Abstract
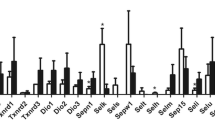
The aim of this study was to investigate the influence of Se deficiency on the transcription of inflammatory factors and selenoprotein genes in the kidneys of broiler chicks. One hundred fifty 1-day-old broiler chicks were randomly assigned to two groups fed with either a low-Se diet (L group, 0.033 mg/kg Se) or an adequate Se diet (C group, 0.2 mg/kg Se). The levels of uric acid (UA) and creatinine (Cr) in the serum and the mRNA levels of 6 inflammatory factors and 25 selenoprotein genes in the kidneys were measured as the clinical signs of Se deficiency occurred at 20 days old. The results indicated that the contents of UA and Cr in the serum increased in L group (p < 0.05), and the mRNA levels of the inflammatory factors (NF-κB, iNOS, COX-2, and TNF-α) increased in L group (p < 0.05). Meanwhile, the mRNA levels of PTGEs and HO-1 were not changed. In addition, 25 selenoprotein transcripts displayed ubiquitous expression in the kidneys of the chicks. The mRNA levels of 14 selenoprotein genes (Dio1, Dio2, GPx3, Sepp1, SelH, SelI, SelK, Sepn1, SelO, SelW, Sep15, SelT, SelU, and SelS) decreased, and 9 selenoprotein genes (GPx1, GPx2, GPx4, SelPb, Txnrd1, Txnrd2, Txnrd3, SPS2, and SelM) increased in L group (p < 0.05), but the Dio3 and Sepx1 mRNA levels did not change. The results indicated that Se deficiency resulted in kidney dysfunction, activation of the NF-κB pathway, and a change in selenoprotein gene expression. The changes of inflammatory factor and selenoprotein gene expression levels were directly related to the abnormal renal functions induced by Se deficiency.



Similar content being viewed by others
References
Mariotti M, Ridge PG, Zhang Y, et al (2012) Composition and evolution of the vertebrate and mammalian selenoproteomes. PLoS One 7:e33066
Rederstorff M, Krol A, Lescure A (2006) Understanding the importance of selenium and selenoproteins in muscle function. Cell Mol Life Sci 63:52–59
Huang JQ, Li DL, Zhao H, et al (2011) The selenium deficiency disease exudative diathesis in chicks is associated with downregulation of seven common selenoprotein genes in liver and muscle. J Nutr 141:1605–1610
Gao X, Xing H, Li S, et al (2012) Selenium regulates gene expression of selenoprotein W in chicken gastrointestinal tract. Biol Trace Elem Res 145:181–188
Sun D, Li C, Gao J, et al (2015) Effects of selenium deficiency on principal indexes of chicken kidney function. Biol Trace Elem Res 164:58–63
Maehira F, Luyo GA, Miyagi I, et al (2002) Alterations of serum selenium concentrations in the acute phase of pathological conditions. Clin Chim Acta 316:137–146
Galley HF, Davies MJ, Webster NR (1996) Xantine oxidase activity and free radical generation in patients with sepsis syndrome. Crit Care Med 24:1649–1653
Stoedter M, Renko K, Hog A, et al (2010) Selenium controls the sex-specific immune response and selenoprotein expression during the acute-phase response in mice. Biochem J 429:43–51
Tirosh O, Levy E, Reifen R (2007) High selenium diet protects against TNBS-induced acute inflammation, mitochondrial dysfunction, and secondary necrosis in rat colon. Nutrition 23:878–886
Sunde RA, Raines AM, Barnes KM, et al (2009) Selenium status highly regulates selenoprotein mRNA levels for only a subset of the selenoproteins in the selenoproteome. Biosci Rep 29:329–338
Liu Y, Zhao H, Zhang Q, et al (2012) Prolonged dietary selenium deficiency or excess does not globally affect selenoprotein gene expression and/or protein production in various tissues of pigs. J Nutr 142:1410–1416
Zhou JC, Zhao H, Li JG, et al (2009) Selenorotein gene expression in thyroid and pituitary of young pigs is not affected by dietary selenium deficiency or excess. J Nutr 139:1061–1066
Zhang JL, Zhang ZW, Shan AS, et al (2014) Effects of dietary selenium deficiency or excess on gene expression of selenoprotein N in chicken muscle tissues 157:234–241
Zhang JL, Li JL, Huang XD, et al (2012) Dietary selenium regulation of transcript abundance of selenoprotein N and selenoprotein Win chicken muscle tissues. Biometals 25:297–307
Liu CP, Fu J, Lin SL, et al (2014) Effects of dietary selenium deficiency on mRNA levels of twenty-one selenoprotein genes in the liver of layer chicken. Biol Trace Elem Res 159:192–198
Lin SL, Wang CW, Tan SR, et al (2014) Selenium deficiency inhibits the conversion of thyroidal thyroxine (T4) to triiodothyronine (T3) in chicken thyroids. Biol Trace Elem Res 161:263–271
Liang Y, Lin SL, Wang CW, et al (2014) Effect of selenium on selenoprotein expression in the adipose tissue of chickens. Biol Trace Elem Res 160:41–48
Yao HD, Zhao WC, Zhao X, et al (2014) Selenium deficiency mainly influences the gene expressions of antioxidative selenoproteins in chicken muscles. Biol Trace Elem Res 161:318–327
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−delta delta C(T)) method. Methods 25:402–408
Najafi M (2014) Serum creatinine role in predicting outcome after cardiac surgery beyond acute kidney injury. World J Cardiol 6:1006–1021
Wahlqvist ML (2013) Antioxidant relevance to human health. Asia Pac J Clin Nutr 22:171–176
Xia Y, Peng C, Zhou Z, et al (2012) Clinical significance of saliva urea, creatinine, and uric acid levels in patients with chronic kidney disease. Zhong Nan Da Xue Xue Bao Yi Xue Ban 37:1171–1176
Makarov SS (2000) NF-κB as a therapeutic target in chronic inflammation: recent advances. Mol Med Today 6:441–448
Pando MP, Verma IM (2000) Signal-dependent and -independent degradation of free and NF-kappa B-bound IkappaBalpha. J. Biol. Chem. 275:21278–21286
Abate A, Oberle S, Schröder H (1998) Lipopolysaccharide-induced expression of cyclooxygenase-2 in mouse macrophages is inhibited by chloromethylketones and a direct inhibitor of NF-kappa B translocation. Prostaglandins Other Lipid Mediat 56:277–290
Israf DA, Khaizurin TA, Syahida A, et al (2007) Cardamonin inhibits COX and iNOS expression via inhibition of p65NF-kappaB nuclear translocation and Ikappa-B phosphorylation in RAW 264.7 macrophage cells. Mol Immunol 44:673–679
Ackerman WE, Summerfield TL, Vandre DD, et al (2008) Nuclear factor-kappa B regulates inducible prostaglandin E synthase expression in human amnion mesenchymalcells. Biol Reprod 78:68–76
Oh YC, Jeong YH, Ha JH, et al (2014) Oryeongsan inhibits LPS-induced production of inflammatory mediators via blockade of the NF-kappa B, MAPK pathways and leads to HO-1 induction in macrophage cells. BMC Complement Altern Med 14:242
Jeong DW, Yoo MH, Kim TS, et al (2002) Protection of mice from allergen-induced asthma by selenite: prevention of eosinophil infiltration by inhibition of NF-kappa B activation. J Biol Chem 277:17871–17876
Hwang JT, Kim YM, Surh YJ, et al (2006) Selenium regulates cyclooxygenase-2 and extracellular signalregulated kinase signaling pathways by activating AMP-activated protein kinase in colon cancer cells. Cancer Res 66:10057–10063
Youn HS, Lim HJ, Choi YJ, et al (2008) Selenium suppresses the activation of transcription factor NF-kappa B and IRF3 induced by TLR3 or TLR4 agonists. Int Immunopharmacol 8(3):495–501
Vincent JL, Forceville X (2008) Critically elucidating the role of selenium. Curr Opin Anesthesiol 21:148–154
Maehira F, Miyagi I, Eguchi Y (2003) Selenium regulates transcription factor NF-kB activation during the acute phase reaction. Clin Chem Acta 334:163–171
Grundemann C, Garcia-Kaufer M, Sauer B, et al (2013) Traditionally used Veronica officinalis inhibits proinflammatory mediators via the NF-κB signalling pathway in a human lung cell line. J Ethnopharmacol 145:118–126
Surh YJ, Chun KS, Cha HH, et al (2001) Molecular mechanisms underlying chemopreventive activities of anti-inflammatory phytochemicals: down-regulation of COX-2 and iNOS through suppression of NF-κB activation. Mutat Res 480-481:243–268
Raabe T, Bukrinsky M, Currie RA (1998) Relative contribution of transcription and translation to the induction of tumor necrosis factor-alpha by lipopolysaccharide. J Biol Chem 273:974–980
Hochberg MC, Tracy JK, Hawkins-Holt M, et al (2003) Comparison of the efficacy of the tumour necrosis factor alpha blocking agents adalimumab, etanercept, and infliximab when added to methotrexate in patients with active rheumatoid arthritis. Ann Rheum Dis 62:213–216
Pae HO, Chung HT (2009) Heme oxygenase-1: its therapeutic roles in inflammatory diseases. Immune Netw 9:12–19
Terry CM, Clikeman JA, Hoidal JR, et al (1998) Effect of tumor necrosis factor-alpha and interleukin-1 alpha on heme oxygenase-1 expression in human endothelial cells. Am J Physiol 274:883–891
Srisook K, Cha YN (2005) Super-induction of HO-1 in macrophages stimulated with lipopolysaccharide by prior depletion of glutathione decreases iNOS expression and NO production. Nitric Oxide 12:70–79
Ashino T, Yamanaka R, Yamamoto M, et al (2008) Negative feedback regulation of lipopolysaccharide-induced inducible nitric oxide synthase gene expression by heme oxygenase-1 induction in macrophages. Mol Immunol 45:2106–2115
Liu T, Laidlaw TM, Feng C, et al (2012) Prostaglandin E2 deficiency uncovers a dominant role for thromboxane A2 in house dust mite-induced allergic pulmonary inflammation. Proc Natl Acad Sci U S A 109:12692–12697
Barnes KM, Evenson JK, Raines AM, et al (2009) Transcript analysis of the selenoproteome indicates that dietary selenium requirements of rats based on elenium-regulated selenoprotein mRNA levels are uniformly less than those based on glutathione peroxidaseactivity. J Nutr 139:199–206
Crosley LK, Méplan C, Nicol F, et al (2007) Differential regulation of expression of cytosolic and mitochondrial thioredoxin reductase in rat liver and kidney. Arch Biochem Biophys 459:178–188
Bermano G, Nicol F, Dyer JA, et al (1995) Tissue-specific regulation of selenoenzyme gene expression during selenium deficiency in rats. Biochem J 311:425–430
Seyedali A, Berry MJ (2014) Nonsense-mediated decay factors are involved in the regulation of selenoprotein mRNA levels during selenium deficiency. RNA 20:1248–1256
Hoffmann PR, Berry MJ (2008) The influence of selenium on immune responses. Mol Nutr Food Res 52:1273–1280
Carlson BA, Yoo MH, Shrimali RK, et al (2010) Role of selenium-containing proteins in T-cell and macrophage function. Proc Nutr Soc 69:300–310
Curran JE, Jowett JB, Elliott KS, et al (2005) Genetic variation in selenoprotein S influences inflammatory response. Nat Genet 37:1234–1241
Duntas LH (2009) Selenium and inflammation: underlying anti-inflammatory mechanisms. Horm Metab Res 41:443–447
Acknowledgments
This study was supported by the Research Fund for the Doctoral Program of Higher Education (20122325110018), the National Natural Science Foundations of China (31472104; 31272626; 30871902), and the National Science and Technology Support Program (2013BAD20B04). The authors thank the members in the Veterinary Internal Medicine Laboratory at the College of Veterinary Medicine, Northeast Agricultural University for their help in analyzing the data.
Conflict of Interest
All authors declare that they have no competing interests.
Compliance with Ethical Standards
All of the procedures used in this experiment were approved by the Institutional Animal Care and Use Committee of Northeast Agricultural University.
Author information
Authors and Affiliations
Corresponding author
Additional information
All authors have read the manuscript and agreed to submit it in its current form for consideration for publication in the journal.
Rights and permissions
About this article
Cite this article
Zhang, Jl., Xu, B., Huang, Xd. et al. Selenium Deficiency Affects the mRNA Expression of Inflammatory Factors and Selenoprotein Genes in the Kidneys of Broiler Chicks. Biol Trace Elem Res 171, 201–207 (2016). https://doi.org/10.1007/s12011-015-0512-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-015-0512-3