Abstract
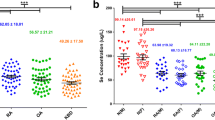
In this study, we aimed to explore the effectiveness of selenium on the chondrocyte glycoprotein glycosylation which plays important roles in the pathogenesis of Kashin–Beck disease (KBD). Cartilage samples were collected from KBD patients after total knee replacement surgery. Chondrocytes were cultured with sodium selenium. The group of chondrocytes which were cultured without adding sodium selenium was considered as control group. Lectin microarray was used to screen the differences in lectin levels between KBD and KBD with selenium groups. Stronger signals for Bandeiraea simplicifolia (BS-I), Hippeastrum hybrid lectin (HHL), Pisum sativum agglutinin (PSA), Psophocarpus tetragonolobus lectin I (PTL-I), Psophocarpus tetragonolobus lectin II (PTL-II), Sophora japonica agglutinin (SJA), Lotus tetragonolobus lectin (LTL), and Triticum vulgaris (WGA) were observed in the KBD group. Meanwhile, Aleuria aurantia lectin (AAL), Lens culinaris agglutinin (LCA), Lycopersicon esculentum (tomato) lectin (LEL), Peanut agglutinin (PNA), and Sambucus nigra lectin (SNA) signals were lower in the KBD group. Selenium may have the function of influence the expression levels of carbohydrate chains Galα1,3-Gal, high mannose, and GlcNAc.



Similar content being viewed by others
Data Availability
The data of this study will be made available on reasonable request.
Code Availability
Not applicable.
References
Yu FF, Qi X, Shang YN, Ping ZG, Guo X (2019) Prevention and control strategies for children Kashin-Beck disease in China: A systematic review and meta-analysis. Medicine (Baltimore) 98(36):e16823. https://doi.org/10.1097/MD.0000000000016823
Zhang R, Guo H, Yang X, Zhang D, Li B, Li Z, Xiong Y (2019) Pathway-based network analyses and candidate genes associated with Kashin-Beck disease. Medicine (Baltimore) 98(18):e15498. https://doi.org/10.1097/MD.0000000000015498
Wang K, Yu J, Liu H, Liu Y, Liu N, Cao Y, Zhang X, Sun D (2020) Endemic Kashin-Beck disease: a food-sourced osteoarthropathy. Semin Arthritis Rheum 50(2):366–372. https://doi.org/10.1016/j.semarthrit.2019.07.014
Wang S, Duan C, Liu H, Shao W, Wu C, Han J, Guo X (2016) The roles of selenium, insulin-like growth factor binding protein 2 and suppressor of cytokine signaling 3 in the pathogenesis of Kashin-Beck disease. Biomarkers 21(5):409–415. https://doi.org/10.3109/1354750X.2016.1141990
Yao Y, Pei F, Kang P (2011) Selenium, iodine, and the relation with Kashin-Beck disease. Nutrition 27(11–12):1095–1100. https://doi.org/10.1016/j.nut.2011.03.002
Wang S, Guo X, Wang W, Wang S (2012) Genome-wide study identifies the regulatory gene networks and signaling pathways from chondrocyte and peripheral blood monocyte of Kashin-Beck disease. Genes Cells 17(8):619–632. https://doi.org/10.1111/j.1365-2443.2012.01620.x
Eichler J (2019) Protein glycosylation. Curr Biol 29(7):R229–R231. https://doi.org/10.1016/j.cub.2019.01.003
Manning JC, Romero A, Habermann FA, Garcia CG, Kaltner H, Gabius HJ (2017) Lectins: a primer for histochemists and cell biologists. Histochem Cell Biol 147(2):199–222. https://doi.org/10.1007/s00418-016-1524-6
Deng W, Wang Y, Liu Z, Cheng H, Xue Y (2014) HemI: a toolkit for illustrating heatmaps. PLoS ONE 9(11):e111988. https://doi.org/10.1371/journal.pone.0111988
Mehdi Y, Hornick JL, Istasse L, Dufrasne I (2013) Selenium in the environment, metabolism and involvement in body functions. Molecules 18(3):3292–3311. https://doi.org/10.3390/molecules18033292
Yiming Z, Qingqing L, Hang Y, Yahong M, Shu L (2020) Selenium deficiency causes immune damage by activating the DUSP1/NF-kappaB pathway and endoplasmic reticulum stress in chicken spleen. Food Funct 11(7):6467–6475. https://doi.org/10.1039/d0fo00394h
Li Y, Shah P, De Marzo AM, Van Eyk JE, Li Q, Chan DW, Zhang H (2015) Identification of glycoproteins containing specific glycans using a lectin-chemical method. Anal Chem 87(9):4683–4687. https://doi.org/10.1021/ac504304v
Diswall M, Benktander J, Angstrom J, Teneberg S, Breimer ME (2014) The alpha1,3GalT knockout/alpha1,2FucT transgenic pig does not appear to have an advantage over the alpha1,3GalT knockout pig with respect to glycolipid reactivity with human serum antibodies. Xenotransplantation 21(1):57–71. https://doi.org/10.1111/xen.12071
Urita A, Matsuhashi T, Onodera T, Nakagawa H, Hato M, Amano M, Seito N, Minami A, Nishimura S, Iwasaki N (2011) Alterations of high-mannose type N-glycosylation in human and mouse osteoarthritis cartilage. Arthritis Rheum 63(11):3428–3438. https://doi.org/10.1002/art.30584
Toegel S, Plattner VE, Wu SQ, Goldring MB, Chiari C, Kolb A, Unger FM, Nehrer S, Gabor F, Viernstein H, Wirth M (2009) Lectin binding patterns reflect the phenotypic status of in vitro chondrocyte models. Vitro Cell Dev Biol Anim 45(7):351–360. https://doi.org/10.1007/s11626-009-9186-5
Ishihara T, Kakiya K, Takahashi K, Miwa H, Rokushima M, Yoshinaga T, Tanaka Y, Ito T, Togame H, Takemoto H, Amano M, Iwasaki N, Minami A, Nishimura S (2014) Discovery of novel differentiation markers in the early stage of chondrogenesis by glycoform-focused reverse proteomics and genomics. Biochim Biophys Acta 1840(1):645–655. https://doi.org/10.1016/j.bbagen.2013.10.027
Igarashi M, Sakamoto K, Nagaoka I (2011) Effect of glucosamine, a therapeutic agent for osteoarthritis, on osteoblastic cell differentiation. Int J Mol Med 28(3):373–379. https://doi.org/10.3892/ijmm.2011.686
Funding
This study was supported by Shaanxi Natural Science Basic Research Projects (2021JM-037 and 2019JM-486), the National Natural Science Foundation of China key international cooperation project (81620108026).
Author information
Authors and Affiliations
Contributions
Sen Wang did the experiments and wrote the manuscript; Lingling Geng did the experiments and revised the manuscript; Guanghui Zhao analyzed the experiment data; Peiling Meng and Linlin Yuan collected the cartilage. Xiong Guo designed the project.
Corresponding author
Ethics declarations
Ethical Approval
Informed consent was obtained from each subject involved in the investigation. This research was approved by the Human Ethics Committee of Xi’an Jiaotong University, Xi’an, Shaanxi, China, and conducted in accordance with the 1964 Helsinki Declaration.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, S., Geng, L., Zhao, G. et al. Effectiveness of Selenium on Chondrocyte Glycoprotein Glycosylation Which Play Important Roles in the Pathogenesis of an Endemic Osteoarthritis, Kashin–Beck Disease. Biol Trace Elem Res 200, 1531–1537 (2022). https://doi.org/10.1007/s12011-021-02778-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-02778-z