Abstract
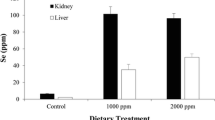
We report here some results of a long-term (19 month) study with cats fed methylmercury (MeHg) in nutritionally balanced diets based on fish. By using either freshwater pike (low in Se) or canned tuna (high in Se) as the major protein source, basal diets with low levels of MeHg were prepared having different Se content, all Se being of natural origin. The basal diets produced no signs of toxicity or pathological changes over the l9-month period. In cats fed basal diets spiked with medium or high levels of MeHg, evidence for delayed onset of toxic effects from the added MeHg was observed with the tuna diets compared to pike diets. In brain, muscle, and blood, the activity of GSH peroxidase, a selenoenzyme, was decreased by Hg. In liver, substantial accumulation of Hg with Se occured (molar Hg/Se ratio approximately 1.4 to 1.8) but GSH peroxidase activity was unaffected. We suggest that the coaccumulation of Hg and Se in liver measures the extent to which MeHg has been metabolically transformed by metabolism to Hg++, and inactivated by deposition as a Hg/Se complex of low bioavailability. The accumulation of Hg and Se in liver was much greater in cats fed tuna compared to pike, out of proportion to the relatively small differences in Hg and Se content of the tuna and pike basal diets. Some mechanisms are described by which selenium, vitamin E, and other factors might facilitate MeHg breakdown to inorganic Hg during long term low level exposure to MeHg.

Similar content being viewed by others
References
Ganther HE, Goudie C, Sunde ML, Kopecky MJ, Wagner P, Oh S-H, Hoekstra WG (l972) Selenium: Relation to decreased toxicity of methylmercury added to diets containing tuna. Science 175:1122–1124
Ganther HE, Sunde ML (l974) Effect of tuna fish and selenium on the toxicity of methylmercury: A progress report. J Food Sci 39:1–5
Chang LW, Yamaguchi S, Dudley, Jr, AW (l974) Neurological changes in cats following long-term diet of mercury contaminated tuna. Acta Neuropath (Berl) 27:171–176
Stillings BR, Lagally H, Bauersfeld P, Soares J (l974) Effect of cystine, selenium, and fish protein on the toxicity and metabolism of methylmercury in rats. Toxicol Appl Pharmacol 30:243–254
El-Begearmi MM, Ganther HE, Sunde ML (l982) Dietary interactions between methylmercury, selenium, arsenic, and sulfur amino acids in Japanese quail. Poultry Sci 61:272–279
Welsh S, Soares, Jr, JH (l976) The protective effect of vitamin E and selenium against methyl mercury toxicity in Japanese quail. Nutr Reports Internat 13:43–51
El-Begearmi MM (1978) Factors modifying mercury toxicity in Japanese quail and rats. Dissertation, University of Wisconsin
Charbonneau SM, Munro IC, Nera EA, Armstrong FAJ, Willes RF, Bryce F, Nelson RF (1976) Chronic toxicity of methylmercury in the adult cat. Interim report. Toxicology 5:337–349
Munns RK, Holland DC (l971) Determination of mercury in fish by flameless atomic absorption: a collaborative study. J Assoc Offic Anal Chem 54:202–205
Oh S-H, Ganther HE, Hoekstra WG (l974) Selenium as a component of glutathione peroxidase isolated from ovine erythrocytes. Biochemistry 13:1825–1829
Prohaska JR, Ganther HE (l976) Selenium and glutathione peroxidase in developing rat brain. J Neurochemistry 27:1379–1387
Chang LW, Dudley, Jr, AW, Dudley MA, Ganther HE, Sunde ML (1977) Modification of the neurotoxic effects of methyl mercury by selenium. In: Roizin L, Shiraki H, Grcevic N (eds) Neurotoxicology. Raven, New York, pp. 275–282
Tsuchiya K (1992) The discovery of the causal agent of Minamata disease. Am J Ind Med 21:275–280
Ohi G, Nishigaki S, Seki H, Tamura Y, Maki T, Minowa K, Shimamura Y, Mizoguchi I, Inaba Y, Takizawa Y, Kawanishi Y (l980) The protective potency of marine animal meat against the neurotoxicity of methylmercury: Its relationship with the organ distribution of mercury and selenium in the rat. Food Cosmetics Toxicol 18:139–145
El-Begearmi MM, Sunde ML, Ganther HE (l977) A mutual protective effect of mercury and selenium in Japanese quail. Poultry Sci 56:313–322
Stoewsand GS, Bache CA, Lisk DJ (l974) Dietary selenium protection of methylmercury intoxication of Japanese quail. Bull Environ Contam Toxicol 11:152–156
Charleston JS, Body RL, Bolender RP, Mottet NK, Vahter ME, Burbacher TM (l996) Changes in the number of astrocytes and microglia in the thalamus of the monkey Macaca fascicularis following long-term subclinical methylmercury exposure. Neurotoxicology 17:127–138
Ralston N (2007) Significance of mercury’s molecular interactions with selenium. Biol Trace Element Res (in press)
Diplock AT (l976) Metabolic aspects of selenium action and toxicity. CRC Crit Rev Toxicol 4:271–329
Wagner PA, Hoekstra WG, Ganther HE (l975) Alleviation of silver toxicity by selenite in the rat in relation to tissue glutathione peroxidase. Proc Soc Exp Biol Med 148:1106–1110
Lind B, Friberg L, Nylander M (l988) Preliminary studies on methylmercury biotransformation and clearance in the brain of primates: II. Demethylation of mercury in brain. J Trace Elem Exp Med 1:49–56
Falnoga I (2007) Selenium-mercury interactions in humans and animals. Biol Trace Element Res (in press)
Nishigaki S (l975) Methylmercury and selenium in umbilical cords of inhabitants of the Minamata area. Nature 258:324–325
Fang SC (l974) Induction of C-Hg cleavage enzymes in rat liver by dietary selenite. Res Comm Chem Pathol Pharmacol 9:579–582
Iwata H, Masukawa T, Kito H, Hayashi M (l982) Degradation of methylmercury by selenium. Life Sci 31:859–866
Hsieh SH, Ganther HE (l975) Acid-volatile selenium formation catalyzed by glutathione reductase. Biochemistry 14:1632–1636
Suzuki KT (2007) Interactions of selenium with mercury during its metabolism Biol. Trace Element Res (in press)
Caygill CPJ, Lucy JA, Diplock AT (l971) The effect of vitamin E on the intracellular distribution of the different oxidation states of selenium in rat liver. Biochem J 125:407–416
Ganther HE (l980) Interactions of vitamin E and selenium with mercury and silver. Ann N Y Acad Sci 355:212–226
Edmonds JS, Francesconi KA (l981) Arseno-sugars from brown kelp as intermediates in cycling of arsenic in a marine ecosystem. Nature 289:602–604
Ganther HE, Levander OA, Baumann CA (l966) Dietary control of selenium volatilization in the rat. J Nutrition 88:55–60
Hsieh HS, Ganther HE (l976) Effects of stock or purified diet on rat liver enzymes involved in the synthesis of dimethyl selenide. J Nutrition 106:1577–1582
Kaderlik RK, Weser E, Ziegler DM (l991) Selective loss of liver flavin-containing monooxygenase in rats on chemically defined diets. In: Hlavica P, Damani LA (eds) Progress in Pharmacology and Clininical Pharmacology vol 8(3). Gustav Fischer, New York, pp 95–103
Acknowledgement
This research was supported by the Food Research Institute, University of Wisconsin-Madison, and by the National Marine Fisheries Service, NOAA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ganther, H.E., Sunde, M.L. Factors in Fish Modifying Methylmercury Toxicity and Metabolism. Biol Trace Elem Res 119, 221–233 (2007). https://doi.org/10.1007/s12011-007-8006-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-007-8006-6