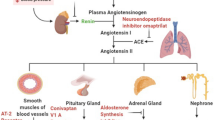
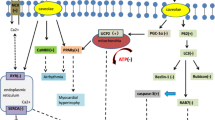
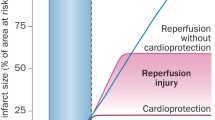
Abstract
Both clinical and experimental findings at the molecular, cellular, tissue, organ and systematic levels have depicted the presence of a contemporary regulatory machinery namely compensation in various forms of cardiovascular diseases. Compensation is believed to be present and regulated within the scope of a biological entity and represents the initiation of dyshomeostasis. Compensation can be identified in multiple categories and organs in cardiovascular diseases at multiple levels. The capacity to reduce the unfavorable pathological compensation may be a criterion to evaluate the therapeutic effectiveness for cardiovascular diseases. This mini-review tries to take compensation into consideration in the understanding of onset and progression of cardiovascular diseases in particular, and thus, better or optimal therapeutic approaches may be achieved for the prevention and management of cardiovascular diseases.


Similar content being viewed by others
References
Gustafson-Wagner, E. A., Sinn, H. W., Chen, Y. L., Wang, D. Z., Reiter, R. S., Lin, J. L., et al. (2007). Loss of mXinalpha, an intercalated disk protein, results in cardiac hypertrophy and cardiomyopathy with conduction defects. American Journal of Physiology. Heart and Circulatory Physiology, 293, H2680–H2692.
Izumo, S., Lompre, A. M., Matsuoka, R., Koren, G., Schwartz, K., Nadal-Ginard, B., et al. (1987). Myosin heavy chain messenger RNA and protein isoform transitions during cardiac hypertrophy. Interaction between hemodynamic and thyroid hormone-induced signals. Journal of Clinical Investigation, 79, 970–977.
Anilkumar, N., Sirker, A., & Shah, A. M. (2009). Redox sensitive signaling pathways in cardiac remodeling, hypertrophy and failure. Frontiers in Bioscience, 14, 3168–3187.
Li, H. H., Willis, M. S., Lockyer, P., Miller, N., McDonough, H., Glass, D. J., et al. (2007). Atrogin-1 inhibits Akt-dependent cardiac hypertrophy in mice via ubiquitin-dependent coactivation of Forkhead proteins. Journal of Clinical Investigation, 117, 3211–3223.
Schwartz, K., de la Bastie, D., Bouveret, P., Oliviero, P., Alonso, S., & Buckingham, M. (1986). Alpha-skeletal muscle actin mRNA’s accumulate in hypertrophied adult rat hearts. Circulation Research, 59, 551–555.
Xia, Y., Wen, H. Y., Young, M. E., Guthrie, P. H., Taegtmeyer, H., & Kellems, R. E. (2003). Mammalian target of rapamycin and protein kinase a signaling mediate the cardiac transcriptional response to glutamine. Journal of Biological Chemistry, 278, 13143–13150.
Bourajjaj, M., Armand, A. S., da Costa Martins, P. A., Weijts, B., van der Nagel, R., Heeneman, S., et al. (2008). NFATc2 is a necessary mediator of calcineurin-dependent cardiac hypertrophy and heart failure. Journal of Biological Chemistry, 283, 22295–22303.
Skurk, C., Izumiya, Y., Maatz, H., Razeghi, P., Shiojima, I., Sandri, M., et al. (2005). The FOXO3a transcription factor regulates cardiac myocyte size downstream of AKT signaling. Journal of Biological Chemistry, 280, 20814–20823.
Bush, E. W., Hood, D. B., Papst, P. J., Chapo, J. A., Minobe, W., Bristow, M. R., et al. (2006). Canonical transient receptor potential channels promote cardiomyocyte hypertrophy through activation of calcineurin signaling. Journal of Biological Chemistry, 281, 33487–33496.
Seth, M., Sumbilla, C., Mullen, S. P., Lewis, D., Klein, M. G., Hussain, A., et al. (2004). Sarco(endo)plasmic reticulum Ca2+ ATPase (SERCA) gene silencing and remodeling of the Ca2+ signaling mechanism in cardiac myocytes. Proceedings of the National academy of Sciences of the United States of America, 101, 16683–16688.
Abe, Y., Ono, K., Kawamura, T., Wada, H., Kita, T., Shimatsu, A., et al. (2007). Leptin induces elongation of cardiac myocytes and causes eccentric left ventricular dilatation with compensation. American Journal of Physiology. Heart and Circulatory Physiology, 292, H2387–H2396.
Karmazyn, M., Purdham, D. M., Rajapurohitam, V., & Zeidan, A. (2007). Leptin as a cardiac hypertrophic factor: a potential target for therapeutics. Trends in Cardiovascular Medicine, 17, 206–211.
Ren, J. (2004). Leptin and hyperleptinemia—from friend to foe for cardiovascular function. Journal of Endocrinology, 181, 1–10.
Muraski, J. A., Fischer, K. M., Wu, W., Cottage, C. T., Quijada, P., Mason, M., et al. (2008). Pim-1 kinase antagonizes aspects of myocardial hypertrophy and compensation to pathological pressure overload. Proceedings of the National academy of Sciences of the United States of America, 105, 13889–13894.
Honsho, S., Nishikawa, S., Amano, K., Zen, K., Adachi, Y., Kishita, E., et al. (2009). Pressure-mediated hypertrophy and mechanical stretch induces IL-1 release and subsequent IGF-1 generation to maintain compensative hypertrophy by affecting Akt and JNK pathways. Circulation Research, 105, 1149–1158.
Kolwicz, S. C., Jr, & Tian, R. (2011). Glucose metabolism and cardiac hypertrophy. Cardiovascular Research, 90, 194–201.
Yamashita, H., Bharadwaj, K. G., Ikeda, S., Park, T. S., & Goldberg, I. J. (2008). Cardiac metabolic compensation to hypertension requires lipoprotein lipase. American Journal of Physiology-Endocrinology and Metabolism, 295, E705–E713.
Lebrin, F. & Mummery, C. L. (2008). Endoglin-mediated vascular remodeling: mechanisms underlying hereditary hemorrhagic telangiectasia. Trends in Cardiovascular Medicine, 18, 25–32.
Sanchez-Elsner, T., Botella, L. M., Velasco, B., Langa, C., & Bernabeu, C. (2002). Endoglin expression is regulated by transcriptional cooperation between the hypoxia and transforming growth factor-beta pathways. Journal of Biological Chemistry, 277, 43799–43808.
ten Dijke, P., Goumans, M. J., & Pardali, E. (2008). Endoglin in angiogenesis and vascular diseases. Angiogenesis, 11, 79–89.
Young, L. H., Renfu, Y., Russell, R., Hu, X., Caplan, M., Ren, J., et al. (1997). Low-flow ischemia leads to translocation of canine heart GLUT-4 and GLUT-1 glucose transporters to the sarcolemma in vivo. Circulation, 95, 415–422.
Young, L. H., Russell, R. R., I. I. I., Yin, R., Caplan, M. J., Ren, J., Bergeron, R., et al. (1999). Regulation of myocardial glucose uptake and transport during ischemia and energetic stress. American Journal of Cardiology, 83, 25H–30H.
Yang, J., & Holman, G. D. (2005). Insulin and contraction stimulate exocytosis, but increased AMP-activated protein kinase activity resulting from oxidative metabolism stress slows endocytosis of GLUT4 in cardiomyocytes. Journal of Biological Chemistry, 280, 4070–4078.
Severino, A., Campioni, M., Straino, S., Salloum, F. N., Schmidt, N., Herbrand, U., et al. (2007). Identification of protein disulfide isomerase as a cardiomyocyte survival factor in ischemic cardiomyopathy. Journal of the American College of Cardiology, 50, 1029–1037.
Feng, H. Z., Chen, M., Weinstein, L. S., & Jin, J. P. (2008). Removal of the N-terminal extension of cardiac troponin I as a functional compensation for impaired myocardial beta-adrenergic signaling. Journal of Biological Chemistry, 283, 33384–33393.
Atluri, P., Morine, K. J., Liao, G. P., Panlilio, C. M., Berry, M. F., Hsu, V. M., et al. (2007). Ischemic heart failure enhances endogenous myocardial apelin and APJ receptor expression. Cellular & Molecular Biology Letters, 12, 127–138.
Chong, A. Y., Caine, G. J., Freestone, B., Blann, A. D., & Lip, G. Y. (2004). Plasma angiopoietin-1, angiopoietin-2, and angiopoietin receptor tie-2 levels in congestive heart failure. Journal of the American College of Cardiology, 43, 423–428.
Peng, W., Zhang, Y., Zhu, W., Cao, C. M., & Xiao, R. P. (2009). AMPK and TNF-alpha at the crossroad of cell survival and death in ischaemic heart. Cardiovascular Research, 84, 1–3.
Seta, Y., Shan, K., Bozkurt, B., Oral, H., & Mann, D. L. (1996). Basic mechanisms in heart failure: the cytokine hypothesis. Journal of Cardiac Failure, 2, 243–249.
Mann, D. L. (2002). Inflammatory mediators and the failing heart: past, present, and the foreseeable future. Circulation Research, 91, 988–998.
Valgimigli, M., Rigolin, G. M., Fucili, A., Porta, M. D., Soukhomovskaia, O., Malagutti, P., et al. (2004). CD34+ and endothelial progenitor cells in patients with various degrees of congestive heart failure. Circulation, 110, 1209–1212.
Nonaka-Sarukawa, M., Yamamoto, K., Aoki, H., Nishimura, Y., Tomizawa, H., Ichida, M., et al. (2007). Circulating endothelial progenitor cells in congestive heart failure. International Journal of Cardiology, 119, 344–348.
Bupha-Intr, T., Wattanapermpool, J., Pena, J. R., Wolska, B. M., & Solaro, R. J. (2007). Myofilament response to Ca2+ and Na+/H+ exchanger activity in sex hormone-related protection of cardiac myocytes from deactivation in hypercapnic acidosis. American Journal of Physiology: Regulatory, Integrative and Comparative Physiology, 292, R837–R843.
Son, S. M. (2007). Role of vascular reactive oxygen species in development of vascular abnormalities in diabetes. Diabetes Research and Clinical Practice, 77(Suppl 1), S65–S70.
Brownlee, M. (2001). Biochemistry and molecular cell biology of diabetic complications. Nature, 414, 813–820.
Cheng, Z., Tseng, Y., & White, M. F. (2010). Insulin signaling meets mitochondria in metabolism. Trends in Endocrinology and Metabolism, 21, 589–598.
Sutak, R., Xu, X., Whitnall, M., Kashem, M. A., Vyoral, D., & Richardson, D. R. (2008). Proteomic analysis of hearts from frataxin knockout mice: marked rearrangement of energy metabolism, a response to cellular stress and altered expression of proteins involved in cell structure, motility and metabolism. Proteomics, 8, 1731–1741.
Duan, D. (2009). Phenomics of cardiac chloride channels: the systematic study of chloride channel function in the heart. Journal of Physiology, 587, 2163–2177.
Yamamoto-Mizuma, S., Wang, G. X., Liu, L. L., Schegg, K., Hatton, W. J., Duan, D., et al. (2004). Altered properties of volume-sensitive osmolyte and anion channels (VSOACs) and membrane protein expression in cardiac and smooth muscle myocytes from Clcn3-/- mice. Journal of Physiology, 557, 439–456.
Riordan, M. M., & Kovacs, S. J. (2008). Elucidation of spatially distinct compensatory mechanisms in diastole: radial compensation for impaired longitudinal filling in left ventricular hypertrophy. Journal of Applied Physiology, 104, 513–520.
AlDabal, L., & BaHammam, A. S. (2010). Cheyne-stokes respiration in patients with heart failure. Lung, 188, 5–14.
Fueger, P. T., Li, C. Y., Ayala, J. E., Shearer, J., Bracy, D. P., Charron, M. J., et al. (2007). Glucose kinetics and exercise tolerance in mice lacking the GLUT4 glucose transporter. Journal of Physiology, 582, 801–812.
Ziegler, M. A., Distasi, M. R., Bills, R. G., Miller, S. J., Alloosh, M., Murphy, M. P., et al. (2010). Marvels, mysteries, and misconceptions of vascular compensation to peripheral artery occlusion. Microcirculation, 17, 3–20.
Bacci, D., Valecchi, D., Sgambati, E., Gulisano, M., Conti, A. A., Molino-Lova, R., et al. (2008). Compensatory collateral circles in vertebral and carotid artery occlusion. Italian Journal of Anatomy and Embryology, 113, 265–271.
Gross, C. G. (2009). Three before their time: Neuroscientists whose ideas were ignored by their contemporaries. Experimental Brain Research, 192, 321–334.
Amit, I., Citri, A., Shay, T., Lu, Y., Katz, M., Zhang, F., et al. (2007). A module of negative feedback regulators defines growth factor signaling. Nature Genetics, 39, 503–512.
Harbuz, M. (2002). Neuroendocrine function and chronic inflammatory stress. Experimental Physiology, 87, 519–525.
Kikkawa, Y., Kameda, K., Hirano, M., Sasaki, T., & Hirano, K. (2010). Impaired feedback regulation of the receptor activity and the myofilament Ca2+ sensitivity contributes to increased vascular reactiveness after subarachnoid hemorrhage. Journal of Cerebral Blood Flow and Metabolism, 30, 1637–1650.
Mancia, G., Seravalle, G., Giannattasio, C., Bossi, M., Preti, L., Cattaneo, B. M., et al. (1992). Reflex cardiovascular control in congestive heart failure. American Journal of Cardiology, 69, 17G–22G.
Nollo, G., Faes, L., Porta, A., Pellegrini, B., Ravelli, F., Del, G. M., et al. (2002). Evidence of unbalanced regulatory mechanism of heart rate and systolic pressure after acute myocardial infarction. American Journal of Physiology. Heart and Circulatory Physiology, 283, H1200–H1207.
Brandman, O., & Meyer, T. (2008). Feedback loops shape cellular signals in space and time. Science, 322, 390–395.
Besedovsky, H., & Sorkin, E. (1977). Network of immune-neuroendocrine interactions. Clinical and Experimental Immunology, 27, 1–12.
Nabel, E. G. (2003). Cardiovascular disease. New England Journal of Medicine, 349, 60–72.
Jackson, J. G., & Pereira-Smith, O. M. (2006). Primary and compensatory roles for RB family members at cell cycle gene promoters that are deacetylated and downregulated in doxorubicin-induced senescence of breast cancer cells. Molecular and Cellular Biology, 26, 2501–2510.
Goldstein, J. L., & Brown, M. S. (1977). The low-density lipoprotein pathway and its relation to atherosclerosis. Annual Review of Biochemistry, 46, 897–930.
Fan, X. J., Yu, H., & Ren, J. (2011). Homeostasis and compensatory homeostasis: bridging Western medicine and traditional chinese medicine. Current Cardiology Reviews, 7, 43–46.
Aviram, M. (1999). Macrophage foam cell formation during early atherogenesis is determined by the balance between pro-oxidants and anti-oxidants in arterial cells and blood lipoproteins. Antioxidants & Redox Signaling, 1, 585–594.
Lecour, S., Smith, R. M., Woodward, B., Opie, L. H., Rochette, L., & Sack, M. N. (2002). Identification of a novel role for sphingolipid signaling in TNF alpha and ischemic preconditioning mediated cardioprotection. Journal of Molecular and Cellular Cardiology, 34, 509–518.
Nakano, M., Knowlton, A. A., Dibbs, Z., & Mann, D. L. (1998). Tumor necrosis factor-alpha confers resistance to hypoxic injury in the adult mammalian cardiac myocyte. Circulation, 97, 1392–1400.
Siragy, H. (1999). Angiotensin II receptor blockers: review of the binding characteristics. American Journal of Cardiology, 84, 3S–8S.
Calhoun, D. A., Jones, D., Textor, S., Goff, D. C., Murphy, T. P., Toto, R. D., et al. (2008). Resistant hypertension: Diagnosis, evaluation, and treatment: a scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Circulation, 117, e510–e526.
Smiley, D., & Umpierrez, G. (2007). Metformin/rosiglitazone combination pill (Avandamet) for the treatment of patients with Type 2 diabetes. Expert Opinion on Pharmacotherapy, 8, 1353–1364.
Blansfield, J. A., Caragacianu, D., Alexander, H. R., I. I. I., Tangrea, M. A., Morita, S. Y., Lorang, D., et al. (2008). Combining agents that target the tumor microenvironment improves the efficacy of anticancer therapy. Clinical Cancer Research, 14, 270–280.
Dorrell, M. I., Aguilar, E., Scheppke, L., Barnett, F. H., & Friedlander, M. (2007). Combination angiostatic therapy completely inhibits ocular and tumor angiogenesis. Proceedings of the National academy of Sciences of the United States of America, 104, 967–972.
Acknowledgments
We thank Dr. Kehong Zhang from the Ivy Editing for insightful comments and suggestions.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Fan, XJ., Ren, J. Compensation: A Contemporary Regulatory Machinery in Cardiovascular Diseases?. Cardiovasc Toxicol 12, 275–284 (2012). https://doi.org/10.1007/s12012-012-9167-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-012-9167-x