Abstract
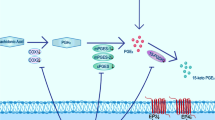
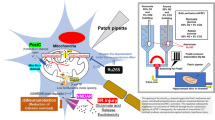
Cerebral ischemia is the primary basis of stroke, both sharing common pathogenic origins leading to irreversible brain damage if blood supply is not restored promptly. Existing evidence indicates that carbonic anhydrase (CA) inhibitors (CAIs) may impart therapeutic benefits against ischemia–reperfusion (I/R) pathology via the adenylyl cyclase-cyclic adenosine monophosphate (cAMP) pathway. We hypothesize that CAI and cAMP activation may enhance the therapeutic outcome against I/R conditions. In this investigation, the potential of dichlorphenamide (CAI) and the role of cAMP against ischemia–reperfusion injury were evaluated using a transient global cerebral I/R (tGCI/R) model. Swiss albino mice were subjected to bilateral common carotid artery occlusion (BCCAo) for 20 min and reperfusion (R) or sham surgery on day 1. Dichlorphenamide (DCPA, 20 mg/kg) and/or forskolin (cAMP agonist, 3 mg/kg) was administered intraperitoneally (i.p.) after BCCAo/R for 14 days. Results showed that tGCI/R impaired neurocognitive functions and lowered brain levels of cAMP and protein kinase A (PKA) that were ameliorated by DCPA and/or forskolin (FSK). DCPA and/or FSK attenuated tGCI/R-induced brain edema, blood–brain barrier dysfunction, oxidative-nitrosative stress, pro-inflammatory cytokines, acetylcholinesterase activity, cell death, and neurotransmitter imbalance (e.g., glutamate, γ-aminobutyric acid). The study showed that DCPA improved neurological and biochemical parameters against tGCI/R injury via cAMP-PKA-mediated activation of protective mechanisms. However, DCPA and FSK in combination showed much enhanced therapeutic outcomes against tGCI/R. Therefore, CA and cAMP present novel targets that may retard the progress of a transient ischemic attack to a full-blown stroke.






Similar content being viewed by others
Data Availability
The data used to support the findings of this study are available from the corresponding author upon a suitable request.
References
Abou-Rjeileh, U., & Contreras, G. A. (2021). Redox regulation of lipid mobilization in adipose tissues. Antioxidants (basel), 10(7), 1090. https://doi.org/10.3390/antiox10071090
Acin-Perez, R., Salazar, E., Kamenetsky, M., Buck, J., Levin, L. R., & Manfredi, G. (2009). Cyclic AMP produced inside mitochondria regulates oxidative phosphorylation. Cell Metabolism, 9(3), 265–276. https://doi.org/10.1016/j.cmet.2009.01.012
Ahad, M. A., Kumaran, K. R., Ning, T., Mansor, N. I., Effendy, M. A., Damodaran, T., & Hassan, Z. (2020). Insights into the neuropathology of cerebral ischemia and its mechanisms. Reviews in the Neurosciences, 31(5), 521–538. https://doi.org/10.1515/revneuro-2019-0099
Battisti-Charbonney, A., Fisher, J., & Duffin, J. (2011). The cerebrovascular response to carbon dioxide in humans. The Journal of Physiology, 589(12), 3039–3048. https://doi.org/10.1113/jphysiol.2011.206052
Cai, L., Chen, W. N., Li, R., Liu, M. M., Lei, C., Li, C. M., & Qiu, Y. Y. (2018). Acetazolamide protects rat articular chondrocytes from IL-1β-induced apoptosis by inhibiting the activation of NF-κB signal pathway. Canadian Journal of Physiology and Pharmacology, 96(11), 1104–1111. https://doi.org/10.1139/cjpp-2018-0334
Chen, Z. Q., Mou, R. T., Feng, D. X., Wang, Z., & Chen, G. (2017). The role of nitric oxide in stroke. Medical Gas Research, 7(3), 194–203. https://doi.org/10.4103/2045-9912.215750
Claiborne, A. (1985). Catalase activity. In R. A. Greenwald (Ed.), CRC Handbook of methods for oxygen radical research (pp. 283–284). CRC Press.
Deng, R. M., Liu, Y. C., Li, J. Q., Xu, J. G., & Chen, G. (2020). The role of carbon dioxide in acute brain injury. Medical Gas Research, 10(2), 81–84. https://doi.org/10.4103/2045-9912.285561
Dhingra, D., & Kumar, V. (2012). Memory-enhancing activity of palmatine in mice using elevated plus maze and Morris water maze. Advances in Pharmacological Sciences. https://doi.org/10.1155/2012/357368
Donkor, E. S. (2018). Stroke in the 21st century: A snapshot of the burden, epidemiology, and quality of life. Stroke Research and Treatment. https://doi.org/10.1155/2018/3238165
Ellman, G. L. (1959). Tissue sulfhydryl groups. Archives of Biochemistry and Biophysics, 82(1), 70–77. https://doi.org/10.1016/0003-9861(59)90090-6
Ellman, G. L., Courtney, K. D., Andres, V., & Featherstone, R. M. (1961). A new and rapid colorimetric determination of acetylcholinesterase activity. Biochemical Pharmacology, 7(2), 88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Ennaceur, A., & Delacour, J. (1988). A new one-trial test for neurobiological studies of memory in rats. 1: Behavioral data. Behavioural Brain Research, 31(1), 47–59. https://doi.org/10.1016/0166-4328(88)90157-x
Fontaine, B., & Phillips, L. H. (2016). A newly approved drug for a rare group of diseases Dichlorphenamide for Periodic Paralysis. Neurological Research, 86(15), 1366–1367. https://doi.org/10.1212/wnl.0000000000002430
Ghorai, S., Pulya, S., Ghosh, K., Panda, P., Ghosh, B., & Gayen, S. (2020). Structure-activity relationship of human carbonic anhydrase-II inhibitors: Detailed insight for future development as anti-glaucoma agents. Bioorganic Chemistry, 95, 103557. https://doi.org/10.1016/j.bioorg.2019.103557
Güneş, E., Aydin, H., & Nizamlioğlu, H. F. (2021). Investigation of the protective effect of acetazolamide and SLC-0111 on carbon tetrachloride-induced toxicity in fruit fly. Toxicology Reports, 8, 1300–1304. https://doi.org/10.1016/j.toxrep.2021.06.014
Guzman-Gutierrez, S. L., & Navarrete, A. (2009). Pharmacological exploration of the sedative mechanism of hesperidin identified as the active principle of Citrus sinensis flowers. Planta Medica, 75(4), 295–301. https://doi.org/10.1055/s-0029-1185306
Horeckerkornberg, B. L. A. (1948). The extinction coefficients of the reduced band of pyridine nucleotides. Journal of Biological Chemistry, 175(1), 385–390. https://doi.org/10.1016/s0021-9258(18)57268-9
Jin, X., Wang, R., Wang, H., Long, C., & Wang, H. (2015). Brain protection against ischemic stroke using choline as a new molecular bypass treatment. Acta Pharmacologica Sinica, 36, 1416–1425. https://doi.org/10.1038/aps.2015.104
Kawabori, M., & Yenari, M. A. (2015). Inflammatory responses in brain ischemia. Current Medicinal Chemistry, 22(10), 1258–1277. https://doi.org/10.2174/0929867322666150209154036
Kuriakose, D., & Xiao, Z. (2020). Pathophysiology and treatment of stroke: Present status and future perspectives. International Journal of Molecular Science, 21(20), 7609. https://doi.org/10.3390/ijms21207609
Lai, T. W., Zhang, S., & Wang, Y. T. (2014). Excitotoxicity and stroke: Identifying novel targets for neuroprotection. Progress in Neurobiology, 115, 157–188. https://doi.org/10.1016/j.pneurobio.2013.11.006
Lee, K. H., Cha, M., & Lee, B. H. (2020). Neuroprotective effect of antioxidants in the brain. International Journal of Molecular Science, 21(19), 7152. https://doi.org/10.3390/ijms21197152
Li, J., Chen, L., Guo, F., Han, X., & Ferreri, F. (2020). The effects of GABAergic system under cerebral ischemia: Spotlight on cognitive function. Neural Plasticity. https://doi.org/10.1155/2020/8856722
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. Journal of Biological Chemistry, 193(1), 265–275. https://doi.org/10.1016/S0021-9258(19)52451-6
Mehan, S., Rahi, S., Tiwari, A., Kapoor, T., Rajdev, K., Sharma, R., Khera, H., Kosey, S., Kukkar, U., & Dudi, R. (2020). Adenylate cyclase activator forskolin alleviates intracerebroventricular propionic acid-induced mitochondrial dysfunction of autistic rats. Neural Regeneration Research, 15(6), 1140–1149. https://doi.org/10.4103/1673-5374.270316
Neumann, J. T., Cohan, C. H., Dave, K. R., Wright, C. B., & Perez-Pinzon, M. A. (2013). Global cerebral ischemia: Synaptic and cognitive dysfunction. Current Drug Targets, 14(1), 20–35. https://doi.org/10.2174/138945013804806514
Ohkawa, H., Ohishi, N., & Yagi, K. (1979). Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry, 95(2), 351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Phipps, M. S., & Cronin, C. A. (2020). Management of acute ischemic stroke. British Medical Journal, 368, l6983. https://doi.org/10.1136/bmj.l6983
Raker, V. K., Becker, C., & Steinbrink, K. (2016). The cAMP pathway as therapeutic target in autoimmune and inflammatory diseases. Frontiers in Immunology, 7, 123. https://doi.org/10.3389/fimmu.2016.00123
Rinaldi, L., Pozdniakova, S., Jayarajan, V., Troidl, C., Abdallah, Y., Aslam, M., & Ladilov, Y. (2019). Protective role of soluble adenylyl cyclase against reperfusion-induced injury of cardiac cells. Biochimica Et Biophysica Acta (BBA) - Molecular Basis of Disease, 1865(1), 252–260. https://doi.org/10.1016/j.bbadis.2018.07.021
Sastry, K. V. H., Moudgal, R. P., Mohan, J., Tyagi, J. S., & Rao, G. S. (2002). Spectrophotometric determination of serum nitrite and nitrate by copper–cadmium alloy. Analytical Biochemistry, 306(1), 79–82. https://doi.org/10.1006/abio.2002.5676
Schaar, K. L., Brenneman, M. M., & Savitz, S. I. (2010). Functional assessments in the rodent stroke model. Experimental & Translational Stroke Medicine, 2, 13. https://doi.org/10.1186/2040-7378-2-13
Scherbakov, N., Sandek, A., & Doehner, W. (2015). Stroke-related sarcopenia: Specific characteristics. Journal of the American Medical Directors Association, 16, 272–276. https://doi.org/10.1016/j.jamda.2014.12.007
Scozzafava, A., & Supuran, C. T. (2014). Glaucoma and the applications of carbonic anhydrase inhibitors. In S. Frost & R. McKenna (Eds.), Carbonic anhydrase Mechanism, regulation, links to disease, and industrial applications. Subcellular Biochemistry (vol 75). Springer.
Torring, M. S., Holmgaard, K., Hessellund, A., Aalkjaer, C., & Bek, T. (2009). The Vasodilating effect of acetazolamide and dorzolamide involves mechanisms other than carbonic anhydrase inhibition. Investigative Ophthalmology & Visual Science, 50, 345–351. https://doi.org/10.1167/iovs.08-2435
Wahul, A. B., Joshi, P. C., Kumar, A., & Chakravarty, S. (2018). Transient global cerebral ischemia differentially affects cortex, striatum and hippocampus in Bilateral Common Carotid Arterial occlusion (BCCAo) mouse model. Journal of Chemical Neuroanatomy, 92, 1–15. https://doi.org/10.1016/j.jchemneu.2018.04.006
Winterbourn, C. C., Hawkins, R. E., Brian, M., & Carrell, R. W. (1975). The estimation of red cell superoxide dismutase activity. Journal of Laboratory and Clinical Medicine, 85(2), 337–341.
Yuan, Y., Shan, X., Men, W., Zhai, H., Qiao, X., Geng, L., & Li, C. (2020). The effect of crocin on memory, hippocampal acetylcholine level, and apoptosis in a rat model of cerebral ischemia. Biomedicine & Pharmacotherapy, 130, 110543. https://doi.org/10.1016/j.biopha.2020.110543
Yucel, M. A. (2009). The possible role of CO2 in producing a post-stimulus CBF and BOLD undershoot. Frontiers in Neuroengineering, 1, 1–14. https://doi.org/10.3389/neuro.14.007.2009
Acknowledgements
The authors are appreciative of Swift School of Pharmacy, Rajpura, for facilitating the required research services.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
MK contributed to conceptualization, design of methodology, supervision, project administration, data analysis, writing—original draft, writing—review & editing, and validation. KK was involved in investigation and data curation. TGS was involved in resources and data validation.
Corresponding author
Ethics declarations
Competing Interest
The authors declare none.
Ethics Approval
The research protocol was approved by Institutional Animal Ethics Committee vide approval reference no. SSP/IAEC/2019/007 on date: 17-11-2019. Animals were housed within the institutional establishment (Animal House Facility) registered under CPCSEA (Regd. 1616/PO/Re/S/12/CPCSEA). All the animal tests were performed following the guiding principles of CPCSEA (Committee for the Purpose of Control and Supervision of Experiments on Animals), Ministry of Environment and Forests (Animal Welfare), Government of India (GOI), New Delhi.
Consent to Participate
Not applicable.
Consent for Publication
It is affirmed that all the authors have seen and agreed to the submission and publication of the research article and their inclusion of name(s) as co-author(s).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, M., Kaur, K. & Singh, T.G. Neuroprotective Effects of Carbonic Anhydrase Inhibition and Cyclic Adenosine Monophosphate Activation in Mouse Model of Transient Global Cerebral Ischemia and Reperfusion. Neuromol Med 25, 217–229 (2023). https://doi.org/10.1007/s12017-022-08728-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-022-08728-9