Abstract
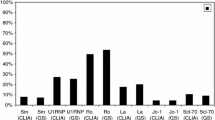
The identification of antinuclear antibodies (ANA) is an essential step in the diagnosis of different autoimmune diseases. The gold standard method for their detection is immunofluorescence assay. However, this is a subjective and laborious method, thus a need for simplified objective methods has aroused. In the current work, we evaluated such automated method, the LIAISON® (DiaSorin, Italy) for the detection of ANA. A total of 242 sera were analyzed including 67 from healthy subjects, 107 from primary biliary cirrhosis (PBC) patients, 20 from scleroderma patients and 48 from patients with Sjögren’s syndrome. All sera were analyzed using the automated chemiluminescent immunoassays, LIAISON® for the presence of ANA (kit No. 310300). Positive samples were further analyzed for the presence of antidouble-stranded DNA (dsDNA) and autoantibodies to 6 extractable nuclear antigens (ENA) of the LIAISON® (kits No. 310330 and 310331). Negative samples were further analyzed by Blueblot ANA assay (D-TEK, Belgium) or BlueDot Liver (D-TEK, Belgium) as appropriate. The LIAISON® specificity for ANA screening was 97 %. ANA positivity was determined in 80 % of all patients. The sensitivity was 95.5 % in scleroderma, 83 % in PBC and 72.9 % in Sjogren’s syndrome. ENA was positive in all ANA-positive scleroderma and Sjögren’s sera and in 27 % of ANA-positive PBC sera. Among scleroderma or Sjögren patients that were ANA negative, 4 samples were positive for anti-SSA and 2 for RNP-68 utilizing Blueblot assays. M2 protein was found in 1 out of the ANA-negative PBC patients. The LIAISON® ANA screen is specific and sensitive for the evaluation of ANA in patients with primary biliary cirrhosis, scleroderma and Sjögren’s syndrome.

Similar content being viewed by others
Abbreviations
- RNP:
-
Ribonucleoprotein particles
- Sm:
-
Smith
- CENP:
-
Centromere protein
- PM:
-
Polymyositis
- PCNA:
-
Proliferating cell nuclear antigen, gp210—nuclear envelope glycoprotein, sp100—nuclear body protein
- LKM1:
-
Liver kidney microsome
- LC1:
-
Liver cytosol
- SLA:
-
Soluble liver antigen, SS-A (Ro), SS-B (La), Jo-1 (histidyl-t-RNA synthetase), Scl-70 (DNA topoisomerase I)
- Scl:
-
Scleroderma
- M2:
-
Mitochondrial protein 2
References
Holborow EJ, Weir DM, Johnson GD. A serum factor in lupus erythematosus with affinity for tissue nuclei. Br Med J. 1957;2:732–4.
Defendenti C, Atzeni F, Spina MF, et al. Clinical and laboratory aspects of Ro/SSA-52 autoantibodies. Autoimmun Rev. 2011;10(3):150–4.
Agmon-Levin N, Shapira Y, Selmi C, et al. A comprehensive evaluation of serum autoantibodies in primary biliary cirrhosis. J Autoimmun. 2010;34(1):55–8.
Von Mühlen CA, Tan EM. Autoantibodies in the diagnosis of systemic rheumatic diseases. Semin Arthritis Rheum. 1995;24(5):323–58.
Fritzler MJ. Challenges to the use of autoantibodies as predictors of disease onset, diagnosis and outcomes. Autoimmun Rev. 2008;7(8):616–20.
Solomon DH, Kavanaugh AJ, Schur PH. Evidence-based guidelines for the use of immunologic tests: antinuclear antibody testing. Arthritis Rheum. 2002;47:434–44.
Clifford BD, Donahue D, Smith L, et al. High prevalence of serological markers of autoimmunity in patients with chronic hepatitis C. Hepatology. 1995;21:613–9.
Burnham TK. Antinuclear antibodies in patients with malignancies. Lancet. 1972;26(2):436.
Tan EM, Feltkamp TE, Smolen JS, et al. Range of antinuclear antibodies in “healthy” individuals. Arthritis Rheum. 1997;40(9):1601–11.
Nilsson BO, Skogh T, Ernerudh J, et al. Antinuclear antibodies in the oldest-old women and men. J Autoimmun. 2006;27(4):281–8.
Lacroix-Desmazes S, Kaveri SV, Mouthon L, et al. Self-reactive antibodies (natural autoantibodies) in healthy individuals. J Immunol Methods. 1998;216(1–2):117–37.
Shoenfeld Yehuda, Cervera Ricard, Eric Gershwin M. Diagnostic criteria in autoimmune diseases. New York: Humana Press; 2008.
Jolly M, Smaron M, Olsen Utset T, Ellman M. Are isolated antinucleolar antibodies a marker of scleroderma? J Clin Rheumatol. 2003;9(5):291–5.
Worman HJ, Courvalin JC. Antinuclear antibodies specific for primary biliary cirrhosis. Autoimmun Rev. 2003;2(4):211–7.
Terjung B, Spengler U. Role of auto-antibodies for the diagnosis of chronic cholestatic liver diseases. Clin Rev Allergy Immunol. 2005;28(2):115–33.
Fritzler MJ. The antinuclear antibody (ANA) test: last or lasting gasp? Arthritis Rheum. 2011;63(1):19–22.
Kavanaugh A, Tomar R, Reveille J, et al. Guidelines for clinical use of the antinuclear antibody test and tests for specific auto-antibodies to nuclear antigens. Arch Pathol Lab Med. 2000;124:71–81.
Bickel YB, Barnett EV, Pearson CM. Immunofluorescent patterns and specificity of human antinuclear antibodies. Clin Exp Immunol. 1968;3(7):641–56.
Fawcett T. An introduction to ROC analysis. Pattern Recognit Lett. 2006;27:861–74.
Wiik AS. Anti-nuclear antibodies: clinical utility for diagnosis, prognosis, monitoring, and planning of treatment strategy in systemic immunoinflammatory diseases. Scand J Rheumatol. 2005;34(4):260–8.
Keren DF. Antinuclear antibody testing. Clin Lab Med. 2002;22:447–74.
Wanchu A. Antinuclear antibodies: clinical applications. J Postgrad Med. 2000;46(2):144–8.
Gniewek RA, Stites DP, McHugh TM, et al. Comparison of antinuclear antibody testing methods: immunofluorescence assay versus enzyme immunoassay. Clin Diagn Lab Immunol. 1997;4(2):185–8.
González C, Guevara P, Alarcón I, et al. Antinuclear antibodies (ANA) screening by enzyme immunoassay with nuclear HEp-2 cell extract and recombinant antigens: analytical and clinical evaluation. Clin Biochem. 2002;35(6):463–9.
Check W. New autoimmune tests seek acceptance. CAP Today. 1995;9:1–36.
Shovman O, Gilburd B, Barzilai O, et al. Evaluation of the BioPlex 2200 ANA screen: analysis of 510 healthy subjects: incidence of natural/predictive autoantibodies. Ann NY Acad Sci. 2005;1050:380–8.
Gilburd B, Abu-Shakra M, Shoenfeld Y, et al. Autoantibodies profile in the sera of patients with Sjogren’s syndrome: the ANA evaluation—a homogeneous, multiplexed system. Clin Dev Immunol. 2004;11(1):53–6.
Nifli AP, Notas G, Mamoulaki M, et al. Comparison of a multiplex, bead-based fluorescent assay and immunofluorescence methods for the detection of ANA and ANCA autoantibodies in human serum. J Immunol Methods. 2006;311(1–2):189–97.
Hiemann R, Buttner T, Krieger T, et al. Challenges of automated screening and differentiation of non-organ specific autoantibodies on HEp-2 cells. Autoimmun Rev. 2009;9(1):17–22.
Melegari A, Bonaguri C, Russo A, et al. A comparative study on the reliability of an automated system for the evaluation of cell-based indirect immunofluorescence. Autoimmun Rev. 2012;11(10):713–6.
Rigon A, Buzzulini F, Soda P, et al. Novel opportunities in automated classification of antinuclear antibodies on HEp-2 cells. Autoimmun Rev. 2011;10(10):647–52.
Op De Beeck K, Vermeersch P, Verschueren P, et al. Detection of antinuclear antibodies by indirect immunofluorescence and by solid phase assay. Autoimmun Rev. 2011;10(12):801–8.
Routsias JG, Tzioufas AG. Sjögren’s syndrome–study of autoantigens and autoantibodies. Clin Rev Allergy Immunol. 2007;32(3):238–51.
Meroni PL, Schur PH. ANA screening: an old test with new recommendations. Ann Rheum Dis. 2010;69(8):1420–2.
Ghillani P, Rouquette AM, Desgruelles C, et al. Evaluation of the LIAISON ANA screen assay for antinuclear antibody testing in autoimmune diseases. Ann NY Acad Sci. 2007;1109:407–13.
Tan EM, Rodnan GP, Garcia I, et al. Diversity of antinuclear antibodies in patients with progressive sclerosis. Arthritis Rheum. 1980;32:617–25.
Basu D, Reveille JD. Anti-scl-70. Autoimmunity. 2005;38(1):65–72.
Riboldi P, Asero R, Origgi L, et al. Antinuclear antibodies in progressive systemic sclerosis. Clin Exp Rheumatol. 1985;3:205–11.
Mahler M, Silverman ED, Schulte-Pelkum J, Fritzler MJ. Anti-Scl-70 (topo-I) antibodies in SLE: myth or reality? Autoimmun Rev. 2010;9(11):756–60.
Dahle C, Skogh T, Aberg AK, et al. Methods of choice for diagnostic antinuclear antibody (ANA) screening: benefit of adding antigen-specific assays to immunofluorescence microscopy. J Autoimmun. 2004;22(3):241–8.
Pollock W, Toh BH. Routine immunofluorescence detection of Ro/SS-A autoantibody using HEp-2 cells transfected with human 60 kDa Ro/SS-A. Clin Pathol. 1999;52:684–7.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zafrir, Y., Gilburd, B., Carrasco, M.G. et al. Evaluation of an automated chemiluminescent immunoassay kit for antinuclear antibodies in autoimmune diseases. Immunol Res 56, 451–456 (2013). https://doi.org/10.1007/s12026-013-8416-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-013-8416-9