Abstract
Aim
To evaluate the prevalence and incidence of type 2 diabetes in patients with nonfunctioning adrenal incidentalomas (NFAI) or adrenal incidentalomas (AI) with autonomous cortisol secretion (ACS).
Methods
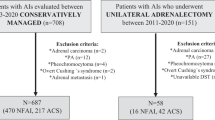
In this single-center retrospective study, all patients with adrenal incidentalomas ≥1 cm and ACS or NFAI studied between 2013 and 2020 were included. ACS was defined by a post-dexamethasone suppression test (DST) serum cortisol concentration ≥1.8 μg/dl, in the absence of signs of hypercortisolism, and NFAI was defined as a DST < 1.8 μg/dl without biochemical evidence of hypersecretion of other hormones.
Results
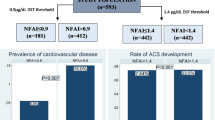
Inclusion criteria were met by 231 patients with ACS and 478 with NFAI. At diagnosis, type 2 diabetes was present in 24.3% of patients. No differences were found in the prevalence of type 2 diabetes (27.7 vs. 22.6%, P = 0.137) between patients with ACS and NFAI. However, fasting plasma glucose values and glycated hemoglobin levels were significantly higher in patients with ACS than with NFAI (112 ± 35.6 vs. 105 ± 29 mg/dl, P = 0.004; and 6.5 ± 1.4 vs. 6.1 ± 0.9%, P = 0.005, respectively). Furthermore, patients with type 2 diabetes had higher urinary free cortisol (P = 0.039) and late-night salivary cortisol levels (P = 0.010) than those without type 2 diabetes. After a median follow-up of 28 months, no differences were found in the incidence of type 2 diabetes between the groups (HR 1.17, 95% 0.52–2.64).
Conclusion
Type 2 diabetes was present in one fourth of our cohort. We found no differences in its prevalence or incidence between the groups. However, glycemic control might be worse among diabetic patients with ACS. Higher concentrations of urinary and salivary cortisol were found in patients with than without type 2 diabetes.

Similar content being viewed by others
References
M. Fassnacht, W. Arlt, I. Bancos, H. Dralle, J. Newell-Price, A. Sahdev, A. Tabarin, M. Terzolo, S. Tsagarakis, O.M. Dekkers, Management of adrenal incidentalomas: European Society of Endocrinology Clinical Practice Guideline in collaboration with the European Network for the Study of Adrenal Tumors. Eur. J. Endocrinol. 175(2 Aug), G1–G34 (2016). https://doi.org/10.1530/EJE-16-0467
J.H. Song, F.S. Chaudhry, W.W. Mayo-Smith, The incidental adrenal mass on CT: prevalence of adrenal disease in 1,049 consecutive adrenal masses in patients with no known malignancy. AJR Am. J. Roentgenol. 190(5 May), 1163–1168 (2008). https://doi.org/10.2214/AJR.07.2799
G. Reimondo, E. Castellano, M. Grosso, R. Priotto, S. Puglisi, A. Pia, M. Pellegrino, G. Borretta, M. Terzolo, Adrenal Incidentalomas are Tied to Increased Risk of Diabetes: Findings from a Prospective Study. J. Clin. Endocrinol. Metab. 105(4 Apr), dgz284 (2020). https://doi.org/10.1210/clinem/dgz284
A. Ebbehoj, D. Li, R.J. Kaur, C. Zhang, S. Singh, T. Li, E. Atkinson, S. Achenbach, S. Khosla, W. Arlt, W.F. Young, W.A. Rocca, I. Bancos, Epidemiology of adrenal tumours in Olmsted County, Minnesota, USA: a population-based cohort study. Lancet Diabetes Endocrinol. 8(11 Nov), 894–902 (2020). https://doi.org/10.1016/S2213-8587(20)30314-4
Araujo-Castro M., García Cano A.M., Escobar-Morreale H.F., Valderrabano P. Predictive model for autonomous cortisol secretion development in non-functioning adrenal incidentalomas. Hormones (Athens) 2022. https://doi.org/10.1007/S42000-022-00406-6
American Diabetes Association Professional Practice Committee, 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2022. Diabetes Care 45(Supplement_1 Jan), S17–S38 (2022). https://doi.org/10.2337/dc22-S002
Third Report of the National Cholesterol Education Program (NCEP), Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 106, 3143–3421 (2002). https://doi.org/10.1161/circ.106.25.3143
Y.S. Elhassan, F. Alahdab, A. Prete, D.A. Delivanis, A. Khanna, L. Prokop, M.H. Murad, M.W. O’Reilly, W. Arlt, I. Bancos, Natural History of Adrenal Incidentalomas With and Without Mild Autonomous Cortisol Excess: A Systematic Review and Meta-analysis. Ann. Intern. Med. 171(2 Jul), 107–116 (2019). https://doi.org/10.7326/M18-3630
D. Lopez, M.A. Luque-Fernandez, A. Steele, G.K. Adler, A. Turchin, A. Vaidya, “Nonfunctional” Adrenal Tumors and the Risk for Incident Diabetes and Cardiovascular Outcomes: A Cohort Study. Ann. Intern. Med. 165(8 Oct), 533–542 (2016). https://doi.org/10.7326/M16-0547
M. Terzolo, A. Pia, A. Alì, G. Osella, G. Reimondo, S. Bovio, F. Daffara, M. Procopio, P. Paccotti, G. Borretta, A. Angeli, Adrenal incidentaloma: a new cause of the metabolic syndrome? J. Clin. Endocrinol. Metab. 87(3 Mar), 998–1003 (2002). https://doi.org/10.1210/jcem.87.3.8277
M. Reincke, M. Fassnacht, S. Väth, P. Mora, B. Allolio, Adrenal incidentalomas: a manifestation of the metabolic syndrome? Endocr. Res. 22(4 Nov), 757–761 (1996). https://doi.org/10.1080/07435809609043773
G. Muscogiuri, G.P. Sorice, A. Prioletta, T. Mezza, C. Cipolla, E. Salomone, A. Giaccari, A. Pontecorvi, Della, S. Casa, The size of adrenal incidentalomas correlates with insulin resistance. Is there a cause-effect relationship? Clin. Endocrinol. (Oxf.) 74(3 Mar), 300–305 (2011). https://doi.org/10.1111/j.1365-2265.2010.03928.x
I.I. Androulakis, G.A. Kaltsas, G.E. Kollias, A.C. Markou, A.K. Gouli, D.A. Thomas, K.I. Alexandraki, C.M. Papamichael, D.J. Hadjidakis, G.P. Piaditis, Patients with apparently nonfunctioning adrenal incidentalomas may be at increased cardiovascular risk due to excessive cortisol secretion. J. Clin. Endocrinol. Metab. 99(8 Aug), 2754–2762 (2014). https://doi.org/10.1210/jc.2013-4064
G.I. Sydney, K.J. Ioakim, S.A. Paschou, Insulin resistance and adrenal incidentalomas: A bidirectional relationship. Maturitas 121(Mar), 1–6 (2019). https://doi.org/10.1016/j.maturitas.2018.12.002
J.A. Higgs, A.P. Quinn, K.D. Seely, Z. Richards, S.P. Mortensen, C.S. Crandall, A.E. Brooks, Pathophysiological Link between Insulin Resistance and Adrenal Incidentalomas. Int J. Mol. Sci. 23(8 Apr), 4340 (2022). https://doi.org/10.3390/ijms23084340
M. Araujo-Castro, H.F. Escobar-Morreale, P. Valderrabano, A Proposal for Nomenclature Revision of Nonfunctioning Adrenal Incidentalomas as Adrenal Lesions of Undetermined Secretion of Adrenal Steroids (ALUSAS). Endocr. Pract. 28(9 Sep), 918–920 (2022). https://doi.org/10.1016/j.eprac.2022.06.007
M. Araujo-Castro, E. Pascual-Corrales, A.M. García Cano, M. Marchan, G. Casals, F.A. Hanzu, M.Á. Gomez-Bermejo, H.F. Escobar Morreale, P. Valderrabano, Evaluation of Body Composition in Patients With and Without Adrenal Tumors and Without Overt Hypersecretory Syndromes. Endocr. Pract. 29(2 Feb), 110–118 (2023). https://doi.org/10.1016/j.eprac.2022.11.009
T. Kamio, K. Shigematsu, K. Kawai, H. Tsuchiyama, Immunoreactivity and receptor expression of insulinlike growth factor I and insulin in human adrenal tumors. An immunohistochemical study of 94 cases. Am. J. Pathol. 138(1 Jan), 83–91 (1991)
A. Penhoat, P.G. Chatelain, C. Jaillard, J.M. Saez, Characterization of insulin-like growth factor I and insulin receptors on cultured bovine adrenal fasciculata cells. Role of these peptides on adrenal cell function. Endocrinology 122(6 Jun), 2518–2526 (1988). https://doi.org/10.1210/endo-122-6-2518
D.J. Pillion, P. Arnold, M. Yang, C.R. Stockard, W.E. Grizzle, Receptors for insulin and insulin-like growth factor-I in the human adrenal gland. Biochem. Biophys. Res. Commun. 165(1 Nov), 204–211 (1989). https://doi.org/10.1016/0006-291x(89)91055-3
M.M. Weber, C.J. Auernhammer, W. Kiess, D. Engelhardt, Insulin-like growth factor receptors in normal and tumorous adult human adrenocortical glands. Eur. J. Endocrinol. 136(3 Mar), 296–303 (1997). https://doi.org/10.1530/eje.0.1360296
B. Altieri, G. Tirabassi, S. Della Casa, C.L. Ronchi, G. Balercia, F. Orio, A. Pontecorvi, A. Colao, G. Muscogiuri, Adrenocortical tumors and insulin resistance: What is the first step? Int J. Cancer 138(12 Jun), 2785–2794 (2016). https://doi.org/10.1002/ijc.29950
K.C. Leung, N. Doyle, M. Ballesteros, M.J. Waters, K.K. Ho, Insulin regulation of human hepatic growth hormone receptors: divergent effects on biosynthesis and surface translocation. J. Clin. Endocrinol. Metab. 85(12 Dec), 4712–4720 (2000). https://doi.org/10.1210/jcem.85.12.7017
A. Belfiore, F. Frasca, G. Pandini, L. Sciacca, R. Vigneri, Insulin receptor isoforms and insulin receptor/insulin-like growth factor receptor hybrids in physiology and disease. Endocr. Rev. 30(6 Oct), 586–623 (2009). https://doi.org/10.1210/er.2008-0047
F. Peruzzi, M. Prisco, M. Dews, P. Salomoni, E. Grassilli, G. Romano, B. Calabretta, R. Baserga, Multiple signaling pathways of the insulin-like growth factor 1 receptor in protection from apoptosis. Mol. Cell Biol. 19(10 Oct), 7203–7215 (1999). https://doi.org/10.1128/MCB.19.10.7203
P. Vigneri, F. Frasca, L. Sciacca, G. Pandini, R. Vigneri, Diabetes and cancer. Endocr. Relat. Cancer 16(4 Dec), 1103–1123 (2009). https://doi.org/10.1677/ERC-09-0087
Funding
Ayuda a la Investigación en Endocrinología, Nutrición y Diabetes 2019, Sociedad de Endocrinología, Nutrición y Diabetes de la Comunidad de Madrid (SENDIMAD). Convocatoria intramural de ayudas a proyectos de investigación de investigadores noveles, investigadores clínicos asociados y/o grupos emergentes del Hospital Universitario Ramón y Cajal 2019, IRYCIS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
All procedures performed in the participants of the study were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was approved by the Ethics Committee of Hospital Universitario Ramón y Cajal on February 14 of 2019.
Informed consent
The Ethical committee of Hospital Universitario Ramón y Cajal approved the waiver for informed consent given the retrospective nature of the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Brox-Torrecilla, N., García Cano, A.M., Valderrábano, P. et al. Prevalence and incidence of type 2 diabetes mellitus in patients with adrenal incidentalomas: a study of 709 cases. Endocrine 81, 484–491 (2023). https://doi.org/10.1007/s12020-023-03396-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-023-03396-6