Abstract
Context
Hypoparathyroidism (hypoPTH) in adults is mainly due to total thyroidectomy. Conventional therapies (calcium, active vitamin D) can fail to normalize calcemia, expose the patient to hypercalciuria and impact quality-of-life. Human parathormone (PTH) replacement therapy is a suitable option in these cases, although few clinical reports have been published so far.
Methods
We describe two cases of patients with refractory postsurgical hypoPTH, in whom subcutaneous infusion of recombinant PTH (teriparatide) through the Omnipod® pump was initiated after failure of all other therapeutic options. Besides, we performed a review of literature of hypoPTH cases treated by continuous infusion of teriparatide.
Results
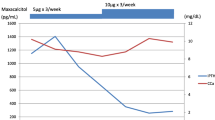
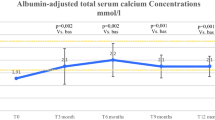
Two women aged 46 and 61 years old failed to normalize calcemia either with conventional treatments (calcium 8 g/day + calcitriol 9 mcg/day and calcium 5 g/day + calcitriol 12 mcg/day) or with thrice daily subcutaneous injections of teriparatide. As a last resort, teriparatide infusion via Omnipod® device normalized their calcemia and allowed calcium/vitamin D withdrawal, with average teriparatide dose of 23 and 32 mcg/day, respectively. The flow of teriparatide was adapted according to a protocol based on measured calcemia, under medical supervision. In the literature, 15 adult cases (13 women, mean age 44.5 ± 5.2 years old) are reported. HypoPTH was consecutive to surgery in all of them. Mean dose of teriparatide administered was 25 ± 6 mcg/day with improvement of calcemia level and quality-of-life in all patients.
Conclusions
Continuous administration of teriparatide through Omnipod® appears as an efficient therapeutic option in refractory hypoPTH, whose administration to the patient can be assisted by medically-supervised protocol.


Similar content being viewed by others
References
M. Mannstadt, J.P. Bilezikian, R.V. Thakker, F.M. Hannan, B.L. Clarke, L. Rejnmark, D.M. Mitchell, T.J. Vokes, K.K. Winer, D.M. Shoback, Nat. Rev. Dis. Prim. 3, 17055 (2017)
B.L. Clarke, E.M. Brown, M.T. Collins, H. Jüppner, P. Lakatos, M.A. Levine, M.M. Mannstadt, J.P. Bilezikian, A.F. Romanischen, R.V. Thakker, J. Clin. Endocrinol. Metab. 101, 2284 (2016)
J. Powers, K. Joy, A. Ruscio, H. Lagast, J. Bone Miner. Res. 28, 2570 (2013)
J.P. Bilezikian, J. Clin. Endocrinol. Metab. 105, 1722–1736 (2020)
L.A. Orloff, S.M. Wiseman, V.J. Bernet, T.J. Fahey, A.R. Shaha, M.L. Shindo, S.K. Snyder, B.C. Stack, J.B. Sunwoo, M.B. Wang, Thyroid 28, 830 (2018)
P. Han, B.J. Trinidad, J. Shi, ASN Neuro. 7, 1–9 (2015)
C. Cipriani, J.P. Bilezikian, J. Endocr. Soc. 5, bvab003 (2021)
L. Underbjerg, T. Sikjaer, L. Mosekilde, L. Rejnmark, J. Bone Miner. Res 28, 2277 (2013)
M.R. Rubin, Endocrinol. Metab. Clin. North Am. 47, 825 (2018)
B.C. Silva, M.R. Rubin, N.E. Cusano, J.P. Bilezikian, Osteoporos. Int. 28, 463 (2017)
W. Arlt, C. Fremerey, F. Callies, M. Reincke, P. Schneider, W. Timmermann, B. Allolio, Eur. J. Endocrinol. 146, 215 (2002)
A. Santonati, A. Palermo, E. Maddaloni, D. Bosco, A. Spada, F. Grimaldi, B. Raggiunti, R. Volpe, S. Manfrini, F. Vescini; Hypoparathyroidism AME Group. J. Clin. Endocrinol. Metab. 100, 3590 (2015)
T. Sikjaer, E. Moser, L. Rolighed, L. Underbjerg, L.S. Bislev, L. Mosekilde, L. Rejnmark, J. Bone Miner. Res 31, 1440 (2016)
M.L. Brandi, J.P. Bilezikian, D. Shoback, R. Bouillon, B.L. Clarke, R.V. Thakker, A.A. Khan, J.T. Potts, J. Clin. Endocrinol. Metab. 101, 2273 (2016)
Masi L. Refractory Hypoparathyroidism. In: Brandi M., Brown E. (eds), Hypoparathyroidism. (Springer, Milano, 2015), pp. 279–286. https://doi.org/10.1007/978-88-470-5376-2_25
A. Palermo, A. Santonati, G. Tabacco, D. Bosco, A. Spada, C. Pedone, B. Raggiunti, T. Doris, D. Maggi, F. Grimaldi, S. Manfrini, F. Vescini, J. Clin. Endocrinol. Metab. 103, 271 (2018)
K.K. Winer, J.A. Yanovski, G.B. Cutler, JAMA 276, 631 (1996)
K.K. Winer, S. Ye, E.M.N. Ferré, M.M. Schmitt, B. Zhang, G.B. Cutler, M.S. Lionakis, Bone 149, 115977 (2021)
K.K. Winer, B. Zhang, J.A. Shrader, D. Peterson, M. Smith, P.S. Albert, G.B. Cutler, J. Clin. Endocrinol. Metab. 97, 391 (2012)
K.K. Winer, K.A. Fulton, P.S. Albert, G.B. Cutler, J. Pediatr. 165, 556 (2014)
M. Puig-Domingo, G. Díaz, J. Nicolau, C. Fernández, S. Rueda, I. Halperin, Eur. J. Endocrinol. 159, 653 (2008)
K. Frey, S. Burger-Stritt, S. Horn, B. Willinger, M. Fassnacht, B. Allolio, S. Hahner, Exp. Clin. Endocrinol. Diabetes 122, poster 01_04 (2015)
M. Saraiva, A.C. Chaves, G. Assunção, J. Saraiva, R. Carvalho, Am. J. Case Rep. 22, e931739 (2021)
H.C. Zisser, Diabetes Ther. 1, 10 (2010)
A. Linglart, A. Rothenbuhler, I. Gueorgieva, P. Lucchini, C. Silve, P. Bougnères, J. Clin. Endocrinol. Metab. 96, 3308 (2011)
M. Laroche, D. Nigon, I. Gennero, S. Lassoued, J.M. Pouilles, F. Trémolières, M. Vallet, I. Tack, Ann. Biol. Clin. 74, 465 (2016)
K.K. Winer, C.W. Ko, J.C. Reynolds, K. Dowdy, M. Keil, D. Peterson, L.H. Gerber, C. McGarvey, G.B. Cutler, J. Clin. Endocrinol. Metab. 88, 4214 (2003)
G. Pellegriti, F. Frasca, C. Regalbuto, S. Squatrito, R. Vigneri, J. Cancer Epidemiol. 2013, 965212 (2013)
R. Mihai, R.V. Thakker, Eur. J. Endocrinol. 184, R165 (2021)
A.A. Khan, L. Rejnmark, M. Rubin, P. Schwarz, T. Vokes, B. Clarke, I. Ahmed, L. Hofbauer, C. Marcocci, U. Pagotto, A. Palermo, E. Eriksen, M. Brod, D. Markova, A. Smith, S. Pihl, S. Mourya, D.B. Karpf, A.D. Shu, J. Clin. Endocrinol. Metab. 107, 372–385 (2021)
F. Albright, R. Ellsworth, J. Clin. Invest. 7, 183 (1929)
K.K. Winer, J.A. Yanovski, B. Sarani, G.B. Cutler, J. Clin. Endocrinol. Metab. 83, 3480 (1998)
K.K. Winer, N. Sinaii, D. Peterson, B. Sainz, G.B. Cutler, J. Clin. Endocrinol. Metab. 93, 3389 (2008)
K.K. Winer, N. Sinaii, J. Reynolds, D. Peterson, K. Dowdy, G.B. Cutler, J. Clin. Endocrinol. Metab. 95, 2680 (2010)
M. Shah, I. Bancos, G.B. Thompson, M.L. Richards, J.L. Kasperbauer, B.L. Clarke, M.T. Drake, M.N. Stan, JAMA Otolaryngol. Head. Neck Surg. 141, 822 (2015)
M. Mannstadt, B.L. Clarke, T. Vokes, M.L. Brandi, L. Ranganath, W.D. Fraser, P. Lakatos, L. Bajnok, R. Garceau, L. Mosekilde, H. Lagast, D. Shoback, J.P. Bilezikian, Lancet Diabetes Endocrinol. 1, 275 (2013)
B.L. Clarke, T.J. Vokes, J.P. Bilezikian, D.M. Shoback, H. Lagast, M. Mannstadt, Endocrine 55, 273 (2017)
B. Cavaco, V. Leite, M.M. Loureiro, M.F. Ferreira, M.C. Pereira, M.A. Santos, L.G. Sobrinho, J. Endocrinol. Invest. 22, 829 (1999)
J. Satterwhite, M. Heathman, P.D. Miller, F. Marín, E.V. Glass, H. Dobnig, Calcif. Tissue Int 87, 485 (2010)
M.H. Samuels, J. Veldhuis, C. Cawley, R.J. Urban, M. Luther, R. Bauer, G. Mundy, J. Clin. Endocrinol. Metab. 77, 399 (1993)
H.M. Harms, U. Kaptaina, W.R. Külpmann, G. Brabant, R.D. Hesch, J. Clin. Endocrinol. Metab. 69, 843 (1989)
Z. Pekkolay, F. Kılınç, H. Soylu, B. Balsak, M. Güven, Ş. Altun Tuzcu, A.V. Kara, A.K. Tuzcu, Turkish J. Phys. Med. Rehabil. 65, 198 (2019)
J. Ilany, I. Vered, O. Cohen, Gynecol. Endocrinol. 29, 807 (2013)
G. Díaz-Soto, M. Mora-Porta, J. Nicolau, V. Perea, I. Halperin, M. Puig-Domingo, Horm. Metab. Res. 44, 708 (2012)
A. Haider, O. Symczyk, Y. Couso, T. Cater, S. Bryan, Case Rep. Endocrinol. 2021, 5593653 (2021)
J.L. Vahle, G.G. Long, G. Sandusky, M. Westmore, Y.L. Ma, M. Sato, Toxicol. Pathol. 32, 426 (2004)
E.B. Andrews, A.W. Gilsenan, K. Midkiff, B. Sherrill, Y. Wu, B.H. Mann, D. Masica, J. Bone Miner. Res 27, 2429 (2012)
C. Goujard, S. Salenave, K. Briot, P. Chanson, G. Grimon, P. Kamenický, Lancet 395, 1304 (2020)
Author information
Authors and Affiliations
Contributions
Conceptualization: K.A. and T.C.; validation: R.R. and T.B.; writing—original draft preparation: K.A. and T.C.; writing—review and editing: R.R., V.A., and T.B.; supervision: R.R. and T.B. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board of Aix-Marseille Université (01/01/2021).
Informed consent
Informed consent was obtained from all subjects involved in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aouchiche, K., Reynaud, R., Amodru, V. et al. Teriparatide administration by the Omnipod pump: preliminary experience from two cases with refractory hypoparathyroidism. Endocrine 76, 179–188 (2022). https://doi.org/10.1007/s12020-021-02978-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-021-02978-6