Abstract
Mice have been used to the study the mechanisms of protective innate and adaptive immunity to larval Strongyloides stercoralis. During primary infection, neutrophils and eosinophils are attracted by parasite components and kill the larvae by release of granule products. Eosinophils also function as antigen-presenting cells for the induction of a Th2 response. B cells produce both IgM and IgG that collaborate with neutrophils to kill worms in the adaptive immune response. Vaccine studies have identified a recombinant diagnostic antigen that induced high levels of immunity to infection with S. stercoralis in mice. These studies demonstrate that there are redundancies in the mechanisms used by the immune response to kill the parasite and that a vaccine with a single antigen may be suitable as a prophylactic vaccine to prevent human strongyloidiasis.
Similar content being viewed by others
Introduction
Strongyloidiasis has been described as the most neglected of the “neglected tropical diseases” [1]. This is surprising given that it has been estimated that Strongyloides stercoralis infects 30–100 million people in both resource-rich and resource-poor nations [2, 3]. One reason this disease has been neglected is that its prevalence has been severely underestimated. Diagnosis of this disease requires special detection methods that are not part of routine screening methods utilized to identify other parasitic infections in population surveys [2]. Also, the parasites are shed in low and inconsistent numbers that further complicate the diagnosis. Use of recombinant antigen-based serological diagnosis has greatly expanded the detection rates of this infection within endemic populations [4, 5]. Another reason this infection is overlooked is that the disease it causes is relatively benign during acute infection. Chronic infections may persist for the lifetime of the host and are commonly subclinical. However, chronically infected individuals who become immunosuppressed, often because of corticosteroid treatment or infection with HTLV-1, can develop hyperinfection syndrome, a condition that is life threatening [6–9]. Although chemotherapy (albendazole or ivermectin) is available for S. stercoralis infections, efficacy is rarely 100% [10, 11].
The life cycle of S. stercoralis is complex with direct, autoinfective, and free-living developmental cycles. The third-stage infective larvae (L3) initiate infection by penetrating the skin of the human host and then undergo a migration through the host tissues, during which the L3 undergo rapid development from the free-living infective third-stage larvae to a postpenetration, host-adapted transformed stage called the L3+ [12, 13]. The L3+ differs from the L3 antigenically, which is intuitive based on the differing environments in which the two worms reside. Similarly, significant physiological differences were seen between the infective larvae of S. venezuelensis and larvae found in the connective tissue, lungs, and intestinal mucosa [14].
After molting twice, the larvae enter the small intestine as mature egg-laying parthenogenetic female adult worms. The eggs hatch in the gut, and first-stage larvae are released in the feces and in the direct developmental cycle they develop into third-stage infective larvae. In the free-living developmental cycle, the first-stage larvae develop into free-living male and female adult worms that reproduce in the environment producing third-stage infective larvae. Alternatively, the first-stage larvae may develop into autoinfective third-stage larvae that never leave the host. Small numbers of these larvae may develop into adult worms, which results in chronic infections that may persist for decades through the continuous controlled replenishment of adult worms from autoinfective third-stage larvae. In patients that are immunosuppressed, hyperinfection may develop by the uncontrolled production of first-stage larvae and their development into autoinfective third-stage larvae that migrate throughout the host.
Another reason that strongyloidiasis has been a neglected disease is the difficulty of performing experiments with the parasite. Only humans, primates, and dogs are naturally susceptible to infection with S. stercoralis, which significantly limits experimentation. Strongyloides ratti and venezuelensis naturally infect rodents, and they have been used to study the biology and immunology of these infections [15–18]. However, a limitation of these studies is that results generated with the rodent parasites S. ratti and S. venezuelensis may not apply to the human pathogen S. stercoralis. Attempts to infect mice with S. stercoralis have revealed that immunologically intact mice and mice deficient in T cells killed the infections within several days [19]. However, SCID mice, which have a deficiency in both T and B cells, could be infected with S. stercoralis, with low numbers of adult worms and first-stage larvae developing in the mice after infection [20]. This indicates that S. stercoralis can develop in mice and that lymphocytes are part of the immune response involved in eliminating the infection.
One of the challenges in studying this infection in mice is the fact that the parasites migrate throughout the body. This makes accurate recovery of the parasites difficult and studying the parasite microenvironment in the host nearly impossible. To overcome these issues, diffusion chambers have been used as a means of containing the parasites in vivo in the subcutaneous tissues, a natural habitat for the larvae. Diffusion chambers are constructed out of Lucite rings to which are adhered membranes that block worms from exiting the diffusion chamber but allow free ingress and egress of host cells and soluble factors. The diffusion chamber system allows a thorough examination of parasite survival in mice and analyses of diffusion chamber contents indicate the innate and adaptive immune factors that are present in the parasite microenvironment.
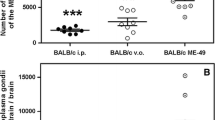
Innate immunity (Fig. 1)
The innate immune response to S. stercoralis larvae in naïve mice is characterized by the recruitment of neutrophils, macrophages, and eosinophils to the parasite, as seen within diffusion chambers. Similarly, S. venezuelensis induced an increase of eosinophils and mononuclear cells in the blood, peritoneal cavity fluid, and bronchoalveolar lavage fluid [21]. Cell contact is required for killing of S. stercoralis larvae, which occurred within 7 days in naïve immunocompetent mice [22]. Elimination of neutrophils and eosinophils from naïve mice by treatment with a monoclonal antibody against Gr-1 resulted in an increase in parasite survival [23], suggesting that these cells are active participants in the protective innate immune response. Likewise, studies with S. ratti have revealed that cellular infiltrates, consisting of neutrophils, eosinophils, and macrophages, were observed in the skin in response to the invading larvae in rats and mice during both primary and secondary responses [24, 25] and that granulocytes are crucial in the early defense against migrating larvae of S. ratti in mice [26].
Innate immune response to Strongyloides stercoralis in mice. During primary infection with larval Strongyloides stercoralis, neutrophils and eosinophils are attracted to the parasite microenvironment by parasite components. Killing of larvae is mediated by both eosinophils and neutrophils through the release of MBP from eosinophils and MPO from neutrophils. Both cell types require complement component C3b to kill the larvae. Eosinophils also have the capacity to function as antigen-presenting cells, presenting parasite antigens to T cells resulting in the induction of a Th2 response
Neutrophils
Studies performed with purified neutrophils indicated that they can independently kill the larvae [27] and that killing of the larvae by neutrophils is dependent on the neutrophil-specific granular protein myeloperoxidase (MPO) [23]. If neutrophil recruitment to the parasites in mice was blocked, either because of a defect in Gαi2 signaling [28] or in the expression of CXCR2 [27], the capacity of mice to kill S. stercoralis larvae was significantly decreased. CXCR2-dependent recruitment of neutrophils to larvae occurs independently of IL-17, and molecules extracted from S. stercoralis are capable of directly recruiting neutrophils through CXCR2, using signaling pathways similar to those used by host chemokines. In addition, the S. stercoralis soluble extract also induced neutrophils to release MIP-2 and KC, which further enhanced the recruitment of neutrophils. The finding that neutrophils produce increased amounts of neutrophil-recruiting chemokines following exposure to S. stercoralis soluble extract suggests an efficiently orchestrated system whereby a primary stimulus from a parasite causes an autocrine amplification of cell recruitment through release of host-derived chemokines. The efficiency of this recruitment strategy is further highlighted by the observation that the CXCR2 receptor has the ability to respond to both parasite- and host-derived factors resulting in highly efficient neutrophil recruitment and control of infection with S. stercoralis [29].
Eosinophils
Several methods have been used to assess the potential of eosinophils to kill the larvae of S. stercoralis and to determine whether they are required for protective innate immunity to the infection. IL-5 plays a key role in the differentiation, maturation, and survival of eosinophils [30, 31]. Treatment of mice with a monoclonal antibody to IL-5 reduced eosinophils and concomitantly reduced the capacity of mice to control the larvae of S. ratti in primary infections [32]. Survival of S. stercoralis was increased in naïve mice deficient in IL-5, and survival of the larvae was diminished in mice overexpressing IL-5 [33], thereby suggesting a direct role for eosinophils in killing the larvae. Treatment of naïve mice with an anti-CCR3 monoclonal antibody specifically eliminated eosinophils and blocked innate protective immunity to the infection [23, 27]. Surprisingly, larval killing in naïve PHIL mice, which constitutively lack eosinophils, was not diminished. Treatment of the PHIL mice with a monoclonal antibody to eliminate neutrophils resulted in a diminished protective innate immune response. This demonstrated that in the complete absence of eosinophils, neutrophils were capable of controlling the infection [23]. It was therefore concluded that both eosinophils and neutrophils can kill the larvae; in untreated mice, both cell types are required for optimal protective innate immunity [27]. However, in mice with a genetic deficiency in eosinophils, neutrophils are sufficient to compensate for the loss of eosinophils and kill the larvae. The mechanism used by mouse eosinophils to kill the larvae was shown to be dependent on the granular protein major basic protein (MBP) and not eosinophil peroxidase (EPO) [23]. MBP, EPO, eosinophil cationic protein (ECP), and eosinophil-derived neurotoxin (EDN), purified from human eosinophils, were tested in vitro for their toxicity to the larvae of S. stercoralis. Only MBP and ECP were toxic to the host-adapted larvae (L3+), while survival of infective larvae remained unaffected [34].
Studies on the schistosomula of S. mansoni have demonstrated that both EPO from eosinophils and MPO from neutrophils are toxic to the parasite. EPO and MPO apparently kill the organism through a similar mechanism dependent on hydrogen peroxide and a halide [35]. Likewise, both EPO and MPO contribute to tyrosine nitration through oxidation of nitrite [36]. Other studies have shown that MPO and EPO form reactive halide species through different mechanisms [37] and that MPO and EPO lyse Mycobacterium tuberculosis through different mechanisms. EPO induced cell wall lesions and lysis in the presence or absence of hydrogen peroxide whereas MPO required hydrogen peroxide to kill M. tuberculosis in the absence of any morphological changes in the pathogen [38]. MPO from neutrophils was required to kill the larvae of S. stercoralis whereas EPO from eosinophils did not function in that capacity, suggesting that the two peroxidases function against the larvae of S. stercoralis through different mechanisms, which results with one peroxidase killing the worm and the other not.
As with neutrophils, eosinophils are directly recruited to the parasite without the need for other host cell assistance. Chemoattractants derived from the larvae and host species stimulate similar receptors and second messenger signals to induce eosinophil chemotaxis. The parasite extract stimulates multiple receptors on the eosinophil surface, which ensures a robust innate immune response to the parasite. The redundancy of the chemotactic factors produced by the parasite and the multiple responding receptors on the eosinophils suggests chemotactic receptors on these pivotal cells may have evolved to ensure a robust response to this infection [39].
It is puzzling as to why the larvae of S. stercoralis recruit both neutrophils and eosinophils, cells that can kill the worm. One possibility is that the efficient recruitment of neutrophils and eosinophils to the larvae explains why mice are refractory to infection with this parasite. It would be interesting to see whether cells from hosts susceptible to the infection also are recruited to the parasite. Alternatively, there may be an advantage for the parasite to recruit the cells either for a direct beneficial effect as seen for Trichinella spiralis [40] or to repair tissue damage caused by the migrating larvae thereby promoting host survival.
Complement
In addition to neutrophils and eosinophils, complement activation is required for innate protective immunity to larval S. stercoralis. In vitro studies have shown that complement component C3 was detected on the surface of the larvae [41] and eosinophils only killed the larvae if a source of complement was provided [23]. Mice deficient in C3 did not kill the parasite whereas mice deficient in C3aR did eliminate the worms, suggesting that C3b is the active component of C3 that is required to mediate larval killing [42]. C3b may function in the larval killing process as an adherence molecule for cells, or it may facilitate activation and degranulation of the cells. Live S. stercoralis larvae activated complement in vitro through both the classical and alternative pathways that promoted the adherence of human monocytes and neutrophils to the surface of S. stercoralis [43]. Complement also promotes the binding of cells to the larvae of S. ratti [44].
Antigen-presenting cells (APC)
The transition from the innate to the adaptive immune response requires the parasite to be: (1) killed, (2) dissociated into a phagocytosable form, and (3) presented by antigen-presenting cells to T cells. Eosinophils are efficiently and independently recruited to the parasite [39] where they have the capacity to kill the larvae through the release of MBP [23]. Interestingly, eosinophils also have the capacity to act as APC inducing parasite-specific Th2 responses and antibody responses in both primary and secondary infections of mice with S. stercoralis [45, 46]. Eosinophils have also been reported to function as APC inducing Th2 responses in several experimental allergy models [47–49]. Therefore, eosinophils have the capacity to chemotax to the parasite microenvironment, kill the parasite, and then present the antigens to naïve T cells to induce adaptive immunity to the infection.
Adaptive immunity (Fig. 2)
The adaptive immune response in mice induced by immunization with live larvae is highly effective with greater than 90% of the challenge larvae killed within 24 h. Interestingly, the immune response generated by immunizing mice with live infective larvae was directed at the L3+ [12]. Combining this observation with the susceptibility of the L3+ to MBP [34] suggests that infective stage L3 are resistant to immune attack whereas the L3+ is the susceptible stage. Furthermore, antigenic differences were seen between the L3, L3+, and autoinfective larvae associated with chronic infections and hyperinfection. Immunity generated with L3 and directed at L3+ did not kill the autoinfective larvae [13]. This might explain how infections persist in human hosts for decades. Incoming infective larvae would be targeted by the adaptive immune response, and this immunity would prevent overwhelming infection with the parasite. The autoinfective larvae would survive in the face of this immune response, thereby perpetuating the parasite within the host. Furthermore, production of autoinfective larvae is apparently controlled by the immune response, based on their uncontrolled development in immunosuppressed individuals. The net result is that the immune response controls infective and autoinfective larvae through different mechanisms that result in infections with S. stercoralis persisting for the lifetime of the host, yet causing only minor pathological effects in most cases.
Adaptive immune response to Strongyloides stercoralis in mice. Immunization of mice with live larvae results in a Th2 response characterized by the production of both IL-4 and IL-5. B cells produce both IgM and IgG that collaborate with neutrophils to kill the worms through an accelerated killing process. As in the protective innate immune response, larval killing is dependent on the release of MPO from neutrophils and on complement component C3b
T cells
Humans appear to control infection with S. stercoralis through a T-helper type 2 (Th2) response based on the observation that patients co-infected with HTLV-1 and S. stercoralis have decreased production of Th2-type cytokines, increased IFN-γ production, and greater susceptibility to hyperinfection [8, 50, 51]. Th2 responses are the hallmark of many helminth infections with expression of IL-4 and IL-5 essential for control of Onchocerca volvulus [52], Heligmosomoides polygyrus [53], Trichuris muris [54], and Angiostrongylus cantonensis [55]. Protective adaptive immunity to S. stercoralis larvae in mice requires CD4+ but not CD8+ T cells [56]. Immunized mice treated with recombinant IL-12 demonstrated a pronounced shift from a Th2 to a Th1 response and thus blocked mice from developing protective adaptive immunity. Furthermore, depletion of the Th2-associated cytokines IL-4 or IL-5 from immunized mice using monoclonal antibodies impaired larval killing [56]. The requirement for a Th2 response for protective immunity also has been reported for S. venezuelensis [57] and S. ratti [58]. S. venezuelensis infections in Lewis rats shift the immune response from Th1 during acute infections to Th2 during the recovery phase [59] and IL-12-deficient mice infected with S. venezuelensis had higher levels of Th2 cytokines and decreased parasite burdens [60]. Primary infections of rats with S. ratti induced a Th2 response within 2–3 weeks postinfection [61], which resulted in the production of IL-4, IL-5, and IL-13 and a suppression of IFN-γ in mice and rats [62, 63].
Eosinophils
Reports on human effector cell responses to S. stercoralis are limited. The frequent absence of eosinophils associated with migrating larvae suggests that while peripheral eosinophilia is a common systemic response, eosinophils are not primarily involved in the tissue response to the parasite. Lymphocytes, macrophages, and neutrophils are frequently seen in close association with the larvae in various tissues; however, their role in controlling the infection has not been elucidated [64, 65]. Cumulatively, the data on human protective immune responses to S. stercoralis are limited, and analysis of the data is restricted to identifying correlations between immune responses and disease states; therefore, in vitro and animal models are required to define the actual role of cells in immunity to the S. stercoralis.
Depleting IL-5 from mice immunized against infection with S. stercoralis, either by monoclonal antibody treatment [34, 56] or by genetically knocking out IL-5 [33], resulted in decreased numbers of eosinophils and an absence of protective adaptive immunity. However, when eosinophils were specifically absent, either due to elimination by monoclonal antibody treatment [27] or the use of PHIL mice that are genetically deficient in eosinophils [23], it was determined that eosinophils were not required as effector cells in the adaptive immune response. Interestingly, immunized IL-5-deficient mice, which had severely reduced numbers of eosinophils, failed to establish protective immunity and had lower levels of parasite-specific IgM [33]. Reconstitution of immunized IL-5-deficient mice with wild-type eosinophils elevated the parasite-specific IgM levels, and the mice were then able to eliminate challenge infections [33]. Similarly, it has been reported that IgM induced by the adjuvant alum is compromised in mice genetically deficient in eosinophils and that transfer of IL-4 expressing eosinophils restored the production of antigen-specific IgM [66], thereby confirming a role for eosinophils in IgM production. Immunized PHIL mice, which have no eosinophils but intact cytokine levels, did not have reduced IgM levels [23]. The immunized PHIL mice appear to have an alternative source for molecules required for the induction of IgM production that IL-5−/− mice do not have. Therefore, eosinophils function as effector cells in the innate immune response, antigen-presenting cells and as sources of cytokines required for IgM production in the adaptive immune response.
Neutrophils
Studies were performed to determine the role of neutrophils in protective adaptive immunity to S. stercoralis. Using CXCR2−/− mice, it was demonstrated that a reduction in recruitment of neutrophils resulted in significantly reduced adaptive protective immunity. Protective antibody developed in immunized CXCR2−/− mice, thereby demonstrating that neutrophils are not required for the induction of the adaptive protective immune response. Moreover, neutrophils from wild-type and CXCR2−/− mice killed the larvae of S. stercoralis at the same rate, thus demonstrating that the defect in the CXCR2−/− mice was in recruitment of neutrophils and not in their ability to kill larvae [27]. Mice deficient in Gαi2 also failed to kill the larvae in a challenge infection with S. stercoralis despite developing an antigen-specific Th2 response characterized by increased IL-4, IL-5, IgM, and IgG. Neutrophils from Gαi2−/− mice were competent in killing larvae; however, immunized Gαi2−/− mice had significantly reduced recruitment of neutrophils to the parasite microenvironment, as seen within the diffusion chamber [28]. These data demonstrate that CXCR2 and Gαi2 are not required for the development of the protective immune responses against S. stercoralis; however, they are essential for the recruitment of neutrophils required for killing of larvae.
Toll-like receptors (TLRs) are pattern recognition receptors that recognize pathogen-associated molecular patterns on pathogens and alert the immune response to the presence of invading pathogens. TLRs are involved in both the innate and adaptive immune responses against several nematode infections including Brugia malayi [67], Onchocerca volvulus [68], Trichuris muris [69], and Syphacia obvelata [70]. Interestingly, TLR4-deficient mice were shown to have impaired neutrophil recruitment in several systems [71, 72]. C3H/HeJ mice, which have a point mutation in the Tlr4 gene, were used to determine the role of TLR4 in protective immunity to S. stercoralis. TLR4 was not required for killing the larvae during the innate immune response but was required for killing the parasites during the adaptive immune response. No differences were seen in the T cell responses, antibody responses, or cell recruitment to the parasite between wild-type and C3H/HeJ mice after immunization. However, it was determined that neutrophils from the C3H/HeJ mice could not participate in killing the worms in the adaptive immune response. The Tlr4 mutation severely alters the effector function, but not recruitment, of cells to the parasite microenvironment [73]. Finally, as in the innate immune response, neutrophils deficient in myeloperoxidase (MPO) had significantly decreased larval killing capacity [23]. Therefore, neutrophils require both MPO and TLR4 to kill the larvae of S. stercoralis in the adaptive immune response.
B cells
The function of B cells during S. stercoralis infection was studied in mice with specific B-cell deficiencies. Protective innate immunity in μMT mice, which lack mature B cells, was sufficient to eliminate all parasites; however, immunized μMT mice that had no detectable antibody levels did not kill the challenge infections. Xid mice, which lack B-1 cells, developed a modest level of parasite-specific IgG with little IgM following immunization and also did not kill worms in the challenge infections. These studies demonstrates that B cells, and specifically B-1 cells, are required for adaptive immunity and not innate immunity to S. stercoralis and suggest that IgM is required for adaptive immunity [74].
Complement and antibody
The protective adaptive immune response in mice to larval S. stercoralis is dependent on complement activation. In the initial studies, immunized mice treated with Cobra venom factor to deplete C3 were shown to be unable to kill the larvae [41]. The necessity of complement was confirmed in immunized C3−/−mice, where larval killing also did not occur. C3aR−/− mice killed larvae during the adaptive immune response as efficiently as wild-type mice thereby suggesting that C3b is the active complement component [42]. C3 has been observed to be on the surface of larvae recovered from immunized mice [41] and possibly serves as an anchor for cells to attach to the larvae in order to mediate killing of the parasite, as has been seen with human complement and cells in response to S. stercoralis [43]. Thus, complement activation is an integral component of both protective innate and adaptive immunity to S. stercoralis in mice.
Mice immunized with S. stercoralis have elevated IgA, IgG1, and IgM levels in the serum [22]. IgM recovered at 1 week postimmunization passively transfers protective immunity to naive mice through a mechanism dependent on granulocytes and complement [41] and both IgM and IgG recovered at 3 and 5 weeks postimmunization passively transfer immunity. IgG requires complement and neutrophils to kill the worms and functions through antibody-dependent cellular cytotoxicity (ADCC) based on studies in FcRγ−/− mice. This is in contrast to IgM from mice immunized with live larvae where protective immunity is ADCC independent. Western blots were performed to determine what antigens the protective IgM and IgG recognized and it was determined that both antibody isotypes recognized some shared antigens, whereas other antigens were recognized independently by either protective IgG or IgM. Furthermore, IgM bound to the surface of the cuticle, basal cuticle-hypodermis, coelomic cavity, and glandular esophagus, whereas the IgG bound only to the basal cuticle-hypodermis and the coelomic cavity [75]. It therefore was concluded that while IgM and IgG antibodies are both protective against larval S. stercoralis, they recognize different antigens and utilize different killing mechanisms. Similarly, (1) a role for antibody and cells in adaptive protective immunity to S. stercoralis in jirds has been observed [76], (2) IgG is required for antibody-dependent immunity to S. ratti [77], and (3) a correlation was observed between protective immunity to S. ratti and IgG in mice and rats [58, 78, 79].
Serum from S. stercoralis seropositive patients was used to evaluate the protective antibody response in humans. Only IgG from humans was able to passively transfer protective immunity to mice through an ADCC-independent mechanism that was dependent on mouse complement and neutrophils. The protective IgG binds to the cuticle and glands surrounding the esophagus. Western blot analysis comparing human and mouse IgG identified unique antigens recognized by human IgG, and few antigens recognized by both human and mouse IgG. Therefore, there are multiple antibody-dependent mechanisms that function in adaptive immunity killing the larvae of S. stercoralis. Both IgM and IgG from mice and IgG from humans participate in killing the parasite, although the antigenic targets and mechanisms are different. Interestingly, human and mouse antibody both require complement and neutrophils to kill the larvae.
Vaccine development
Studies were performed to identify antigens from S. stercoralis that would induce protective immunity and thereby be components of a vaccine against the infection. Mice immunized with soluble antigens derived from S. stercoralis larvae, administered with alum as the adjuvant, had a 50% reduction in larval survival. Purified IgG from mice immunized with the soluble antigens passively transferred immunity to naïve mice and was ADCC independent. Immunization of mice with the small pool of antigens specifically recognized by the protective IgG induced a level of parasite killing comparable to live larval immunization [75]. These studies demonstrated that a limited pool of native antigens, identified by mouse protective antibody, were capable of inducing a high level of protective immunity to S. stercoralis in mice.
Using protective human IgG, seven proteins were recognized in the pool of soluble S. stercoralis antigens, but only three were identified in the S. stercoralis EST database. The three proteins, tropomyosin (Sstmy-1), Na+–K+ATPase (Sseat-6), and LEC-5 (Sslec-5), were constructed into DNA plasmids. Sseat-6 was the only plasmid that induced a limited, but statistically significant, level of protective immunity against the S. stercoralis larvae [80], showing that the DNA encoding a single antigen could induce the development of protective immunity.
Single recombinant purified protein antigens were tested for efficacy as a vaccine against S. stercoralis. Ss-EAT-6, Ss-TMY-1, and Ss-LEC-5 were selected as they were recognized by human IgG and there was success with Ss-eat-6 using DNA immunization [80]. In addition, the recombinant diagnostic antigens Ss-NIE-1 and Ss-IR [4] were included in the study. Immunization with the recombinant antigens in alum revealed that only immunization with the diagnostic antigen Ss-IR stimulated high and reproducible levels of protective immunity to infection. IgG from mice immunized with Ss-IR could transfer protective immunity and was found to bind to the larval surface and to the granules in the glandular esophagus. Interestingly, this is the same location that the protective human IgG bound to the worms [81, 82]. Ss-IR has been identified as a highly effective recombinant antigen for use in a vaccine against S. stercoralis. The mechanism of action appears to be antibody dependent and the observation that it functions so successfully with alum as its adjuvant suggests that a Th2 response may be required [83]. In addition, the fact that alum is widely used as an adjuvant in humans adds to the potential for this vaccine to be used as a prophylactic vaccine in humans protecting them from infection with S. stercoralis.
Conclusions
It is clear from the studies on both innate and adaptive immunity that there is redundancy in the mechanisms used by the immune response to kill S. stercoralis in mice. In the innate response, both eosinophils and neutrophils can kill the larvae. In the adaptive immune response, neutrophils were required but functioned with both IgM and IgG. Furthermore, IgG induced by live parasites, IgG induced by soluble parasite antigens, and IgG from infected humans all seemed to kill the larvae through different mechanisms. It is possible that this vast armamentaria against the parasite is only present in mice, which might explain why they are naturally resistant to this infection. Alternatively, susceptible human hosts also may have these methods to control the infection; however, the worm has developed immune evasion mechanisms to specifically combat them in the human host.
It is interesting that complement and neutrophils were required by the protective adaptive immune response regardless of the source or isotype of the antibody. The observation that human and mouse IgG functioned in a similar manner provides evidence that the mouse model used for studying immunity to S. stercoralis is providing information that will translate into the human response to the infection. This is further confirmed by the discovery that an antigen that functions as a diagnostic indicator of human infection with S. stercoralis also functions as a vaccine against infection in mice. The SsIR antigen is clearly immunogenic in humans, which supports the concept that the antigen will be suitable as a prophylactic vaccine to prevent human strongyloidiasis.
References
Olsen A, van Lieshout L, Marti H, Polderman T, Polman K, Steinmann P, Stothard R, Thybo S, Verweij JJ, Magnussen P. Strongyloidiasis—the most neglected of the neglected tropical diseases? Trans R Soc Trop Med Hyg. 2009;103:967–72.
Bethony J, Brooker S, Albonico M, Geiger SM, Loukas A, Diemert D, Hotez PJ. Soil-transmitted helminth infections: ascariasis, trichuriasis, and hookworm. Lancet. 2006;367:1521–32.
Croker C, Reporter R, Redelings M, Mascola L. Strongyloidiasis-related deaths in the United States, 1991–2006. Am J Trop Med Hyg. 2010;83:422–6.
Krolewiecki AJ, Ramanathan R, Fink V, McAuliffe I, Cajal SP, Won K, Juarez M, Di Paolo A, Tapia L, Acosta N, Lee R, Lammie P, Abraham D, Nutman TB. Improved diagnosis of Strongyloides stercoralis using recombinant antigen-based serologies in a community-wide study in northern Argentina. Clin Vaccine Immunol. 2010;17:1624–30.
Montes M, Sawhney C, Barros N. Strongyloides stercoralis: there but not seen. Curr Opin Infect Dis. 2010;23:500–4.
Keiser PB, Nutman TB. Strongyloides stercoralis in the immunocompromised population. Clin Microbiol Rev. 2004;17:208–17.
Marcos LA, Terashima A, Dupont HL, Gotuzzo E. Strongyloides hyperinfection syndrome: an emerging global infectious disease. Trans R Soc Trop Med Hyg. 2008;102:314–8.
Montes M, Sanchez C, Verdonck K, Lake JE, Gonzalez E, Lopez G, Terashima A, Nolan T, Lewis DE, Gotuzzo E, White AC Jr. Regulatory T cell expansion in HTLV-1 and strongyloidiasis co-infection is associated with reduced IL-5 responses to Strongyloides stercoralis antigen. PLoS Negl Trop Dis. 2009;3:e456.
Ramanathan R, Nutman T. Strongyloides stercoralis infection in the immunocompromised host. Curr Infect Dis Rep. 2008;10:105–10.
Horton J. Albendazole: a review of anthelmintic efficacy and safety in humans. Parasitology. 2000;121(Suppl):S113–32.
Ikeda T. Pharmacological effects of ivermectin, an antiparasitic agent for intestinal strongyloidiasis: its mode of action and clinical efficacy. Nihon Yakurigaku Zasshi. 2003;122:527–38.
Brigandi RA, Rotman HL, Leon O, Nolan TJ, Schad GA, Abraham D. Strongyloides stercoralis host-adapted third-stage larvae are the target of eosinophil-associated immune-mediated killing in mice. J Parasitol. 1998;84:440–5.
Brigandi RA, Rotman HL, Nolan TJ, Schad GA, Abraham D. Chronicity in Strongyloides stercoralis infections: dichotomy of the protective immune response to infective and autoinfective larvae in a mouse model. Am J Trop Med Hyg. 1997;56:640–6.
Maruyama H, Nishimaki A, Takuma Y, Kurimoto M, Suzuki T, Sakatoku Y, Ishikawa M, Ohta N. Successive changes in tissue migration capacity of developing larvae of an intestinal nematode, Strongyloides venezuelensis. Parasitology. 2006;132:411–8.
Machado ER, Carlos D, Sorgi CA, Ramos SG, Souza DI, Soares EG, Costa-Cruz JM, Ueta MT, Aronoff DM, Faccioli LH. Dexamethasone effects in the Strongyloides venezuelensis infection in a murine model. Am J Trop Med Hyg. 2011;84:957–66.
Viney M. How do host immune responses affect nematode infections? Trends Parasitol. 2002;18:63–6.
Viney ME. The biology and genomics of Strongyloides. Med Microbiol Immunol. 2006;195:49–54.
Yoshida A, Nagayasu E, Nishimaki A, Sawaguchi A, Yanagawa S, Maruyama H. Transcripts analysis of infective larvae of an intestinal nematode, Strongyloides venezuelensis. Parasitol Int. 2011;60:75–83.
Dawkins HJ, Grove DI. Attempts to establish infections with Strongyloides stercoralis in mice and other laboratory animals. J Helminthol. 1982;56:23–6.
Rotman HL, Yutanawiboonchai W, Brigandi RA, Leon O, Nolan TJ, Schad GA, Abraham D. Strongyloides stercoralis: complete life cycle in SCID mice. Exp Parasitol. 1995;81:136–9.
Machado ER, Carlos D, Lourenco EV, Souza GE, Sorgi CA, Silva EV, Ueta MT, Ramos SG, Aronoff DM, Faccioli LH. Cyclooxygenase-derived mediators regulate the immunological control of Strongyloides venezuelensis infection. FEMS Immunol Med Microbiol. 2010;59:18–32.
Abraham D, Rotman HL, Haberstroh HF, Yutanawiboonchai W, Brigandi RA, Leon O, Nolan TJ, Schad GA. Strongyloides stercoralis: protective immunity to third-stage larvae inBALB/cByJ mice. Exp Parasitol. 1995;80:297–307.
O’Connell AE, Hess JA, Santiago GA, Nolan TJ, Lok JB, Lee JJ, Abraham D. Major basic protein from eosinophils and myeloperoxidase from neutrophils are required for protective immunity to Strongyloides stercoralis in mice. Infect Immun. 2011;79:2770–8.
Dawkins HJ, Muir GM, Grove DI. Histopathological appearances in primary and secondary infections with Strongyloides ratti in mice. Int J Parasitol. 1981;11:97–103.
McHugh TD, Jenkins T, McLaren DJ. Strongyloides ratti: studies of cutaneous reactions elicited in naive and sensitized rats and of changes in surface antigenicity of skin-penetrating larvae. Parasitology. 1989;98(Pt 1):95–103.
Watanabe K, Noda K, Hamano S, Koga M, Kishihara K, Nomoto K, Tada I. The crucial role of granulocytes in the early host defense against Strongyloides ratti infection in mice. Parasitol Res. 2000;86:188–93.
Galioto AM, Hess JA, Nolan TJ, Schad GA, Lee JJ, Abraham D. Role of eosinophils and neutrophils in innate and adaptive protective immunity to larval Strongyloides stercoralis in mice. Infect Immun. 2006;74:5730–8.
Padigel UM, Stein L, Redding K, Lee JJ, Nolan TJ, Schad GA, Birnbaumer L, Abraham D. Signaling through Galphai2 protein is required for recruitment of neutrophils for antibody-mediated elimination of larval Strongyloides stercoralis in mice. J Leukoc Biol. 2007;81:1120–6.
O’Connell AE, Redding KM, Hess JA, Lok JB, Nolan TJ, Abraham D. Soluble extract from the nematode Strongyloides stercoralis induces CXCR2 dependent/IL-17 independent neutrophil recruitment. Microbes Infect. 2011;13:536–44.
Lalani T, Simmons RK, Ahmed AR. Biology of IL-5 in health and disease. Ann Allergy Asthma Immunol. 1999;82:317–32, 332–3.
Roboz GJ, Rafii S. Interleukin-5 and the regulation of eosinophil production. Curr Opin Hematol. 1999;6:164–8.
Watanabe K, Sasaki O, Hamano S, Kishihara K, Nomoto K, Tada I, Aoki Y. Strongyloides ratti: the role of interleukin-5 in protection against tissue migrating larvae and intestinal adult worms. J Helminthol. 2003;77:355–61.
Herbert DR, Lee JJ, Lee NA, Nolan TJ, Schad GA, Abraham D. Role of IL-5 in innate and adaptive immunity to larval Strongyloides stercoralis in mice. J Immunol. 2000;165:4544–51.
Rotman HL, Yutanawiboonchai W, Brigandi RA, Leon O, Gleich GJ, Nolan TJ, Schad GA, Abraham D. Strongyloides stercoralis: eosinophil-dependent immune-mediated killing of third stage larvae in BALB/cByJ mice. Exp Parasitol. 1996;82:267–78.
Jong EC, Mahmoud AA, Klebanoff SJ. Peroxidase-mediated toxicity to schistosomula of Schistosoma mansoni. J Immunol. 1981;126:468–71.
Brennan ML, Wu W, Fu X, Shen Z, Song W, Frost H, Vadseth C, Narine L, Lenkiewicz E, Borchers MT, Lusis AJ, Lee JJ, Lee NA, Abu-Soud HM, Ischiropoulos H, Hazen SL. A tale of two controversies: defining both the role of peroxidases in nitrotyrosine formation in vivo using eosinophil peroxidase and myeloperoxidase-deficient mice, and the nature of peroxidase-generated reactive nitrogen species. J Biol Chem. 2002;277:17415–27.
Spalteholz H, Panasenko OM, Arnhold J. Formation of reactive halide species by myeloperoxidase and eosinophil peroxidase. Arch Biochem Biophys. 2006;445:225–34.
Borelli V, Vita F, Shankar S, Soranzo MR, Banfi E, Scialino G, Brochetta C, Zabucchi G. Human eosinophil peroxidase induces surface alteration, killing, and lysis of Mycobacterium tuberculosis. Infect Immun. 2003;71:605–13.
Stein LH, Redding KM, Lee JJ, Nolan TJ, Schad GA, Lok JB, Abraham D. Eosinophils utilize multiple chemokine receptors for chemotaxis to the parasitic nematode Strongyloides stercoralis. J Innate Immun. 2009;1:618–30.
Fabre V, Beiting DP, Bliss SK, Gebreselassie NG, Gagliardo LF, Lee NA, Lee JJ, Appleton JA. Eosinophil deficiency compromises parasite survival in chronic nematode infection. J Immunol. 2009;182:1577–83.
Brigandi RA, Rotman HL, Yutanawiboonchai W, Leon O, Nolan TJ, Schad GA, Abraham D. Strongyloides stercoralis: role of antibody and complement in immunity to the third stage of larvae in BALB/cByJ mice. Exp Parasitol. 1996;82:279–89.
Kerepesi LA, Hess JA, Nolan TJ, Schad GA, Abraham D. Complement component C3 is required for protective innate and adaptive immunity to larval Strongyloides stercoralis in mice. J Immunol. 2006;176:4315–22.
de Messias IJ, Genta RM, Mohren WD. Adherence of monocytes and polymorphonuclear cells to infective larvae of Strongyloides stercoralis after complement activation. J Parasitol. 1994;80:267–74.
Grove DI, Northern C, Dawkins HJ. Interactions of Strongyloides ratti free-living and skin-penetrated infective larvae and parasitic adults with serum and cells in vitro. Aust J Exp Biol Med Sci. 1985;63(Pt 5):521–9.
Padigel UM, Hess JA, Lee JJ, Lok JB, Nolan TJ, Schad GA, Abraham D. Eosinophils act as antigen-presenting cells to induce immunity to Strongyloides stercoralis in mice. J Infect Dis. 2007;196:1844–51.
Padigel UM, Lee JJ, Nolan TJ, Schad GA, Abraham D. Eosinophils can function as antigen-presenting cells to induce primary and secondary immune responses to Strongyloides stercoralis. Infect Immun. 2006;74:3232–8.
MacKenzie JR, Mattes J, Dent LA, Foster PS. Eosinophils promote allergic disease of the lung by regulating CD4(+) Th2 lymphocyte function. J Immunol. 2001;167:3146–55.
Shi HZ. Eosinophils function as antigen-presenting cells. J Leukoc Biol. 2004;76:520–7.
Xie ZF, Shi HZ, Qin XJ, Kang LF, Huang CP, Chen YQ. Effects of antigen presentation of eosinophils on lung Th1/Th2 imbalance. Chin Med J (Engl). 2005;118:6–11.
Neva FA, Filho JO, Gam AA, Thompson R, Freitas V, Melo A, Carvalho EM. Interferon-gamma and interleukin-4 responses in relation to serum IgE levels in persons infected with human T lymphotropic virus type I and Strongyloides stercoralis. J Infect Dis. 1998;178:1856–9.
Porto AF, Neva FA, Bittencourt H, Lisboa W, Thompson R, Alcantara L, Carvalho EM. HTLV-1 decreases Th2 type of immune response in patients with strongyloidiasis. Parasite Immunol. 2001;23:503–7.
Lange AM, Yutanawiboonchai W, Scott P, Abraham D. IL-4- and IL-5-dependent protective immunity to Onchocerca volvulus infective larvae in BALB/cBYJ mice. J Immunol. 1994;153:205–11.
Urban JF Jr, Katona IM, Paul WE, Finkelman FD. Interleukin 4 is important in protective immunity to a gastrointestinal nematode infection in mice. Proc Natl Acad Sci USA. 1991;88:5513–7.
Else KJ, Grencis RK. Cellular immune responses to the murine nematode parasite Trichuris muris. I. Differential cytokine production during acute or chronic infection. Immunology. 1991;72:508–13.
Sasaki O, Sugaya H, Ishida K, Yoshimura K. Ablation of eosinophils with anti-IL-5 antibody enhances the survival of intracranial worms of Angiostrongylus cantonensis in the mouse. Parasite Immunol. 1993;15:349–54.
Rotman HL, Schnyder-Candrian S, Scott P, Nolan TJ, Schad GA, Abraham D. IL-12 eliminates the Th-2 dependent protective immune response of mice to larval Strongyloides stercoralis. Parasite Immunol. 1997;19:29–39.
Fernandes A, Pereira AT, Eschenazi PD, Schilter HC, Sousa AL, Teixeira MM, Negrao-Correa D. Evaluation of the immune response against Strongyloides venezuelensis in antigen-immunized or previously infected mice. Parasite Immunol. 2008;30:139–49.
Bleay C, Wilkes CP, Paterson S, Viney ME. Density-dependent immune responses against the gastrointestinal nematode Strongyloides ratti. Int J Parasitol. 2007;37:1501–9.
Chiuso-Minicucci F, Marra NM, Zorzella-Pezavento SF, Franca TG, Ishikawa LL, Amarante MR, Amarante AF, Sartori A. Recovery from Strongyloides venezuelensis infection in Lewis rats is associated with a strong Th2 response. Parasite Immunol. 2010;32:74–8.
Machado ER, Carlos D, Lourenco EV, Sorgi CA, Silva EV, Ramos SG, Ueta MT, Aronoff DM, Faccioli LH. Counterregulation of Th2 immunity by interleukin 12 reduces host defenses against Strongyloides venezuelensis infection. Microbes Infect. 2009;11:571–8.
Wilkes CP, Bleay C, Paterson S, Viney ME. The immune response during a Strongyloides ratti infection of rats. Parasite Immunol. 2007;29:339–46.
Eschbach ML, Klemm U, Kolbaum J, Blankenhaus B, Brattig N, Breloer M. Strongyloides ratti infection induces transient nematode-specific Th2 response and reciprocal suppression of IFN-gamma production in mice. Parasite Immunol. 2010;32:370–83.
Paterson S, Wilkes C, Bleay C, Viney ME. Immunological responses elicited by different infection regimes with Strongyloides ratti. PLoS One. 2008;3:e2509.
Genta RM, Gomes MC. Pathology. In: Grove DI, editor. Strongyloidiasis a major roundworm infection of man. Philadelphia, PA, USA: Taylor & Francis Inc.; 1989. p. 105–132.
Haque AK, Schnadig V, Rubin SA, Smith JH. Pathogenesis of human strongyloidiasis: autopsy and quantitative parasitological analysis. Mod Pathol. 1994;7:276–88.
Wang HB, Weller PF. Pivotal advance: eosinophils mediate early alum adjuvant-elicited B cell priming and IgM production. J Leukoc Biol. 2008;83:817–21.
Hayashi Y, Nakagaki K, Nogami S, Hammerberg B, Tanaka H. Protective immunity against Brugia malayi infective larvae in mice. I. Parameters of active and passive immunity. Am J Trop Med Hyg. 1989;41:650–6.
Kerepesi LA, Leon O, Lustigman S, Abraham D. Protective immunity to the larval stages of Onchocerca volvulus is dependent on Toll-like receptor 4. Infect Immun. 2005;73:8291–7.
Helmby H, Grencis RK. Essential role for TLR4 and MyD88 in the development of chronic intestinal nematode infection. Eur J Immunol. 2003;33:2974–9.
Stewart PW, Chapes SK. Role of major histocompatibility complex class II in resistance of mice to naturally acquired infection with Syphacia obvelata. Comp Med. 2003;53:70–4.
Fan J, Malik AB. Toll-like receptor-4 (TLR4) signaling augments chemokine-induced neutrophil migration by modulating cell surface expression of chemokine receptors. Nat Med. 2003;9:315–21.
Kirimanjeswara GS, Mann PB, Pilione M, Kennett MJ, Harvill ET. The complex mechanism of antibody-mediated clearance of Bordetella from the lungs requires TLR4. J Immunol. 2005;175:7504–11.
Kerepesi LA, Hess JA, Leon O, Nolan TJ, Schad GA, Abraham D. Toll-like receptor 4 (TLR4) is required for protective immunity to larval Strongyloides stercoralis in mice. Microbes Infect. 2007;9:28–34.
Herbert DR, Nolan TJ, Schad GA, Abraham D. The role of B cells in immunity against larval Strongyloides stercoralis in mice. Parasite Immunol. 2002;24:95–101.
Ligas JA, Kerepesi LA, Galioto AM, Lustigman S, Nolan TJ, Schad GA, Abraham D. Specificity and mechanism of immunoglobulin M (IgM)- and IgG-dependent protective immunity to larval Strongyloides stercoralis in mice. Infect Immun. 2003;71:6835–43.
Nolan TJ, Rotman HL, Bhopale VM, Schad GA, Abraham D. Immunity to a challenge infection of Strongyloides stercoralis third-stage larvae in the jird. Parasite Immunol. 1995;17:599–604.
Murrell KD. Protective role of immunoglobulin G in immunity to Strongyloides ratti. J Parasitol. 1981;67:167–73.
Dawkins HJ, Grove DI. Transfer by serum and cells of resistance to infection with Strongyloides ratti in mice. Immunology. 1981;43:317–22.
Uchikawa R, Ichiki H, Komaki E. Antibody responses and protective immunity in rats receiving repeated inoculations of Strongyloides ratti. J Parasitol. 1991;77:737–41.
Kerepesi LA, Keiser PB, Nolan TJ, Schad GA, Abraham D, Nutman TB. DNA immunization with Na+–K+ ATPase (Sseat-6) induces protective immunity to larval Strongyloides stercoralis in mice. Infect Immun. 2005;73:2298–305.
Abraham D, Hess JA, Mejia R, Nolan TJ, Lok JB, Lustigman S, Nutman TB. Immunization with the recombinant antigen Ss-IR induces protective immunity to infection with Strongyloides stercoralis in mice. Vaccine. 2011;29:8134–40.
Kerepesi LA, Nolan TJ, Schad GA, Lustigman S, Herbert DR, Keiser PB, Nutman TB, Krolewiecki AJ, Abraham D. Human immunoglobulin G mediates protective immunity and identifies protective antigens against larval Strongyloides stercoralis in mice. J Infect Dis. 2004;189:1282–90.
Brewer JM, Conacher M, Hunter CA, Mohrs M, Brombacher F, Alexander J. Aluminium hydroxide adjuvant initiates strong antigen-specific Th2 responses in the absence of IL-4- or IL-13-mediated signaling. J Immunol. 1999;163:6448–54.
Acknowledgments
This research was supported in part by the Edna McConnell Foundation and by National Institutes of Health grants RO1AI47189 and 1R56AI076345. We acknowledge the contributions made by past members of the laboratory to the findings presented in this review: Richard Brigandi, Ann Marie Galioto, Frances Haberstroh, De’Broski Herbert, Larua Kerepesi, Alejandro Krolewiecki, Ofra Leon, Amy O’Connell, Udaikumar Padigel, Kevin Redding, Harris Rotman, Louis Stein, and Wiboonchai Yutanawiboonchai. We thank our past and present collaborators for their assistance with the studies discussed in this review: James Lee, James Lok, Sara Lustigman, Thomas Nolan, Thomas Nutman, and Gerhard Schad,.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bonne-Année, S., Hess, J.A. & Abraham, D. Innate and adaptive immunity to the nematode Strongyloides stercoralis in a mouse model. Immunol Res 51, 205–214 (2011). https://doi.org/10.1007/s12026-011-8258-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-011-8258-2