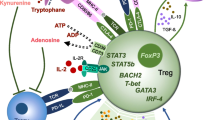
Abstract
The immune system has evolved to respond to pathogens (nonself) and unresponsive to self-antigens (tolerance). During the development of T and B cells in the thymus and bone marrow, respectively, self-reactive T and B cells are deleted by a process of apoptosis (both T and B cells) and become unresponsive to self-antigen by receptor editing (for B cells). However, few self-reactive T cells are leaked into the periphery. A number of mechanisms are responsible to ensure that self-reactive T and B cells remain unresponsive to self-antigens. In the central tolerance, major mechanisms include apoptosis (for T cells) and receptor editing (for B cells), and in the peripheral tolerance, a major mechanism appears to be regulated by Treg cells. In T cell central tolerance, one of the most important molecules is a transcription factor, autoimmune regulator, which is selectively expressed in medullary thymic epithelial cells (mTECs) and constitutively regulates the transcription of hundreds of self-antigens in mTECs, thereby inducing central tolerance, negative selection, and Treg differentiation from some self-reactive thymocytes. Primary immunodeficiency diseases are a group of monogenic diseases where mutations of certain genes have resulted in the loss of central and/or peripheral tolerance. As a result autoimmunity and autoimmune diseases are common among patients with primary immunodeficiency diseases. Here, we have provided a comprehensive review of the mechanisms of central and peripheral tolerance and autoimmune manifestations and mechanisms of autoimmunity in primary immunodeficiency diseases.
Similar content being viewed by others
References
Nortarangelo LD, Gambineri E, Badolato R (2006) Immunodeficiencies with autoimmune consequences. Adv Immunol 89:321–370
Westerberg LS, Klein C, Snapper SB (2008) Breakdown of T cell tolerance and autoimmunity in primary immunodeficiency—lessons learned from monogenic disorders in mice and men. Curr Opin Immunol 20:646–654
Ulmanen I, Halonen M, Ilmarinen T, Peltonen L (2005) Monogenic autoimmune diseases—lessons of self-tolerance. Curr Opin Immunol 17:609–615
Derbinski J, Schulte A, Kyewski B, Klein L (2001) Promiscuous gene expression in medullary thymic epithelial cells mirrors the peripheral self. Nat Immunol 2:1032–1039
Mathis D, Benoist C (2009) Aire. Ann Rev Immuno 27:287–312
Hogquist KA, Baldwin TA, Jameson SC (2005) Central tolerance: learning self-control in the thymus. Nat Rev Immunol 5:772–782
Baba T, Nakamoto Y, Mukaida N (2009) Crucial contribution of thymic Sirp alpha + conventional dendritic cells to central tolerance against blood-borne antigens I a CCR2-dependent manner. J Immunol 183:3053–3063
Pillai S, Mattoo H, Cariappa A (2011) B cells and autoimmunity. Curr Opin Immunol 23:721–731
Mauri C, Bosma A (2012) Immune regulatory function of B cells. Ann Rev Immunol 30:221–241
Sakaguchi S, Yamaguchi T, Nomura T, Ono R (2008) Regulatory T cells and immune tolerance. Cell 133:775–787
Moraes-Vasconcelos D, Costa-Carvalho BT, Torgerson TR, Ochs HD (2008) Primary immune deficiency disorders presenting as autoimmune diseases: IPEX and APECED. J Clin Immunol 28(Suppl 1):S11–S19
Brunkow ME, Jeffrey EW, Hjerrild KA, Paeper B, Clark LB, Yasayko SA et al (2001) Disruption of a new forkhead/winged-helix protein, scurfin, results in fatal lymphoproliferative disorder of the scurfy mouse. Nat Genet 27:68–73
Notarangelo JD (2010) Primary immunodeficiencies. J Allergy Clin Immunol 125:S182–S194
Finish-German APECED Consortium (1997) An autoimmune disease, APECED, caused by mutations in a novel gene featuring two PHD-type zing-finger domains. Nat Genet 17:399–403
Villaseñor J, Benoist C, Mathis D (2005) AIRE and APECED: molecular insights into an autoimmune disease. Immunol Rev 204:156–164
Wildin RS, Smyk-Pearson S, Filipovich AH (2002) Clinical and molecular features of the immunodysregulation, polyendocrinopathy, enteropathy, X linked (IPEX) syndrome. J Med Gent 39:537–545
Bosco JB, Gupta S (2008) Disorders of apoptosis: mechanisms for autoimmunity in primary immunodeficiency diseases. J Clin Immunol 28:S20–S28
Poliani PL, Facchetti F, Ravanini M, Gennery AR, Villa A, Roifman CM et al (2009) Eraly defects in T cell development severely affect distribution and maturation of thymic stromal cells: possible implications for the pathophysiology of Omenn syndrome. Blood 114:105–108
Nadeau K, Hwa V, Rosenfeld RG (2011) STAT5b deficiency: an unsuspected cause of growth failure, immunodeficiency, and sever pulmonary disease. J Ped 158:701–708
Cohen AC, Nadeau KC, Tu W, Hwa V, Dionis K, Dezrodnik I et al (2006) Decreased accumulation and regulatory function of CD4+ CD25 (high) T cells in human STAT5b deficiency. J Immunol 177:2770–2774
Willerford DM, Chen J, Ferry JA, Davidson L, Ma A, Alt FW (1995) Interleukin 2 receptor alpha chain regulate the size and content of the peripheral lymphoid compartment. Immunity 3:521–530
Sharfe N, Dadi HK, Shahar M, Roifman CM (1997) Human immune disorder arising from mutation of the α chain of the interleukin-2 receptor. Proc Natl Acad Sci (USA) 94:3168–3171
Roifman CM (2000) Human IL-2 receptor alpha chain deficiency. Pediatr Res 48:6–11
Feske S, Picard C, Fischer A (2010) Immunodeficiency due to mutations in ORAI1 and STIM1. Clin Immunol 135:162–182
Picard C, McCarl CA, Papolos A, Khalil S, Luthy K, Hivroz C, LeDeist F et al (2009) STIM1 mutation associated with a syndrome of immunodeficiency and autoimmunity. N Eng J Med 360:1971–1980
Jesus AA, Duarte AJ, Oliveira JB (2008) Autoimmunity in hyper-IgM syndrome. J Clin Immunol 28(Suppl 1):S62–S66
Notarangelo LD, Miao CH, Ochs HD (2008) Wiskott–Aldrich syndrome. Curr Opin Hematol 15:30–36
Freeman AF, Holland SH (2008) The hyper-IgE syndromes. Immunol Allergy Clin North Am 28:277–291
De Ravin SS, Naumann N, Cowen EW et al (2008) Chronic granulomatous disease as a risk for autoimmune disease. J Allergy Clin Immunol 122:1097–1103
Kraaij MD, Savageb NDL, van der Kooija SW, Koekkoeka K, Wangc J, van den Bergd JM et al (2010) Induction of regulatory T cells by macrophages is dependent on production of reactive oxygen species. Proc Natl Acad Sci (USA) 107:17686–17691
Collin M, Bigley V, Haniffa M, Hambleton S (2011) Human dendritic cell deficiency: the missing ID? Nature Rev Immunol 11:575–583
Barilla-LaBarca MI, Atkinson JP (2003) Rheumatic syndromes associated with complement deficiency. Curr Opin Rheumatol 15:55–60
Unsworth DJ (2008) Complement deficiency and disease. J Clin Pathol 61:1013–1017
Knight AK, Cunningham-Rundles C (2006) Inflammatory and autoimmune complications of common variable immune deficiency. Autoimmune Rev 5:156–159
Baccheli C, Buckridge S, Thrasher AJ, Gasper HB (2007) Molecular defects in common variable immunodeficiency. J Exp Immunol 149:401–409
Grimbacher B, Hutloff A, Schlesier M, Glocker E, Warnatz K, Drager R et al (2003) Homozygous loss of ICOS is associated with adult onset common variable immunodeficiency. Nat Immunol 4:261–268
Van Zelm MC, Reisli I, van der Burg M, Castano D, van Noesel CJ, van Tol MJ et al (2006) An antibody deficiency syndrome due to mutation in the CD19 gene. N Eng J Med 354:1901–1912
Castigli E, Wilson SA, Garibyan I, Rachid R, Bonilla F, Schneider L et al (2005) TACI is mutant in common variable immunodeficiency and IgA deficiency. Nat Gen 37:829–834
Losi CG, Silini A, Fiorini C, Soresina A, Meini A, Meini A, Ferrari S et al (2005) Mutational analysis of human BAFF receptor TNFRSF13C (BAFF-R) in patients with common variable immunodeficiency. J Clin Immunol 25:496–502
Yu GP, Chiang D, Song SJ, Hoyte EG, Huang J, Vanisharn C et al (2009) Regulator T cell dysfunction in subjects with common variable immunodeficiency complicated by autoimmune disease. Clin Immunol 131:240–253
Yel L (2010) Selective IgA deficiency. J Clin Immunol 30:10–16
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gupta, S., Louis, A.G. Tolerance and Autoimmunity in Primary Immunodeficiency Disease: a Comprehensive Review. Clinic Rev Allerg Immunol 45, 162–169 (2013). https://doi.org/10.1007/s12016-012-8345-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-012-8345-8