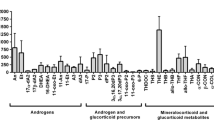
Abstract
Purpose
To review the literature assessing the diagnostic performance of urinary steroid profiling (USP) by high-performance liquid chromatography (LC–MS) or gas chromatography (GC) coupled to mass spectrometry (MS) in the evaluation of adrenal lesions, both in terms of functionality and malignancy.
Results
The evaluation of adrenal incidentalomas (AI) aims to rule out malignancy and hormone excess. Current diagnostic protocols have several limitations and include time consuming and relatively complicated multi-step processes in most cases. On the contrary, USP by LC–MS/MS or LC-GC/MS offer an easy, comprehensive and non-invasive assessment of adrenal steroid secretion. USP complements current workups used in the evaluation of AIs by improving our ability to identify malignancy and/or autonomous hormone secretion.
Conclusions
Urine steroid profiling by LC–MS/MS and GC–MS allows a thorough, non-invasive, assessment of adrenal steroidogenesis as a whole which complement the current evaluation of AIs, and holds a promising role in the diagnosis of autonomous cortisol secretion, primary aldosteronism, and adrenal malignancy.

Similar content being viewed by others
References
M. Araujo-Castro, M.A. Sampedro Núñez, M. Marazuela, Autonomous cortisol secretion in adrenal incidentalomas. Endocrine 64(1), 1–13 (2019). https://doi.org/10.1007/s12020-019-01888-y
M. Araujo-Castro, M. Iturregui Guevara, M. Calatayud Gutiérrez, P. Parra Ramírez, P. Gracia Gimeno, F.A. Hanzu et al. Practical guide on the initial evaluation, follow-up, and treatment of adrenal incidentalomas Adrenal Diseases Group of the Spanish Society of Endocrinology and Nutrition. Endocrinol. Diabetes Nutr. 67(6), 408–419 (2020). https://doi.org/10.1016/j.endinu.2020.03.002
M. Fassnacht, W. Arlt, I. Bancos, H. Dralle, J. Newell-Price, A. Sahdev et al. Management of adrenal incidentalomas: European Society of Endocrinology Clinical Practice Guideline in collaboration with the European Network for the Study of Adrenal Tumors. Eur. J. Endocrinol. 175(2), G1–G34 (2016). https://doi.org/10.1530/EJE-16-0467
J.M. Lee, M.K. Kim, S.H. Ko, J.M. Koh, B.Y. Kim, S.W. Kim et al. Clinical guidelines for the management of adrenal incidentaloma. Endocrinol Metab (Seoul). 32(2), 200–218 (2017). https://doi.org/10.3803/EnM.2017.32.2.200
M.M. Grumbach, B.M.K. Biller, G.D. Braunstein, K.K. Campbell, J. Aidan Carney, P.A. Godley et al. Management of the clinically inapparent adrenal mass (“incidentaloma”). Ann. Intern. Med. 138(5), 424–429 (2003). https://doi.org/10.7326/0003-4819-138-5-200303040-00013
A.H. Hamrahian, A.G. Ioachimescu, E.M. Remer, G. Motta-Ramirez, H. Bogabathina, H.S. Levin et al. Clinical utility of noncontrast computed tomography attenuation value (hounsfield units) to differentiate adrenal adenomas/hyperplasias from nonadenomas: Cleveland clinic experience. J. Clin. Endocrinol. Metab. 90(2), 871–877 (2005). https://doi.org/10.1210/jc.2004-1627
T.J. Cawood, P.J. Hunt, D. O’Shea, D. Cole, S. Soule, Recommended evaluation of adrenal incidentalomas is costly, has high false-positive rates and confers a risk of fatal cancer that is similar to the risk of the adrenal lesion becoming malignant; time for a rethink?. Eur. J. Endocrinol. 161(4), 513–527 (2009). https://doi.org/10.1530/EJE-09-0234
J.W. Funder, R.M. Carey, F. Mantero, M.H. Murad, M. Reincke, H. Shibata et al. The management of primary aldosteronism: Case detection, diagnosis, and treatment: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 101(5), 1889–1916 (2016). https://doi.org/10.1210/jc.2015-4061
E. Fischer, F. Beuschlein, M. Bidlingmaier, M. Reincke, Commentary on the Endocrine Society Practice Guidelines: Consequences of adjustment of antihypertensive medication in screening of primary aldosteronism. Rev. Endocr. Metab. Disord. 12(1), 43–48 (2011). https://doi.org/10.1007/s11154-011-9163-7
I. Chiodini, Diagnosis and treatment of subclinical hypercortisolism. J. Clin. Endocrinol. Metab. 96(5), 1223–1236 (2011). https://doi.org/10.1210/jc.2010-2722
A.A. Gheorghisan-galateanu, M. Carsote, A. Valea, Incidentaloma: from general practice to specific endocrine frame. J. Pak. Med. Assoc. 67(6), 917–922 (2017)
J.W. Funder, R.M. Carey, C. Fardella, C.E. Gomez-Sanchez, F. Mantero, M. Stowasser et al. Case detection, diagnosis, and treatment of patients with primary aldosteronism: an endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 93(9), 3266–3281 (2008). https://doi.org/10.1210/jc.2008-0104
M. Peppa, E. Boutati, C. Koliaki, N. Papaefstathiou, E. Garoflos, T. Economopoulos et al. Insulin resistance and metabolic syndrome in patients with nonfunctioning adrenal incidentalomas: a cause-effect relationship? Metabolism 59(10), 1435–1441 (2010). https://doi.org/10.1016/j.metabol.2010.01.007
T.M.A. Kerkhofs, M.N. Kerstens, I.P. Kema, T.P. Willems, H.R. Haak, Diagnostic value of urinary steroid profiling in the evaluation of adrenal tumors. Horm. Cancer 6(4), 168–175 (2015). https://doi.org/10.1007/s12672-015-0224-3
W. Arlt, M. Biehl, A.E. Taylor, S. Hahner, R. Libé, B.A. Hughes et al. Urine steroid metabolomics as a biomarker tool for detecting malignancy in adrenal tumors. J. Clin. Endocrinol. Metab. 96(12), 3775–3784 (2011). https://doi.org/10.1210/jc.2011-1565
L.I. Velikanova, Z.R. Shafigullina, A.A. Lisitsin, N.V. Vorokhobina, K. Grigoryan, E.A. Kukhianidze et al. Different types of urinary steroid profiling obtained by high-performance liquid chromatography and gas chromatography-mass spectrometry in patients with adrenocortical carcinoma. Horm. Cancer 7(5-6), 327–335 (2016). https://doi.org/10.1007/s12672-016-0267-0
Z.R. Shafigullina, L.I. Velikanova, N.V. Vorokhobina, S.B. Shustov, A.A. Lisitsin, E.V. Malevanaia et al. Urinary steroid profiling by gas chromatography mass spectrometry: Early features of malignancy in patients with adrenal incidentalomas. Steroids 135, 31–35 (2018). https://doi.org/10.1016/j.steroids.2018.04.006
V. Chortis, I. Bancos, T. Nijman, L.C. Gilligan, A.E. Taylor, C.L. Ronchi et al. Urine steroid metabolomics as a novel tool for detection of recurrent adrenocortical carcinoma. J. Clin. Endocrinol. Metab. 105(3), e307–e318 (2020). https://doi.org/10.1210/clinem/dgz141
A. Khorram-Manesh, H. Ahlman, S. Jansson, B. Wängberg, O. Nilsson, C.E. Jakobsson et al. Adrenocortical carcimona: surgery and mitotane for treatment and steroid profiles for follow-up. World J. Surg. 22(6), 605–611 (1998). https://doi.org/10.1007/s002689900442.
A. Kotłowska, E. Maliński, K. Sworczak, J. Kumirska, P. Stepnowski, The urinary steroid profile in patients diagnosed with adrenal incidentaloma. Clin. Biochem. 42(6), 448–454 (2009). https://doi.org/10.1016/j.clinbiochem.2008.12.027
A. Kotłowska, K. Sworczak, P. Stepnowski, Urine metabolomics analysis for adrenal incidentaloma activity detection and biomarker discovery. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 879(5–6), 359–363 (2011)
F. Holler, D.A. Heinrich, C. Adolf, B. Lechner, M. Bidlingmaier, G. Eisenhofer et al. Steroid profiling and immunohistochemistry for subtyping and outcome prediction in primary aldosteronism—a review. Curr. Hypertens. Rep. 21(10), 77 (2019). https://doi.org/10.1007/s11906-019-0985-0
W. Arlt, K. Lang, A.J. Sitch, A.S. Dietz, Y. Rhayem, I. Bancos et al. Steroid metabolome analysis reveals prevalent glucocorticoid excess in primary aldosteronism. JCI Insight 2(8), e93136 (2017). https://doi.org/10.1172/jci.insight.93136
L.S. Meyer, X. Wang, E. Sušnik, J. Burrello, A. Burrello, I. Castellano et al. Immunohistopathology and steroid profiles associated with biochemical outcomes after adrenalectomy for unilateral primary aldosteronism. Hypertension 72(3), 650–657 (2018). https://doi.org/10.1161/HYPERTENSIONAHA.118.11465
P. Mulatero, S.M. Di Cella, S. Monticone, D. Schiavone, M. Manzo, G. Mengozzi et al. 18-hydroxycorticosterone, 18-hydroxycortisol, and 18-oxocortisol in the diagnosis of primary aldosteronism and its subtypes. J. Clin. Endocrinol. Metab. 97(3), 881–889 (2012). https://doi.org/10.1210/jc.2011-2384
C. Rossi, I. Cicalini, S. Verrocchio, G. Di Dalmazi, L. Federici, I. Bucci, The potential of steroid profiling by mass spectrometry in the management of adrenocortical carcinoma. Biomedicines 8(9), 314 (2020). https://doi.org/10.3390/biomedicines8090314
G. Casals, F.A. Hanzu, Cortisol measurements in Cushing’s syndrome: immunoassay or mass spectrometry? Ann. Lab. Med. 40(4), 285–296 (2020). https://doi.org/10.3343/alm.2020.40.4.285
G. Aranda, M. Careaga, F.A. Hanzu, I. Patrascioiu, P. Ríos, M. Mora et al. Accuracy of immunoassay and mass spectrometry urinary free cortisol in the diagnosis of Cushing’s syndrome. Pituitary 19(5), 496–502 (2016). https://doi.org/10.1007/s11102-016-0730-5
P.C. White, Trastornos de la glándula suprarrenal suprarrenal. In: Nelson. Tratado de pediatría, pp. 2403–2420. (Elsevier, España, S.L.U. 2020)
W. Arlt, P.M. Stewart, Adrenal corticosteroid biosynthesis, metabolism, and action. Endocrinol. Metab. Clin. N. Am. 34(2), 293–313 (2005). https://doi.org/10.1016/j.ecl.2005.01.002
C. Shackleton, O.J. Pozo, J. Marcos, GC/MS in recent years has defined the normal and clinically disordered steroidome: will it soon be surpassed by LC/tandem MS in this role? J. Endocr. Soc. 2(8), 974–996 (2018)
N. Krone, B.A. Hughes, G.G. Lavery, P.M. Stewart, W. Arlt, C.H.L. Shackleton, Gas chromatography/mass spectrometry (GC/MS) remains a pre-eminent discovery tool in clinical steroid investigations even in the era of fast liquid chromatography tandem mass spectrometry (LC/MS/MS). J. Steroid Biochem. Mol. Biol. 121(3–5), 496–504 (2010)
C.H.L. Shackleton, Profiling steroid hormones and urinary steroids. J. Chromatogr. 379, 91–156 (1986). https://doi.org/10.1016/s0378-4347(00)80683-0
C.H.L. Shackleton, Mass spectrometry in the diagnosis of steroid-related disorders and in hypertension research. J. Steroid Biochem. Mol. Biol. 45(1-3), 127–140 (1993). https://doi.org/10.1016/0960-0760(93)90132-g
K.-H. Storbeck, L. Schiffer, E.S. Baranowski, V. Chortis, A. Prete, L. Barnard et al. Steroid metabolome analysis in disorders of adrenal steroid biosynthesis and metabolism. Endocr. Rev. 40(6), 1605–1625 (2019). https://doi.org/10.1210/er.2018-00262
J.W. Honour, E. Conway, R. Hodkinson, F. Lam, The evolution of methods for urinary steroid metabolomics in clinical investigations particularly in childhood. J. Steroid Biochem. Mol. Biol. 181, 28–51 (2018). https://doi.org/10.1016/j.jsbmb.2018.02.013
D.J. Handelsman, L. Wartofsky, Requirement for mass spectrometry sex steroid assays in the journal of clinical endocrinology and metabolism. J. Clin. Endocrinol. Metab. 98(10), 3971–3973 (2013). https://doi.org/10.1210/jc.2013-3375
D. Lopez, M.A. Luque-Fernandez, A. Steele, G.K. Adler, A. Turchin, A. Vaidya, “Nonfunctional” adrenal Tumors and the risk for incident diabetes and cardiovascular outcomes: a cohort study. Ann. Intern. Med. 165(8), 533–542 (2016). https://doi.org/10.7326/M16-0547
L.K. Nieman, B.M.K.K. Biller, J.W. Findling, J. Newell-Price, M.O. Savage, P.M. Stewart et al. The diagnosis of Cushing’s Syndrome: an Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 93(5), 1526–1540 (2008). https://doi.org/10.1210/jc.2008-0125
R. Gatti, G. Antonelli, M. Prearo, P. Spinella, E. Cappellin, E.F. De Palo, Cortisol assays and diagnostic laboratory procedures in human biological fluids. Clin. Biochem. 42(12), 1205–1217 (2009). https://doi.org/10.1016/j.clinbiochem.2009.04.011
A. Kotłowska, T. Puzyn, K. Sworczak, P. Stepnowski, P. Szefer, Metabolomic biomarkers in urine of cushing’s syndrome patients. Int. J. Mol. Sci. 18(2), 1–15. (2017)
J. Homoki, R. Holl, W.M. Teller, Urinary steroid profiles in Cushing’s syndrome and tumors of the adrenal cortex. Klin. Wochenschr. 65(15), 719–726 (1987). https://doi.org/10.1007/BF01736807
E. Kikuchi, H. Yanaihara, J. Nakashima, K. Homma, T. Ohigashi, H. Asakura et al. Urinary steroid profile in adrenocortical tumors. Biomed. Pharmacother. 54(1), 194s–197s (2000). https://doi.org/10.1016/s0753-3322(00)80043-8
P.M. Stewart, B.R. Walker, G. Holder, D. O’Halloran, C.H.L. Shackleton, 11 beta-Hydroxysteroid dehydrogenase activity in Cushing’s syndrome: explaining the mineralocorticoid excess state of the ectopic adrenocorticotropin syndrome. J. Clin. Endocrinol. Metab. 80(12), 3617–3620 (1995). https://doi.org/10.1210/jcem.80.12.8530609
J. Brossaud, D. Ducint, J.B. Corcuff, Urinary glucocorticoid metabolites: biomarkers to classify adrenal incidentalomas. Clin. Endocrinol. (Oxf.) 84(2), 236–43. (2016)
J.M. Hines, I. Bancos, C. Bancos, R.D. Singh, A.V. Avula, W.F. Young et al. High-resolution, accurate-mass (HRAM) mass spectrometry urine steroid profiling in the diagnosis of adrenal disorders. Clin. Chem. 63(12), 1824–1835 (2017). https://doi.org/10.1373/clinchem.2017.271106
F. Ceccato, G. Antonelli, M. Barbot, M. Zilio, L. Mazzai, R. Gatti et al. The diagnostic performance of urinary free cortisol is better than the cortisol: Cortisone ratio in detecting de novo Cushing’s Syndrome: The use of a LC-MS/MS method in routine clinical practice. Eur J Endocrinol 171(1), 1–7 (2014). https://doi.org/10.1530/EJE-14-0061
Araujo-castro M. Treatment of primary hyperaldosteronism. Med. Clin. (Barc). S0025-7753(20)30323-7,(2020). https://doi.org/10.1016/j.medcli.2020.04.029
T. Wannachalee, A.F. Turcu, Developments in primary aldosteronism subtyping using steroid profiling. Horm. Metab. Res. 52(6), 373–378 (2020). https://doi.org/10.1055/a-1141-3526
S. Ulick, J.D. Blumenfeld, S.A. Atlas, J.Z. Wang, E.D. Vaughan, The unique steroidogenesis of the aldosteronoma in the differential diagnosis of primary aldosteronism. J. Clin. Endocrinol. Metab. 76(4), 873–878 (1993). https://doi.org/10.1210/jcem.76.4.8473399
M. Wolley, M. Thuzar, M. Stowasser, Controversies and advances in adrenal venous sampling in the diagnostic workup of primary aldosteronism. Best. Pract. Res. Clin. Endocrinol. Metab. 34(3), 101400 (2020). https://doi.org/10.1016/j.beem.2020.101400
S. Ulick, M.D. Chu, Hypersecretion of a new corticosteroid, 18-hydroxycortisol in two types of adrenocortical hypertension. Clin. Exp. Hypertens. A. 4(9-10), 1771–1777 (1982). https://doi.org/10.3109/10641968209061640
R.D. Gordon, S.M. Hamlet, T.J. Tunny, C.E. Gomez‐Sanchez, L.S. Jayasinghed̊, Distinguishing aldosterone-producing adenoma from other forms of hyperaldosteronism and lateralizing the tumour pre-operatively. Clin. Exp. Pharmacol. Physiol. 13(4), 325–328 (1986). https://doi.org/10.1111/j.1440-1681.1986.tb00357.x
J.L. Benham, M. Eldoma, B. Khokhar, D.J. Roberts, D.M. Rabi, G.A. Kline, Proportion of patients with hypertension resolution following adrenalectomy for primary aldosteronism: a systematic review and meta-analysis. J. Clin. Hypertens. (Greenwich) 18(12), 1205–1212 (2016). https://doi.org/10.1111/jch.12916
M. Terzolo, A. Stigliano, I. Chiodini, P. Loli, L. Furlani, G. Arnaldi et al. AME position statement on adrenal incidentaloma. Eur. J. Endocrinol. 164(6), 851–870 (2011). https://doi.org/10.1530/EJE-10-1147
S. Minowada, K. Kinoshita, M. Hara, K. Isurugi, T. Uchikawa, T. Niijima, Measurement of urinary steroid profile in patients with adrenal tumor as a screening method for carcinoma. Endocrinol. Jpn. 32(1), 29–37 (1985). https://doi.org/10.1507/endocrj1954.32.29
S. Grondal, B. Eriksson, L. Hagenas, S. Werner, T. Curstedt, Steroid profile in urine: a useful tool in the diagnosis and follow up of adrenocortical carcinoma. Acta Endocrinol. (Copenh) 122(5), 656–663 (1990). https://doi.org/10.1530/acta.0.1220656
I. Bancos, W. Arlt, Diagnosis of a malignant adrenal mass: the role of urinary steroid metabolite profiling. Curr. Opin. Endocrinol. Diabetes Obes. 24(3), 200–207 (2017). https://doi.org/10.1097/MED.0000000000000333
S.C. Tiu, A.O.K. Chan, N.F. Taylor, C.Y. Lee, P.Y. Loung, C.H. Choi et al. Use of urinary steroid profiling for diagnosing and monitoring adrenocortical tumours. Hong Kong Med. J. 15(6), 463–470 (2009)
I. Bancos, A.E. Taylor, V. Chortis, A.J. Sitch, C. Jenkinson, C.J. Davidge-Pitts et al. Urine steroid metabolomics for the differential diagnosis of adrenal incidentalomas in the EURINE-ACT study: a prospective test validation study. Lancet Diabetes Endocrinol. 8(9), 773–781 (2020). https://doi.org/10.1016/S2213-8587(20)30218-7
H. Sasano, T. Suzuki, H. Nagura, T. Nishikawa, Steroidogenesis in human adrenocortical carcinoma: Biochemical activities, immunohistochemistry, and in situ hybridization of steroidogenic enzymes and histopathologic study in nine cases. Hum. Pathol. 24(4), 397–404 (1993). https://doi.org/10.1016/0046-8177(93)90088-x
V. Chortis, A.E. Taylor, P. Schneider, J.W. Tomlinson, B.A. Hughes, D.M. O’Neil et al. Mitotane therapy in adrenocortical cancer induces CYP3A4 and inhibits 5α-reductase, explaining the need for personalized glucocorticoid and androgen replacement. J. Clin. Endocrinol. Metab. 98(1), 161–171 (2013). https://doi.org/10.1210/jc.2012-2851
Funding
Supported by Grants “BECA SENDIMAD de Ayuda a la Investigación en Endocrinología, Nutrición y Diabetes 2019”, Sociedad de Endocrinología, Nutrición y Diabetes de Madrid, Madrid, Spain, and “Convocatoria intramural de ayudas a proyectos de investigación de investigadores noveles, investigadores clínicos asociados y/o grupos emergentes del Hospital Universitario Ramón y Cajal”, Instituto Ramón y Cajal de Investigación Sanitaria (IRYCIS), Madrid, Spain.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Araujo-Castro, M., Valderrábano, P., Escobar-Morreale, H.F. et al. Urine steroid profile as a new promising tool for the evaluation of adrenal tumors. Literature review. Endocrine 72, 40–48 (2021). https://doi.org/10.1007/s12020-020-02544-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-020-02544-6