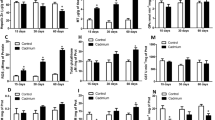
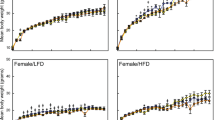
Abstract
Cadmium is a nonessential transition metal considered one of the more hazardous environmental contaminants. The population is chronically exposed to this metal at low concentrations, designated as the LOAEL (lowest observable adverse effect level) dose. We aimed to investigate whether oral subacute exposure to cadmium LOAEL disrupts hormonal and metabolic effects of the liver-adipose axis in Wistar rats. Fifty male Wistar rats were separated into two groups: control (standard normocalorie diet + water free of cadmium) and cadmium (standard normocalorie diet + drinking water with 32.5 ppm CdCl2). After 1 month, zoometry, a serum lipid panel, adipokines, and proinflammatory cytokines were evaluated. Tests of glucose and insulin tolerance (ITT) and insulin resistance were performed. Histological studies on structure, triglyceride distribution, and protein expression of the insulin pathway were performed in the liver and retroperitoneal adipose tissue. In both tissues, the cadmium, triglyceride, glycogen, and proinflammatory cytokine contents were also quantified. The cadmium group developed dyslipidemia, glucose intolerance, hyperinsulinemia, hyperleptinemia, inflammation, and selective insulin resistance in the liver and adipose tissue. In the liver, glycogen synthesis was diminished, while de novo lipogenesis increased, which was associated with low GSK3β-pS9 and strong expression of SREBP-1c. Dysfunctional adipose tissue was observed with hypertrophy and lipolysis, without changes in SREBP-1c expression and low glycogen synthesis. Both tissues accumulated cadmium and developed inflammation. In conclusion, oral subacute cadmium LOAEL dose exposure induces inflammation, insulin signaling modifications, an early insulin resistance stage (insensibility), and impairment of the hormonal and metabolic liver-adipose axis in Wistar rats.







Similar content being viewed by others
References
ATSDR (2012) Toxicological profile for cadmium. Agency Toxic Subst Dis Regist Public Heal Serv US Dep Heal Hum Serv 2012:1–487
Moulis J-M, Thévenod F (2010) New perspectives in cadmium toxicity: an introduction. BioMetals 23:763–768. https://doi.org/10.1007/s10534-010-9365-6
Järup L, Åkesson A (2009) Current status of cadmium as an environmental health problem. Toxicol Appl Pharmacol 238:201–208. https://doi.org/10.1016/j.taap.2009.04.020
Satarug S, Nishijo M, Ujjin P, Moore MR (2018) Chronic exposure to low-level cadmium induced zinc-copper dysregulation. J Trace Elem Med Biol 46:32–38. https://doi.org/10.1016/j.jtemb.2017.11.008
Treviño S, Waalkes MP, Flores Hernández JA et al (2015) Chronic cadmium exposure in rats produces pancreatic impairment and insulin resistance in multiple peripheral tissues. Arch Biochem Biophys 583:27–35. https://doi.org/10.1016/j.abb.2015.07.010
Sarmiento-Ortega VE, Treviño S, Flores-Hernández JÁ et al (2017) Changes on serum and hepatic lipidome after a chronic cadmium exposure in Wistar rats. Arch Biochem Biophys 635:52–59. https://doi.org/10.1016/j.abb.2017.10.003
Santamaria-Juarez C, Atonal-Flores F, Diaz A et al (2020) Aortic dysfunction by chronic cadmium exposure is linked to multiple metabolic risk factors that converge in anion superoxide production. Arch Physiol Biochem 1–9. https://doi.org/10.1080/13813455.2020.1726403
Barregard L, Bergström G, Fagerberg B (2013) Cadmium exposure in relation to insulin production, insulin sensitivity and type 2 diabetes: a cross-sectional and prospective study in women. Environ Res 121:104–109. https://doi.org/10.1016/J.ENVRES.2012.11.005
Fitzgerald R, Olsen A, Nguyen J et al (2020) Pancreatic islets accumulate cadmium in a rodent model of cadmium-induced hyperglycemia. Int J Mol Sci 22:1–16. https://doi.org/10.3390/IJMS22010360
Chapatwala KD, Rajanna E, Desaiah D (1980) Cadmium Induced Changes in Gluconeogenic Enzymes in Rat Kidney and Liver. Drug Chem Toxicol 3:407–420. https://doi.org/10.3109/01480548009030129
Chapatwala KD, Hobson M, Desaiah D, Rajanna B (1982) Effect of cadmium on hepatic and renal gluconeogenic enzymes in female rats. Toxicol Lett 12:27–34. https://doi.org/10.1016/0378-4274(82)90194-1
Jacquet A, Arnaud J, Hininger-Favier I, Hazane-Puch F, Couturier K, Lénon M, Lamarche F, Ounnas F, Fontaine E, Jean-Marc Moulis CD (2018) Impact of chronic and low cadmium exposure of rats: sex specific disruption of glucose metabolism. Chemosphere 207:764–773. https://doi.org/10.1016/j.chemosphere.2018.05.099
Ayoub N, Mantash H, Dhaini HR et al (2021) Serum cadmium levels and risk of metabolic syndrome: a cross-sectional study. Biol Trace Elem Res 199:3625–3633. https://doi.org/10.1007/S12011-020-02502-3
Jacquet A, Barbeau D, Arnaud J, Hijazi S, Hazane-Puch F, Lamarche F, Quiclet C, Couturier K, Fontaine E, Jean-Marc Moulis CD (2019) Impact of maternal low-level cadmium exposure on glucose and lipid metabolism of the litter at different ages after weaning. Chemosphere 219:109–121. https://doi.org/10.1016/j.chemosphere.2018.11.137
Han JC, Park SY, Hah BG, Choi GH, Kim YK, Kwon TH, Kim EK, Lachaal M, Jung WL CY (2003) Cadmium induces impaired glucose tolerance in rat by down-regulating GLUT4 expression in adipocytes. Arch Biochem Biophys 413:213–220. https://doi.org/10.1016/s0003-9861(03)00120-6
Treviño S, Diaz A (2020) Vanadium and insulin: Partners in metabolic regulation. J Inorg Biochem 208:111094
Moslehi A, Hamidi-Zad Z (2018) Role of SREBPs in liver diseases: a mini-review. J Clin Transl Hepatol 6:332–338
Wang Y, Viscarra J, Kim SJ, Sul HS (2015) Transcriptional regulation of hepatic lipogenesis. Nat Rev Mol Cell Biol 16:678–689
Mashek DG (2013) Hepatic fatty acid trafficking: multiple forks in the road. Adv Nutr 4:697–710
Tiwari S, Siddiqi SA (2012) Intracellular trafficking and secretion of VLDL. Arterioscler Thromb Vasc Biol 32:1079–1086
Onal G, Kutlu O, Gozuacik D, Dokmeci Emre S (2017) Lipid droplets in health and disease. Lipids Health Dis 16:128
Olzmann JA, Carvalho P (2019) Dynamics and functions of lipid droplets. Nat Rev Mol Cell Biol 20:137–155
Kioumourtzoglou D, Pryor PR, Gould GW, Bryant NJ (2015) Alternative routes to the cell surface underpin insulin-regulated membrane trafficking of GLUT4. J Cell Sci 128:2423–2429. https://doi.org/10.1242/jcs.166561
Kuri-Harcuch W, Velez-delValle C, Vazquez-Sandoval A et al (2019) A cellular perspective of adipogenesis transcriptional regulation. J Cell Physiol 234:1111–1129
de Sá Mota P, Richard AJ, Hang H, Stephens JM (2017) Transcriptional regulation of adipogenesis. Compr Physiol 7:635–674. https://doi.org/10.1002/cphy.c160022
Frühbeck G, Catalán V, Rodríguez A, Gómez-Ambrosi J (2018) Adiponectin-leptin ratio: a promising index to estimate adipose tissue dysfunction. Relation with obesity-associated cardiometabolic risk. Adipocyte 7:57–62
Blüher M, Mantzoros CS (2015) From leptin to other adipokines in health and disease: facts and expectations at the beginning of the 21st century. Metabolism. 64:131–145
Turer AT, Scherer PE (2012) Adiponectin: mechanistic insights and clinical implications. Diabetologia 55:2319–2326
Achari AE, Jain SK (2017) Adiponectin, a therapeutic target for obesity, diabetes, and endothelial dysfunction. Int J Mol Sci 18:1321
Thomas D, Apovian C (2017) Macrophage functions in lean and obese adipose tissue. Metabolism. 72:120–143
Chait A, den Hartigh LJ (2020) Adipose tissue distribution, inflammation and its metabolic consequences, including diabetes and cardiovascular disease. Front Cardiovasc Med 7:22. https://doi.org/10.3389/fcvm.2020.00022
Matulewicz N, Karczewska-Kupczewska M (2016) Insulin resistance and chronic inflammation. Postepy Hig Med Dosw (Online) 70:1245–1258
Asghar A, Sheikh N (2017) Role of immune cells in obesity induced low grade inflammation and insulin resistance. Cell Immunol 315:18–26
Sarmiento-Ortega V, Brambila E, Flores-Hernández J et al (2018) The NOAEL metformin dose is ineffective against metabolic disruption induced by chronic cadmium exposure in Wistar rats. Toxics 6:55. https://doi.org/10.3390/toxics6030055
Martin SS, Blaha MJ, Elshazly MB et al (2013) Comparison of a novel method vs the Friedewald equation for estimating low-density lipoprotein cholesterol levels from the standard lipid profile. JAMA - J Am Med Assoc 310:2061–2068. https://doi.org/10.1001/jama.2013.280532
Abdul-Ghani MA, Molina-Carrion M, Jani R et al (2008) Adipocytes in subjects with impaired fasting glucose and impaired glucose tolerance are resistant to the anti-lipolytic effect of insulin. Acta Diabetol 45:147–150. https://doi.org/10.1007/s00592-008-0033-z
Treviño S, Sánchez-Lara E, Sarmiento-Ortega VE et al (2015) Hypoglycemic, lipid-lowering and metabolic regulation activities of metforminium decavanadate (H2Metf) 3 [V10O28]·8H2O using hypercaloric-induced carbohydrate and lipid deregulation in Wistar rats as b. J Inorg Biochem 147:85–92. https://doi.org/10.1016/j.jinorgbio.2015.04.002
Sarmiento-Ortega VE, Moroni-González D, Díaz A et al (2021) Sodium metavanadate treatment improves glycogen levels in multiple tissues in a model of metabolic syndrome caused by chronic cadmium exposure in Wistar rats. BioMetals. https://doi.org/10.1007/s10534-020-00276-8
Treviño S, Díaz A, Sánchez-Lara E et al (2018) Pharmacological and Toxicological Threshold of Bisammonium Tetrakis 4-(N,N-Dimethylamino)pyridinium Decavanadate in a Rat Model of Metabolic Syndrome and Insulin Resistance. Bioinorg Chem Appl 2018:2151079. https://doi.org/10.1155/2018/2151079
Schwartz GG, Il’yasova D, Ivanova A (2003) Urinary cadmium, impaired fasting glucose, and diabetes in the NHANES III. Diabetes Care 26:468–470. https://doi.org/10.2337/diacare.26.2.468
Xiong X, Zhang Y, Xing H, Xu S (2019) Ameliorative Effect of Selenomethionine on Cadmium-Induced Hepatocyte Apoptosis via Regulating PI3K/AKT Pathway in Chickens. Biol Trace Elem Res. https://doi.org/10.1007/s12011-019-01858-5
Hildebrand J, Thakar S, Watts TL et al (2019) The impact of environmental cadmium exposure on type 2 diabetes risk: A protocol for an overview of systematic reviews. Syst Rev 8:309. https://doi.org/10.1186/s13643-019-1246-7
Fatima G, Raza AM, Hadi N et al (2019) Cadmium in human diseases: it’s more than just a mere metal. Indian J Clin Biochem 34:371–378
Shin D, Eom YS, Chon S et al (2019) Factors influencing insulin sensitivity during hyperinsulinemic-euglycemic clamp in healthy Korean male subjects. Diabetes, Metab Syndr Obes Targets Ther 12:469–476. https://doi.org/10.2147/DMSO.S195350
Samuel VT, Shulman GI (2012) Mechanisms for insulin resistance: Common threads and missing links. Cell 148:852–871
Samuel VT, Shulman GI (2016) The pathogenesis of insulin resistance: integrating signaling pathways and substrate flux. J Clin Invest 126:12–22. https://doi.org/10.1172/JCI77812
Horita S, Nakamura M, Suzuki M et al (2016) Selective insulin resistance in the kidney. Biomed Res Int 2016:5825170
Gehart H, Kumpf S, Ittner A, Ricci R (2010) MAPK signalling in cellular metabolism: Stress or wellness? EMBO Rep 11:834–840
Fang X, Yu SX, Lu Y et al (2000) Phosphorylation and inactivation of glycogen synthase kinase 3 by protein kinase A. Proc Natl Acad Sci U S A 97:11960–11965. https://doi.org/10.1073/pnas.220413597
Petersen MC, Shulman GI (2018) Mechanisms of insulin action and insulin resistance. Physiol Rev 98:2133–2223
Eberlé D, Hegarty B, Bossard P et al (2004) SREBP transcription factors: master regulators of lipid homeostasis. Biochimie 86:839–848
Owen JL, Zhang Y, Bae SH et al (2012) Insulin stimulation of SREBP-1c processing in transgenic rat hepatocytes requires p70 S6-kinase. Proc Natl Acad Sci U S A 109:16184–16189. https://doi.org/10.1073/pnas.1213343109
Haeusler RA, Hartil K, Vaitheesvaran B et al (2014) Integrated control of hepatic lipogenesis versus glucose production requires FoxO transcription factors. Nat Commun 5:5190. https://doi.org/10.1038/ncomms6190
Benedict M, Zhang X (2017) Non-alcoholic fatty liver disease: an expanded review. World J Hepatol 9:715–732
Marchisello S, Di Pino A, Scicali R et al (2019) Pathophysiological, molecular and therapeutic issues of nonalcoholic fatty liver disease: An overview. Int J Mol Sci 20:1948. https://doi.org/10.3390/ijms20081948
Tanase DM, Gosav EM, Costea CF et al (2020) The intricate relationship between type 2 diabetes mellitus (T2DM), insulin resistance (IR), and nonalcoholic fatty liver disease (NAFLD). J Diabetes Res 2020:3920196
Bashir N, Shagirtha K, Manoharan V, Miltonprabu S (2019) The molecular and biochemical insight view of grape seed proanthocyanidins in ameliorating cadmium-induced testes-toxicity in rat model: Implication of PI3K/Akt/Nrf-2 signaling. Biosci Rep 39:BSR20180515. https://doi.org/10.1042/BSR20180515
Shati AA, Alfaifi MY (2019) Trans-resveratrol inhibits tau phosphorylation in the brains of control and cadmium chloride-treated rats by activating PP2A and PI3K/Akt induced-inhibition of GSK3β. Neurochem Res 44:357–373. https://doi.org/10.1007/s11064-018-2683-8
Xin C, Guangliang S, Qing Z et al (2020) Astilbin protects chicken peripheral blood lymphocytes from cadmium-induced necroptosis via oxidative stress and the PI3K/Akt pathway. Ecotoxicol Environ Saf 190:110064. https://doi.org/10.1016/j.ecoenv.2019.110064
Rajanna B, Hobson M, Reese J et al (1984) Chronic hepatic and renal toxicity by cadmium in rats. Drug Chem Toxicol 7:229–241. https://doi.org/10.3109/01480548409035105
Larregle EV, Varas SM, Oliveros LB et al (2008) Lipid metabolism in liver of rat exposed to cadmium. Food Chem Toxicol 46:1786–1792. https://doi.org/10.1016/j.fct.2008.01.018
Prabu SM, Shagirtha K, Renugadevi J (2010) Amelioration of cadmium-induced oxidative stress, impairment in lipids and plasma lipoproteins by the combined treatment with quercetin and α-tocopherol in rats. J Food Sci 75:T132–T140. https://doi.org/10.1111/j.1750-3841.2010.01757.x
Hammarstedt A, Gogg S, Hedjazifar S et al (2018) Impaired adipogenesis and dysfunctional adipose tissue in human hypertrophic obesity. Physiol Rev 98:1911–1941. https://doi.org/10.1152/physrev.00034.2017
Zhu S, Sun F, Li W et al (2011) Apelin stimulates glucose uptake through the PI3K/Akt pathway and improves insulin resistance in 3T3-L1 adipocytes. Mol Cell Biochem 353:305–313. https://doi.org/10.1007/s11010-011-0799-0
Tsuchiya A, Kanno T, Nishizaki T (2014) PI3 kinase directly phosphorylates Akt1/2 at Ser473/474 in the insulin signal transduction pathway. J Endocrinol 220:49–59. https://doi.org/10.1530/JOE-13-0172
Yaribeygi H, Simental-Mendía LE, Barreto GE, Sahebkar A (2019) Metabolic effects of antidiabetic drugs on adipocytes and adipokine expression. J Cell Physiol 234:16987–16997
Summers SA, Whiteman EL, Birnbaum MJ (2000) Insulin signaling in the adipocyte. Int J Obes 24:S67–S70. https://doi.org/10.1038/sj.ijo.0801509
Arner E, Westermark PO, Spalding KL et al (2010) Adipocyte turnover: relevance to human adipose tissue morphology. Diabetes 59:105–109. https://doi.org/10.2337/db09-0942
White U, Ravussin E (2019) Dynamics of adipose tissue turnover in human metabolic health and disease. Diabetologia 62:17–23
Horton JD, Shimomura I, Ikemoto S et al (2003) Overexpression of sterol regulatory element-binding protein-1a in mouse adipose tissue produces adipocyte hypertrophy, increased fatty acid secretion, and fatty liver. J Biol Chem 278:36652–36660. https://doi.org/10.1074/jbc.M306540200
Frühbeck G, Méndez-Giménez L, Fernández-Formoso JA et al (2014) Regulation of adipocyte lipolysis. Nutr Res Rev 27:63–93. https://doi.org/10.1017/S095442241400002X
Zhang Y, Su X, Dong Y et al (2020) Cytological and functional characteristics of fascia adipocytes in rats: a unique population of adipocytes. Biochim Biophys Acta Mol Cell Biol Lipids 1865:158585. https://doi.org/10.1016/j.bbalip.2019.158585
Rutkowski JM, Stern JH, Scherer PE (2015) The cell biology of fat expansion. J Cell Biol 208:501–512
Haczeyni F, Bell-Anderson KS, Farrell GC (2018) Causes and mechanisms of adipocyte enlargement and adipose expansion. Obes Rev 19:406–420
Kusminski CM, Bickel PE, Scherer PE (2016) Targeting adipose tissue in the treatment of obesity-associated diabetes. Nat Rev Drug Discov 15:639–660
Eckel N, Mühlenbruch K, Meidtner K et al (2015) Characterization of metabolically unhealthy normal-weight individuals: risk factors and their associations with type 2 diabetes. Metabolism 64:862–871. https://doi.org/10.1016/J.METABOL.2015.03.009
Tinkov AA, Filippini T, Ajsuvakova OP et al (2018) Cadmium and atherosclerosis: a review of toxicological mechanisms and a meta-analysis of epidemiologic studies. Environ Res 162:240–260. https://doi.org/10.1016/J.ENVRES.2018.01.008
Hossein-Khannazer N, Azizi G, Eslami S et al (2020) The effects of cadmium exposure in the induction of inflammation. Immunopharmacol Immunotoxicol 42:1–8
Moynihan M, Telléz-Rojo MM, Colacino J et al (2019) Prenatal cadmium exposure is negatively associated with adiposity in girls not boys during adolescence. Front Public Healh 7:61. https://doi.org/10.3389/FPUBH.2019.00061
Van Cauwenberghe J, Den Hond E, Schoeters G et al (2014) Prenatal exposure to environmental contaminants and body composition at age 7-9 years. Environ Res 132:24–32. https://doi.org/10.1016/J.ENVRES.2014.03.019
Reilly SM, Saltiel AR (2017) Adapting to obesity with adipose tissue inflammation. Nat Rev Endocrinol 13:633–643
Longo M, Zatterale F, Naderi J et al (2019) Adipose tissue dysfunction as determinant of obesity-associated metabolic complications. Int J Mol Sci 20:2358. https://doi.org/10.3390/ijms20092358
Gustafson B, Gogg S, Hedjazifar S et al (2009) Inflammation and impaired adipogenesis in hypertrophic obesity in man. Am J Physiol Endocrinol Metab 297:E999–E1003
Laurencikiene J, Van Harmelen V, Nordström EA et al (2007) NF-κB is important for TNF-α-induced lipolysis in human adipocytes. J Lipid Res 48:1069–1077. https://doi.org/10.1194/jlr.M600471-JLR200
Lumeng CN, Bodzin JL, Saltiel AR (2007) Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J Clin Invest 117:175–184. https://doi.org/10.1172/JCI29881
Oliveira AG, Araujo TG, Carvalho BM et al (2013) Acute exercise induces a phenotypic switch in adipose tissue macrophage polarization in diet-induced obese rats. Obesity 21:2545–2556. https://doi.org/10.1002/oby.20402
Park SE, Park CY, Sweeney G (2015) Biomarkers of insulin sensitivity and insulin resistance: past, present and future. Crit Rev Clin Lab Sci 52:180–190
Olszowski T, Baranowska-Bosiacka I, Gutowska I, Chlubek D (2012) Pro-inflammatory properties of cadmium. Acta Biochim Pol 59:475–482
Kataranovski M, Janković S, Kataranovski D et al (2009) Gender differences in acute cadmium-induced systemic inflammation in rats. Biomed Environ Sci 22:1–7. https://doi.org/10.1016/S0895-3988(09)60014-3
Zhang J, Wang Y, Fu L et al (2019) Chronic cadmium exposure induced hepatic cellular stress and inflammation in aged female mice. J Appl Toxicol 39:498–509. https://doi.org/10.1002/jat.3742
Huang YY, Xia MZ, Wang H et al (2014) Cadmium selectively induces MIP-2 and COX-2 through PTEN-mediated akt activation in RAW264.7 cells. Toxicol Sci 138:310–321. https://doi.org/10.1093/toxsci/kfu013
Ali I, Damdimopoulou P, Stenius U, Halldin K (2015) Cadmium at nanomolar concentrations activates Raf-MEK-ERK1/2 MAPKs signaling via EGFR in human cancer cell lines. Chem Biol Interact 231:44–52. https://doi.org/10.1016/j.cbi.2015.02.014
Souza V, del Carmen Escobar M, Bucio L et al (2009) NADPH oxidase and ERK1/2 are involved in cadmium induced-STAT3 activation in HepG2 cells. Toxicol Lett 187:180–186. https://doi.org/10.1016/j.toxlet.2009.02.021
Escobar MDC, Souza V, Bucio L et al (2009) MAPK activation is involved in cadmium-induced Hsp70 expression in HepG2 cells. Toxicol Mech Methods 19:503–509. https://doi.org/10.3109/15376510903325670
Liu C, Zhu Y, Lu Z et al (2020) Cadmium induces acute liver injury by inhibiting Nrf2 and the role of NF-κB, NLRP3, and MAPKS signaling pathway. Int J Environ Res Public Health 17:138. https://doi.org/10.3390/ijerph17010138
Genchi G, Sinicropi MS, Lauria G et al (2020) The effects of cadmium toxicity. Int J Environ Res Public Health 17:3782. https://doi.org/10.3390/IJERPH17113782
Rani A, Kumar A, Lal A, Pant M (2014) Cellular mechanisms of cadmium-induced toxicity: a review. Int J Environ Health Res 24:378–399. https://doi.org/10.1080/09603123.2013.835032
Ashrafizadeh M, Ahmadi Z, Farkhondeh T, Samarghandian S (2020) Back to nucleus: combating with cadmium toxicity using Nrf2 Signaling pathway as a promising therapeutic target. Biol Trace Elem Res 197:52–62. https://doi.org/10.1007/S12011-019-01980-4
Dai S, Yin Z, Yuan G et al (2013) Quantification of metallothionein on the liver and kidney of rats by subchronic lead and cadmium in combination. Environ Toxicol Pharmacol 36:1207–1216. https://doi.org/10.1016/J.ETAP.2013.10.003
Sabolić I, Breljak D, Skarica M, Herak-Kramberger CM (2010) Role of metallothionein in cadmium traffic and toxicity in kidneys and other mammalian organs. Biometals 23:897–926. https://doi.org/10.1007/S10534-010-9351-Z
Gupta DK, Pena LB, Romero-Puertas MC et al (2017) NADPH oxidases differentially regulate ROS metabolism and nutrient uptake under cadmium toxicity. Plant Cell Environ 40:509–526. https://doi.org/10.1111/PCE.12711
Okoye CN, MacDonald-Jay N, Kamunde C (2019) Effects of bioenergetics, temperature and cadmium on liver mitochondria reactive oxygen species production and consumption. Aquat Toxicol 214:105264. https://doi.org/10.1016/J.AQUATOX.2019.105264
Cannino G, Ferruggia E, Luparello C, Rinaldi AM (2009) Cadmium and mitochondria. Mitochondrion 9:377–384. https://doi.org/10.1016/J.MITO.2009.08.009
Beyrami M, Karimi E, Oskoueian E (2020) Synthesized chrysin-loaded nanoliposomes improves cadmium-induced toxicity in mice. Environ Sci Pollut Res Int 27:40643–40651. https://doi.org/10.1007/S11356-020-10113-7
Zhang D, Gao J, Zhang K et al (2012) Effects of chronic cadmium poisoning on Zn, Cu, Fe, Ca, and metallothionein in liver and kidney of rats. Biol Trace Elem Res 149:57–63. https://doi.org/10.1007/S12011-012-9394-9
Pal M, Febbraio MA, Lancaster GI (2016) The roles of c-Jun NH2-terminal kinases (JNKs) in obesity and insulin resistance. J Physiol 594:267–279. https://doi.org/10.1113/JP271457
Solinas G, Becattini B (2017) JNK at the crossroad of obesity, insulin resistance, and cell stress response. Mol Metab 6:174–184
Schmitz-Peiffer C, Laybutt DR, Burchfield JG et al (2007) Inhibition of PKCε Improves Glucose-Stimulated Insulin Secretion and Reduces Insulin Clearance. Cell Metab 6:320–328. https://doi.org/10.1016/j.cmet.2007.08.012
Acknowledgments
The authors thank Vicerrectoria de Investigación y Posgrado (VIEP; TRMS-NAT19-1) through Ygnacio Martınez Laguna, CONACyT, and the “Sistema Nacional de Investigadores” of Mexico for the financial support of this research project (VESO, 533291) and Dr. Francisco Ramos Collazo (Bioterio “Claude Bernard”, BUAP) for his assistance and the donation of the animals used in this work. We express our gratitude to the Clinical Laboratory “Los Ángeles” for the biochemical determinations. The workgroup is grateful to Jesús Alberto Abrego Maldonado and Álan Josué Rodríguez Velázquez for their help to histological processing and analysis. We thank Professor Thomas Edwards, Ph.D., for editing the English language text.
Funding
CONACyT and the “Sistema Nacional de Investigadores” of Mexico for the financial support of this research project [VESO, 533291].
Author information
Authors and Affiliations
Contributions
Victor Enrique Sarmiento-Ortega, Eduardo Brambila, and Samuel Treviño designed the study and wrote the protocol. Victor Enrique Sarmiento-Ortega, Alfonso Díaz, Diana Moroni-González, and Samuel Treviño performed the experiments. Victor Enrique Sarmiento-Ortega, Eduardo Brambila, and Samuel Treviño managed the literature searches and analysis. Eduardo Brambila and Diana Moroni-González undertook the statistical analysis. Alfonso Díaz, Victor Enrique Sarmiento-Ortega, Eduardo Brambila, and Samuel Treviño wrote the first draft of the manuscript. All contributing authors have approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sarmiento-Ortega, V.E., Moroni-González, D., Díaz, A. et al. Oral Subacute Exposure to Cadmium LOAEL Dose Induces Insulin Resistance and Impairment of the Hormonal and Metabolic Liver-Adipose Axis in Wistar Rats. Biol Trace Elem Res 200, 4370–4384 (2022). https://doi.org/10.1007/s12011-021-03027-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-03027-z