain trauma. Ann Neurol. 2014;76:681–94.
Klem GH, Luders HO, Jasper HH, Elger C. The ten-twenty electrode system of the International Federation. The International Federation of clinical neurophysiology. Electroencephalogr Clin Neurophysiol. 1999;52:3–6.
Oostenveld R, Praamstra P. The five percent electrode system for high-resolution EEG and ERP measurements. Clin Neurophysiol. 2001;112:713–9.
Hofmeijer J, van Kaam CR, van de Werff B, Vermeer SE, Tjepkema-Cloostermans MC, van Putten MJAM. Detecting cortical spreading depolarization with full band scalp electroencephalography: An illusion? Front Neurol. 2018;9: https://www.frontiersin.org/articles/https://doi.org/10.3389/fneur.2018.00017/full.
Hartings JA, Ngwenya LB, Watanabe T, Foreman B. Commentary: detecting cortical spreading depolarization with full band scalp electroencephalography: an illusion? Front Syst Neurosci. 2018;12: https://www.frontiersin.org/articles/https://doi.org/10.3389/fnsys.2018.00019/full.
Robinson D, Hartings J, Foreman B. First report of spreading depolarization correlates on scalp EEG confirmed with a depth electrode. Neurocrit Care. 2021.
Milakara D, Grozea C, Dahlem M, et al. Simulation of spreading depolarization trajectories in cerebral cortex: correlation of velocity and susceptibility in patients with aneurysmal subarachnoid hemorrhage. Neuroimage Clin. 2017;16:524–38.
Dahlem MA, Graf R, Strong AJ, et al. Two-dimensional wave patterns of spreading depolarization: retracting, re-entrant, and stationary waves. Physica D. 2010;239:889–903.
Kenny A, Plank MJ, David T. The effects of cerebral curvature on cortical spreading depression. J Theor Biol. 2019;472:11–26.
Santos E, Sanchez-Porras R, Sakowitz OW, Dreier JP, Dahlem MA. Heterogeneous propagation of spreading depolarizations in the lissencephalic and gyrencephalic brain. J Cereb Blood Flow Metab. 2017;37:2639–43.
Chamanzar A, George S, Venkatesh P, et al. An algorithm for automated, noninvasive detection of cortical spreading depolarizations based on EEG simulation. IEEE Trans Biomed Eng. 2019;66:1115–26.
Bures J, Buresova O, Krivanek J. The Mechanism and application of Leao’s spreading depression of electroencephalographic activity. Prague: Academia; 1974.
Brinley FJ Jr, Kandel ER, Marshall WH. Potassium outflux from rabbit cortex during spreading depression. J Neurophysiol. 1960;23:246–56.
Mestre H, Du T, Sweeney AM, et al. Cerebrospinal fluid influx drives acute ischemic tissue swelling. Science. 2020;367: https://doi.org/10.1126/science.aax7171.
Jones SC, Hund SJ, Singer E, Brown BR. Non-invasive detection of spreading depolarization: Initial results. 3rd International Conference on Spreading Depolarization (iCSD-2019); Yokohama, Japan: http://www.cosbid.org/wp-content/uploads/2019/07/iCSD-2019-Program.pdf.
Jones SC, Hund SJ, Singer E, Brown BR. Non-invasive detection of spreading depolarization: further results. 4th International Conference on Spreading Depolarization (iCSD-2021); Sep 20, 2021; Lyon, France: Abstract of accepted oral presentation available from author.
James MF, Smith MI, Bockhorst KH, et al. Cortical spreading depression in the gyrencephalic feline brain studied by magnetic resonance imaging. J Physiol. 1999;519(Pt 2):415–25.
Picton TW, Hillyard SA. Cephalic skin potentials in electroencephalography. Electroencephalogr Clin Neurophysiol. 1972;33:419–24.
Tallgren P. DC-stable electrode-skin interface for human EEG recording. 2005; University of Helsinki, Department of Biological and Environmental Sciences, Finland: Report # E5: http://lib.tkk.fi/Diss/2006/isbn9512269562/article4.pdf
Jones SC, Kharlamov A, Chung DY, Boada F, Yushmanov VE. Types of peri-infarct depolarization trajectories in experimental ischemic stroke. 4th International Conference on Spreading Depolarization (iCSD-2021); Sep 20, 2021; Lyon, France: Abstract of accepted poster presentation available from author.
Dahnke R, Yotter RA, Ziegler G, Gaser C. Brain Tissue Thickness Estimation using a Projection Scheme. 16th Annual Meeting of the Organization for Human Brain Mapping, Program #1458; Jun 6, 2010; Barcelona: Organization of Human Brain Mapping: https://www.researchgate.net/profile/Robert_Dahnke/publication/336513396_Brain_Tissue_Thickness_Estimation_Using_a_Projection_Scheme/links/5da45dde45851553ff8f658d/Brain-Tissue-Thickness-Estimation-Using-a-Projection-Scheme.pdf.
Bashkatov AN, Genina EA, Sinichkin YP, Kochubey VI, Lakodina NA, Tuchin VV. Glucose and mannitol diffusion in human dura mater. Biophys J. 2003;85:3310–8.
Mahinda HAM, Murty OP. Variability in thickness of human skull bones and sternum—an autopsy experience. J Forensic Med Toxicol. 2009;26:26–31.
Hori H, Moretti G, Rebora A, Crovato F. The thickness of human scalp: normal and bald. J Invest Dermatol. 1972;58:396–9.
Acknowledgements
We thank Thomas Ferguson, Richard Kraig, Douglas Smith, and Joel Greenberg for reading and commenting on the manuscript and Hiba Al-Ashtal for editorial services.
Funding
This project was conducted for CerebroScope, a medical device company developing a scalp direct current electroencephalography system for detecting SDs in severe acute brain injury, concussion, and migraine. This work was partially supported by grants from the United States Public Health Service National Institutes of Health; NS30839, NS30839-14S1, and NS66292 to the SCJ while at the Allegheny-Singer Research Institute; and 5R43NS092181 and 3R43NS092181-02S1 to SCJ for CerebroScope; Deutsche Forschungsgemeinschaft, German Research Council: DFG DR 323/5–1 and DFG DR 323/10–1 to JPD; and Bundesministerium fuer Bildung und Forschung (Era-Net Neuron EBio2), with funds from Bundesministerium fuer Bildung und Forschung (0101EW2004) to JPD.
Author information
Authors and Affiliations
Contributions
SJH developed the code with input from SCJ and PGM and wrote the first draft. BRB contributed additional analysis. KAE conceived of and performed the statistical analysis. JPD and CLL supplied the comparison data. SCJ, SJH, BRB, and JPD edited the article. SCJ conceived of the project, provided supervision, and revised the article. The final manuscript was approved by all authors.
Corresponding author
Ethics declarations
Conflicts of interest
Samuel J. Hund, Prahlad G. Menon, and Stephen C. Jones are founding partners and shareholders of CerebroScope. Benjamin R. Brown is a consultant to and shareholder of CerebroScope.
Ethical Approval/Informed Consent
All ethical standards have been met. See section “Retrospective Evaluation of Human Data.”
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the collection “Spreading Cortical Depolarization”.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MP4 6051 KB)
Rights and permissions
About this article
Cite this article
Hund, S.J., Brown, B.R., Lemale, C.L. et al. Numerical Simulation of Concussive-Generated Cortical Spreading Depolarization to Optimize DC-EEG Electrode Spacing for Noninvasive Visual Detection. Neurocrit Care 37 (Suppl 1), 67–82 (2022). https://doi.org/10.1007/s12028-021-01430-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-021-01430-x