Abstract
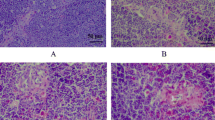
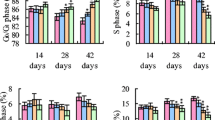
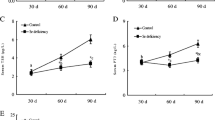
The purpose of this 42-day study was to investigate the effects of low selenium (Se) on immune function by determining histopathological changes of thymus, apoptosis of thymic cells, and subpopulation of peripheral blood T cells. One hundred twenty 1-day-old avian broilers were randomly assigned to two groups of 60 each and were fed on a low Se diet (0.0342 mg/kg Se) or a control diet (0.2 mg/kg Se), respectively. The relative weight of thymus was significantly decreased in low Se group from 21 days of age in time-dependent manner when compared with that of control group. Histopathologically, lymphopenia in the cortex and medulla of thymus was observed in low Se group. In comparison with those of control group, the percentage of Annexin-V positive cells was increased, and the percentages of CD3+ and CD3+CD8+ T cells of the peripheral blood were decreased in low Se group, as measured by flow cytometry. These data suggested that low dietary Se induced histological lesions of thymus, increased apoptosis of thymic cells, and decreased T cell subsets. The cellular immune function was finally impaired in broilers.



Similar content being viewed by others
References
McKenzie RC, Becket GJ, Arthur JR (2006) Effect of selenium on immunity and aging. In: Hatfield DL, Berry MJ, Gladyshev VN (eds) Selenium: its molecular biology and role in human health. Springer, New York, pp 311–322
Thompson JN, Scott ML (1970) Impaired lipid and vitamin E absorption related to atrophy of the pancreas in selenium-deficient chicks. J Nutr 100:797–809
Marsh JA, Combs GF Jr, Whitacre ME et al (1986) Effect of selenium and vitamin E dietary deficiencies on chick lymphoid organ development. Proc Soc Exp Biol Med 182:425–436
Cui HM (1988) The histopathologic study of selenium deficiency in chicks. Acta Veterinaria et Zootechnica Sinica 1:52–56
Peng X, Cui Y, Cui W et al (2010) The cell cycle arrest and apoptosis of bursa of Fabricius induced by low selenium in chickens. Biol Trace Elem Res. doi:10.1007/s12011-010-8639-8
Kiremidjian-Schumacher L, Roy M, Wishe HI et al (1994) Supplementation with selenium and human immune cell functions, II: effect on cytotoxic lymphocytes and natural killer cells. Biol Trace Elem Res 41:115–127
Vega L, Rodríguez-Sosa M, García-Montalvo EA et al (2007) Non-optimal levels of dietary selenomethionine alter splenocyte response and modify oxidative stress markers in female mice. Food Chem Toxicol 45:1147–1153
Spallholz JE, Stewart JE (1989) Advances in the role of minerals in immunobiology. Biol Trace Elem Res 19:129–151
Marsh JR, Dietert RR, Combs GF Jr (1981) Influence of dietary selenium and vitamin E on the humoral immune response of the chick. Proc Soc Exp Biol Med 166:228–236
Larsen HJ, Tollersrud S (1981) Effect of dietary vitamin E and selenium on the phytohaemagglutination response of pig lymphocytes. Res Vet Sci 31:301–305
Serfass RE, Ganther HE (1975) Defective microbicidal activity in glutathione peroxidase-deficient neutrophils of selenium-deficient rats. Nature 255:640–641
Arthur JR, McKenzie RC, Beckett GJ (2003) Selenium in the immune system. J Nutr 133:1457S–1459S
Peng X, Cui Y, Cui W et al (2009) The decrease of relative weight, lesions, and apoptosis of bursa of Fabricius induced by excess dietary selenium in chickens. Biol Trace Elem Res 131:33–42
Chen T, Cui Y, Bai C et al (2009) Decreased percentages of the peripheral blood T-cell subsets and the serum IL-2 contents in chickens fed on diets excess in fluorine. Biol Trace Elem Res 132:122–128
Li P (1998) A review of development, function and derivative structure of vertebrate's thymic epithelial cell. Yantai Teachers University Journal 14:226–231
Molinero P, Osuna C, Guerrero JM (1995) Type II thyroxine 5'-deiodinase in the rat thymus. J Endocrinol 146:105–111
Zeng H (2002) Selenite and selenomethionine promote HL-60 cell cycle progression. J Nutr 132:74–679
Cheng WH, Quimby FW, Lei XG (2003) Impacts of glutathione peroxidase-1 knockout on the protection by injected selenium against the pro-oxidant-induced liver aponecrosis and signaling in selenium-deficient mice. Free Radic Biol Med 34:918–927
Green DR (2000) Apoptotic pathways: paper wraps stone blunts scissors. Cell 102:1–4
Rao L, Puschner B, Prolla TA (2001) Gene exprssion profiling of low selenium status in the mouse intestine: transcriptional activation of genes linked to DNA damage, cell cycle control and oxidative stress. J Nutr 131:3175–3181
Shrimali RK, Irons RD, Carlson BA et al (2008) Selenoproteins mediate T cell immunity through an antioxidant mechanism. J Biol Chem 283:20181–20185
Pavelka M, Roth J (2005) Functional ultrastructure—atlas of tissue biology and pathology. Springer, New York, pp 312–313
Gisela FE, Walter GB, Tina KB (1998) CD4, CD8 and TCR defined T-cell subsets in thymus and spleen of 2- and 7-week old commercial broiler chickens. Vet Immunol Immunopathol 62:339–348
Petrie HT, Klassen LW, Klassen PS et al (1989) Selenium and the immune response: 2. Enhancement of murine cytotoxic T-lymphocyte and natural killer cell cytotoxicity in vivo. J Leukoc Biol 45:215
Acknowledgement
This work was supported by Program for Changjiang Scholars and Innovative Research Team in University (IRT 0848) and the Education Department of Sichuan Province (09ZZ017).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peng, X., Cui, Hm., Deng, J. et al. Low Dietary Selenium Induce Increased Apoptotic Thymic Cells and Alter Peripheral Blood T Cell Subsets in Chicken. Biol Trace Elem Res 142, 167–173 (2011). https://doi.org/10.1007/s12011-010-8756-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-010-8756-4