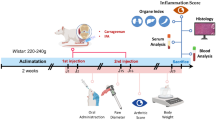
Abstract
Oxidative stress has been implicated in deadly lifestyle diseases, and antioxidants from plant sources are the primary option in the treatment regime. Kenaf seeds are the storehouse of potential natural antioxidant phytoconstituents. Perhaps, none of the studies documented the phytoconstituents and their antioxidant potential from Kenaf seed coat so far. Thus, the current study focuses on exploring the protective effect of Kenaf Seed Coat Ethanol Extract (KSCEE) against sodium nitrite and diclofenac-induced oxidative stress in vitro (red blood cell and platelets model) and in vivo (female Sprague Dawely rat’s model) along with the antithrombotic activity. The infrared spectrophotometry data showed the heterogeneous functional groups (CH, OH, C = C, C = C–C) and aromatic rings. Reverse phase high-performance liquid chromatography and gas chromatography–mass spectrometry chromatogram of KSCEE also evidenced the presence of several phytochemicals. KSCEE displayed about 76% of DPPH scavenging activity with an IC50 value of 34.94 µg/ml. KSCEE significantly (***p < 0.001) normalized the stress markers such as lipid peroxidation, protein carbonyl content, superoxide dismutase, and catalase in sodium nitrite and diclofenac-induced oxidative stress in RBC, platelets, liver, kidney, and small intestine, respectively. Furthermore, KSCEE was found to protect the diclofenac-induced tissue destruction of the liver, kidney, and small intestine obtained from seven groups of female Sprague Dawely rats. KSCEE delayed the clotting time of platelet-rich plasma and platelet-poor plasma and activated partial thromboplastin time, suggesting its anticoagulant property. In addition, KSCEE also exhibited antiplatelet activity by inhibiting both adenosine diphosphate and epinephrine-induced platelet aggregation. In conclusion, KSCEE ameliorates the sodium nitrite and diclofenac-induced oxidative stress in red blood cells, platelets, and experimental animals along with antithrombotic properties.









Similar content being viewed by others
Data Availability
The data that support the finding of this study are available from the corresponding author upon reasonable request.
Abbreviations
- ADP:
-
Adenosine diphosphate
- ALP:
-
Alkaline phosphatase
- APTT:
-
Activated partial thromboplastin time
- CAT:
-
Catalase
- DFC:
-
Diclofenac
- DNPH:
-
Di-Nitro-Phenyl-Hydrazine
- DPPH:
-
Di-Phenyl-2-Picryl-Hdrazyl
- ECNICI:
-
Electron capture negative ion chemical ionization
- EDTA:
-
Ethylene diamine tetraacetic acid
- FTIR:
-
Fourier transform infrared spectrophotometry
- GC–MS:
-
Gas chromatography-mass spectrometry
- HPLC:
-
High-performance liquid chromatography
- KSCEE:
-
Kenaf seed coat ethanol extract
- LDH:
-
Lactate dehydrogenase
- LPO:
-
Lipid per oxidation
- MED:
-
Minimum edema dose
- MHD:
-
Minimum hemorrhagic dose
- PCC:
-
Protein carbonyl content
- PDA:
-
Photometric diode array
- PPP:
-
Platelet-poor plasma
- PRP:
-
Platelet rich plasma
- PT:
-
Prothrombin time
- RBC:
-
Red blood cells
- RNS:
-
Reactive nitrogen species
- ROS:
-
Reactive oxygen species
- SDS:
-
Sodium dodecyl sulfate
- SGOT:
-
Serum glutamic oxaloacetic transamylase
- SGPT:
-
Serum glutamate pyruvate transferase
- SOD:
-
Super oxide dismutase
- SYL:
-
Silymarin
References
Carvalho, C., & Moreira, P. I. (2018). Oxidative stress: A major player in cerebrovascular alterations associated to neurodegenerative events. Frontiers in Physiology, 9, 806. https://doi.org/10.3389/fphys.2018.00806
Lozano, R., Naghavi, M., Foreman, K., Lim, S., Shibuya, K., Aboyans, V., & Murray, C. J. (2012). Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. The Lancet, 380(9859), 2095–2128. https://doi.org/10.1016/S0140-6736(12)61728-0
Evans, N. S., & Ratchford, E. V. (2014). Vascular disease patient information page: Venous thromboembolism (deep vein thrombosis and pulmonary embolism). Vascular Medicine, 19(2), 148–150. https://doi.org/10.1177/1358863X14529007
Alfhili, M. A., Weidner, D. A., & Lee, M.-H. (2019). Disruption of erythrocyte membrane asymmetry by triclosan is preceded by calcium dysregulation and p38 MAPK and RIP1 stimulation. Chemosphere, 229, 103–111. https://doi.org/10.1016/j.chemosphere.2019.04.21
Cosby, K., Partovi, K. S., Crawford, J. H., Patel, R. P., Reiter, C. D., Martyr, S., Yang, B. K., Waclawiw, M. A., Zalos, G., Xu, X., & Huang, K. T. (2003). Nitrite reduction to nitric oxide by deoxyhemoglobin vasodilates the human circulation. Nature Medicine, 9(12), 1498–1505. https://doi.org/10.1038/nm954
Buerck, J. P., Burke, D. K., Schmidtke, D. W., Snyder, T. A., Papavassiliou, D. V., & O’Rear, E. A. (2021). Production of erythrocyte microparticles in a sub-hemolytic environment. Journal of Artificial Organs, 24(2), 135–145. https://doi.org/10.1007/s10047-020-01231-7
Qiao, J., Arthur, J. F., Gardiner, E. E., Andrews, R. K., Zeng, L., & Xu, K. (2018). Regulation of platelet activation and thrombus formation by reactive oxygen species. Redox Biology, 14, 126–130. https://doi.org/10.1016/j.redox.2017.08.021
Fuentes, E., Gibbins, J. M., Holbrook, L. M., & Palomo, I. (2018). NADPH oxidase 2 (NOX2): A key target of oxidative stress-mediated platelet activation and thrombosis. Trends in Cardiovascular Medicine, 28(7), 429–434. https://doi.org/10.1016/j.tcm.2018.03.001
Wang, L., Chen, J., Xie, H., Ju, X., & Liu, R. H. (2013). Phytochemical profiles and antioxidant activity of Adlay varieties. Journal of Agricultural and Food Chemistry, 61(21), 5103–5113. https://doi.org/10.1021/jf400556s
Seca, A. M., Silva, A. M., Silvestre, A. J., Cavaleiro, J. A., Domingues, F. M., & Pascoal-Neto, C. (2000). Chemical composition of the light petroleum extract of Hibiscus cannabinus bark and core. Phytochemical Analysis, 11, 345. https://doi.org/10.1016/s0031-9422(01)00311-9
Kareru, P., Keriko, J., Gachanja, A., & Kenji, G. (2008). Direct detection of triterpenoid saponins in medicinal plants. African Journal of Traditional, Complementary and Alternative Medicines, 5(1), 56–60. https://doi.org/10.4314/ajtcam.v5i1.31257
Okoh, S. O., Asekun, O. T., Familoni, O. B., & Afolayan, A. J. (2011). Composition and antioxidant activities of leaf and root volatile oils of Morinda lucida. Natural Product Communications, 6(10), 1934578X1100601. https://doi.org/10.1177/1934578X1100601032
Ohkawa, H., Ohishi, N., & Yagi, K. (1979). Assay for lipid peroxides in animal tissues by the thiobarbituric acid reaction. Analytical Biochemistry, 95, 351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Levine, R. L., Garland, D., Oliver, C. N., Amici, A., Climent, I., et al. (1951). Determination of carbonyl content in oxidatively modified proteins. Methods in Enzymology, 186, 464–478. https://doi.org/10.1016/0076-6879(90)86141-H
Sundaram, M. S., Hemshekhar, M., Santhosh, M. S., Paul, M., Sunitha, K., Thushara, R. M., & Girish, K. S. (2015). Tamarind seed (Tamarindus indica) extract ameliorates adjuvant-induced arthritis via regulating the mediators of cartilage/bone degeneration, inflammation and oxidative stress. Scientific Reports, 5(1), 11117. https://doi.org/10.1038/srep11117
Gangaraju, S., Manjappa, B., Subbaiah, G. K., Kempaiah, K., Shashidharamurthy, R., Plow, J. H., & Sannaningaiah, D. (2015). Jackfruit (Artocarpus heterophyllus) seed extract exhibits fibrino (geno)lytic activity. Pharmacognosy Journal, 7(3), 171–177. https://doi.org/10.5530/pj.2015.3.5
Quick, A. J., Stanley-Brown, M., & Bancroft, F. W. (1935). A study of the coagulation defect in hemophilia and jaundice. The American Journal Of The Medical Sciences, 190(4), 501–510. https://doi.org/10.1097/00000441-193510000-00009
Born, G. V. R. (1962). Aggregation of blood platelets by adenosine diphosphate and its reversal. Nature, 194, 927–929. https://doi.org/10.1038/194927b0
Kondo, H., Kondo, S., Ikezawa, H., Murata, R., & Ohsaka, A. (1960). Studies on the quantitative method for determination of the hemorrhagic activity of habu snake venom. Japanese Journal of Medical Science and Biology, 13(1–2), 43–51. https://doi.org/10.7883/yoken1952.13.43
Vishwanath, S. B., Manjunatha Kini, R., & Veerabasappa Gowda, T. (1987). Characterization of three edema-inducing phospholipase A2 enzymes from habu (Trimeresurus flavoviridis) venom and their interaction with the alkaloid aristolochic acid. Toxicon, 25(5), 501–515. https://doi.org/10.1016/0041-0101(87)90286-8
Sies, H. (1997). Oxidative stress: Oxidants and antioxidants. Experimental Physiology, 82, 291–295. https://doi.org/10.1113/expphysiol.1997.sp004024
Hybertson, B. M., Gao, B., Bose, S. K., & McCord, J. M. (2011). Oxidative stress in health and disease: The therapeutic potential of Nrf2 activation. Molecular Aspects of Medicine, 32(4–6), 234–246. https://doi.org/10.1016/j.mam.2011.10.006
Subramanion, L. J., Zakaria, Z., & Sreenivasan, S. (2011). Phytochemicals screening, DPPH free radical scavenging and xanthine oxidase inhibitory activities of Cassia fistula seeds extract. Journal of Medicinal Plants Research, 5(10), 1941–1947.
Dravie, E. E., Kortei, N. K., Essuman, E. K., Tettey, C. O., Boakye, A. A., & Hunkpe, G. (2020). Antioxidant, phytochemical and physicochemical properties of a sesame seed (Sesamum indicum L). Scientific African, 8, e00349. https://doi.org/10.1016/j.sciaf.2020.e00349
Bianchini Silva, L. S., Perasoli, F. B., Carvalho, K. V., Vieira, K. M., Paz Lopes, M. T., Bianco de Souza, G. H., Henrique Dos Santos, O. D., & Freitas, K. M. (2020). Melaleuca leucadendron (L.) L. flower extract exhibits antioxidant and photoprotective activities in human keratinocytes exposed to ultraviolet B radiation. Free Radical Biology and Medicine, 159, 54–65. https://doi.org/10.1016/j.freeradbiomed.2020.07.022
Jimoh, M. A., Idris, O. A., & Jimoh, M. O. (2020). Cytotoxicity, Phytochemical, Antiparasitic Screening, and Antioxidant Activities of Mucuna pruriens (Fabaceae). Plants, 9(9), 1249. https://doi.org/10.3390/plants9091249
Hayes, J. E., Allen, P., Brunton, N., O’Grady, M. N., & Kerry, J. P. (2011). Phenolic composition and in vitro antioxidant capacity of four commercial phytochemical products: Olive leaf extract (Olea europaea L.), lutein, sesamol and ellagic acid. Food Chemistry, 126(3), 948–955. https://doi.org/10.1016/j.foodchem.2010.11.092.
Salahuddin, M. A. H., Ismail, A., Kassim, N. K., Hamid, M., & Ali, M. S. M. (2020). Phenolic profiling and evaluation of in vitro antioxidant, α-glucosidase and α-amylase inhibitory activities of Lepisanthes fruticosa (Roxb) Leenh fruit extracts. Food Chemistry, 331, 127240. https://doi.org/10.1016/j.foodchem.2020.127
Kawamoto, E. M., Munhoz, C. D., Glezer, I., Bahia, V. S., Caramelli, P., Nitrini, R., & Marcourakis, T. (2005). Oxidative state in platelets and erythrocytes in aging and Alzheimer’s disease. Neurobiology of Aging, 26(6), 857–864. https://doi.org/10.1016/j.neurobiolaging.2004.08.011
Chandramma, S., Kengaiah, J., Nandish, S. K. M., Ramachandraiah, C., Hanumegowda, S. M., Shivaiah, A., Santhosh, S., & Sannaningaiah, D. (2020). Caesalpinia crista coat extract protects red blood cells from sodium nitrite-induced oxidative stress and exhibits antiplatelet activity. Blood Coagulation & Fibrinolysis, 31, 293–302. https://doi.org/10.1097/MBC.0000000000000913
Girish, T. K., Vasudevaraju, P., & Prasada Rao, U. J. S. (2012). Protection of DNA and erythrocytes from free radical induced oxidative damage by black gram (Vigna mungo L.) husk extract. Food and Chemical Toxicology, 50(5), 1690–1696. https://doi.org/10.1016/j.fct.2012.01.043
Thushara, R. M., Hemshekhar, M., Paul, M., Shanmuga Sundaram, M., Shankar, R. L., Kemparaju, K., & Girish, K. S. (2014). Crocin prevents sesamol-induced oxidative stress and apoptosis in human platelets. Journal of Thrombosis and Thrombolysis, 38(3), 321–330. https://doi.org/10.1007/s11239-014-1056-7
Ashwini, S., Chandramma, S., Sujatha, M.H., Jayanna, K., Sharath Kumar, M.N., Ramachandraiah, C., Hanumegowda, S.M., Santhosh S, Sannaningaiah D. (2020). Protein extract of Pennisetum glaucum (Pearl millet) protects RBC, liver, kidney, and small intestine from oxidative stress and exhibits anticoagulant, and antiplatelet activity. J Am Coll Nutr, 1541–1087.
Liu, L., Gao, Q., Zhang, Z., & Zhang, X. (2022). Elsholtzia rugulosa: Phytochemical profile and antioxidant, anti-Alzheimer’s disease, antidiabetic, antibacterial, cytotoxic and hepatoprotective activities. Plant Foods for Human Nutrition, 77(1), 62–67. https://doi.org/10.1007/s11130-021-00941-4
Sinha, M., Manna, P., & Sil, P. C. (2007). Aqueous extract of the bark of Terminalia arjuna plays a protective role against sodium-fluoride-induced hepatic and renal oxidative stress. Journal of Natural Medicines, 61(3), 251–260. https://doi.org/10.1007/s11418-007-0133-z
Korish, A. A. (2010). Effect of caffeic acid phenethyl ester on the hemostatic alterations associated with toxin-induced acute liver failure. Blood Coagulation & Fibrinolysis, 21(2), 158–163. https://doi.org/10.1097/MBC.0b013e32833678be
Jerine, S. P., Parthasarathy, M., Nithyanandham, S., Katturaja, R., Namachivayam, A., & Prince, S. E. (2019). Protective effect of the ethanolic and methanolic leaf extracts of Madhuca longifolia against diclofenac-induced toxicity in female Wistar albino rats. Pharmacological Reports, 71(6), 983–993. https://doi.org/10.1016/j.pharep.2019.05.013
Hasan, M. M., Tasmin, M. S., El-Shehawi, A. M., Elseehy, M. M., Reza, M. A., & Haque, A. (2021). R. vesicarius L. exerts nephroprotective effect against cisplatin-induced oxidative stress. BMC Complementary Medicine and Therapies, 21(1), 225. https://doi.org/10.1186/s12906-021-03398-9
Xue, W., Lei, J., Li, X., & Zhang, R. (2011). Trigonella foenum graecum seed extract protects kidney function and morphology in diabetic rats via its antioxidant activity. Nutrition Research, 31(7), 555–562. https://doi.org/10.1016/j.nutres.2011.05.010
Akacha, A., Rebai, T., Zourgui, L., & Amri, M. (2018). Preventive effect of ethanolic extract of cactus (Opuntia ficus-indica) cladodes on methotrexate-induced oxidative damage of the small intestine in Wistar rats. Journal of Cancer Research and Therapeutics, 14(10), 779.
Hu, Y., Li, M., Lu, Q., Weng, H., Wang, J., & Zhao, H. (2019). A statistical framework for cross-tissue transcriptome-wide association analysis. Nature Genetics, 51(3), 568–576. https://doi.org/10.1038/s41588-019-0345-7
Ganesan, P., Matsubara, K., Ohkubo, T., Tanaka, Y., Noda, K., Sugawara, T., & Hirata, T. (2010). Anti-angiogenic effect of siphonaxanthin from the green alga Codium fragile. Phytomedicine, 17(14), 1140–1144. https://doi.org/10.1016/j.phymed.2010.05.005
Azam, G., Jayanna, S. G., Nelliankla, A., Boraiah, V., Hanumegowda, S. M., Sannaningaiah, D., Vijendra, P. D., Kumar, V., & Mahmood, R. (2021). Evaluation of in vitro antioxidant, anti-inflammatory, anticoagulant and antiplatelet potential of Rhus mysorensis. Biomedicine, 41(4), 724–731. https://doi.org/10.51248/.v41i4.1365
Lamponi, S., Baratto, M. C., Miraldi, E., Baini, G., & Biagi, M. (2021). Chemical profile, antioxidant, anti-proliferative, anticoagulant and mutagenic effects of a hydroalcoholic extract of Tuscan Rosmarinus officinalis. Plants, 10(1), 97. https://doi.org/10.3390/plants10010097
Hanumegowda, S., Srinivasa, C., Shivaiah, A., Venkatappa, M., Hanumanthappa, R., Rangappa, R., Sannaningaiah, D., et al. (2022). Protein extract of kenaf seed exhibits anticoagulant, antiplatelet and antioxidant activities. Asian Pacific Journal of Tropical Biomedicine, 12(2), 47. https://doi.org/10.4103/2221-1691.335693
Anastasiou, G., Gialeraki, A., Merkouri, E., Politou, M., & Travlou, A. (2012). Thrombomodulin as a regulator of the anticoagulant pathway: Implication in the development of thrombosis. Blood Coagulation & Fibrinolysis, 23(1), 1–10. https://doi.org/10.1097/MBC.0b013e32834cb271
Ardlie, N. G., & Han, P. (1974). Enzymatic basis for platelet aggregation and release: The significance of the ‘platelet atmosphere’ and the relationship between platelet function and blood coagulation. British journal of hematology, 26, 331–356.
Beers, R. F., & Sizer, I. W. (1952). A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. Journal of Biological Chemistry, 195, 133–140. https://doi.org/10.1016/S0021-9258(19)50881-X
Bergendi, L., Beneš, L., Ďuračková, Z., & Ferenčik, M. (1999). Chemistry, physiology and pathology of free radicals. Life Sciences, 65, 1865–1874. https://doi.org/10.1016/S0024-3205(99)00439-7
Davidović-Plavšić, B., Kukavica, B., Škondrić, S., Jimenez-Gallardo, C., & Žabić, M. (2021). Wild garlic extract reduces lipid peroxidation in terbuthylazine-treated human erythrocytes. Biomarkers, 26(7), 617–624. https://doi.org/10.1080/1354750X.2021.1953598
Denis, C., Methia, N., Frenette, P. S., et al. (1998). A mouse model of severe von Willebrand disease: Defects in hemostasis and thrombosis. PNAS, 95, 9524–9529. https://doi.org/10.1073/pnas.95.16.9524
Edziri, H., Jaziri, R., Haddad, O., Anthonissen, R., Aouni, M., Mastouri, M., & Verschaeve, L. (2019). Phytochemical analysis, antioxidant, anticoagulant and in vitro toxicity and genotoxicity testing of methanolic and juice extracts of Beta vulgaris L. South African Journal of Botany, 126, 170–175. https://doi.org/10.1016/j.sajb.2019.01.017
Elshopakey, G. E., & Elazab, S. T. (2021). Cinnamon aqueous extract attenuates diclofenac sodium and oxytetracycline mediated hepato-renal toxicity and modulates oxidative stress, cell apoptosis, and inflammation in male albino rats. Veterinary Sciences, 8(1), 9. https://doi.org/10.3390/vetsci8010009
Harborne, J. B. (1980). Phytochemical methods. Springer.
Iloki-Assanga, S. B., Lewis-Luján, L. M., Lara-Espinoza, C. L., Gil-Salido, A. A., Fernandez-Angulo, D., Rubio-Pino, J. L., & Haines, D. D. (2015). Solvent effects on phytochemical constituent profiles and antioxidant activities, using four different extraction formulations for analysis of Bucida buceras L. and Phoradendron californicum. BMC Research Notes, 8(1), 396. https://doi.org/10.1186/s13104-015-1388-1
Justesen, U., Knuthsen, P., & Leth, T. (1998). Quantitative analysis of flavonols, flavones, and flavanones in fruits, vegetables and beverages by high-performance liquid chromatography with photo-diode array and mass spectrometric detection. Journal of Chromatography A, 799(1–2), 101–110. https://doi.org/10.1016/S0021-9673(97)01061-3
Kauffman, D. (2020). Cardiovascular disease burden, deaths are rising around the world. American College of Cardiology, 9.
Kiefmann, R., Rifkind, J. M., Nagababu, E., & Bhattacharya, J. (2008). Red blood cells induce hypoxic lung inflammation. Blood, The Journal of the American Society of Hematology, 111(10), 5205–5214.
Kengaiah, J., Nandish, S. K. M., Ramachandraiah, C., Srinivasa, C., Shivaiah, A., & Sannaningaiah, D. (2019). In-vitro and in-vivo studies of Tamarind seed edible extract reveals anti-oxidative, anticoagulant, antiplatelet events. Asian Journal of Pharmacy and Pharmacology, 5(6), 1104–1116. https://doi.org/10.31024/ajpp.2019.5.6.5
Kim, K., Li, J., Tseng, A., Andrews, R. K., & Cho, J. (2015). NOX2 is critical for heterotypic neutrophil-platelet interactions during vascular inflammation. Blood, 126(16), 1952–1964. https://doi.org/10.1182/blood-2014-10-605261
Lakshmi, T., Ramasamy, R., & Thirumalaikumaran, R. (2015). Preliminary phytochemical analysis and in vitro antioxidant, FTIR spectroscopy, anti-diabetic activity of Acacia catechu ethanolic seed extract. Pharmacognosy Journal, 7(6), 356–362. https://doi.org/10.5530/pj.2015.6.7
Pham-Huy, L. A., He, H., & Pham-Huy, C. (2008). Free radicals, antioxidants in disease and health. International Journal of Biomedical Science: IJBS, 4(2), 89.
Luqman, S., & Rizvi, S. I. (2006). Protection of lipid peroxidation and carbonyl formation in proteins by capsaicin in human erythrocytes subjected to oxidative stress. Phytotherapy Research: An International Journal Devoted to Pharmacological and Toxicological Evaluation of Natural Product Derivatives, 20(4), 303–306.
Madamanchi, N. R., Hakim, Z. S., & Runge, M. S. (2005). Oxidative stress in atherogenesis and arterial thrombosis: The disconnect between cellular studies and clinical outcomes. Journal of Thrombosis and Haemostasis, 3(2), 254–267. https://doi.org/10.1111/j.1538-7836.2004.01085.x
Medini, H., Marzouki, H., Chemli, R., Khouja, M. L., Marongiu, B., Piras, A., & Tuveri, E. (2009). Comparison of the antimicrobial activity and the essential oil composition of Juniperus oxycedrus subsp macrocarpa and J oxycedrus subsp rufescens obtained by hydrodistillation and supercritical carbon dioxide extraction methods. Chemistry of Natural Compounds, 45(5), 739–741.
Mohamed, A., Bhardwaj, H., Hamama, A., & Webber, C. (1995). Chemical composition of kenaf (Hibiscus cannabinus L) seed oil. Industrial Crops and Products, 4(3), 157–165. https://doi.org/10.1016/0926-6690(95)00027-A
Nandish, S. M., Kengaiah, J., Ramachandraiah, C., Shivaiah, A., Chandramma, Girish, K., Kemparaju, K., & Sannaningaiah, D. (2018). Anticoagulant, antiplatelet and fibrin clot hydrolyzing activities of flax seed buffer extract. Pharmacognosy Magazine, 14(55), 175. https://doi.org/10.4103/pm.pm_320_17
Nerdy, N., & Ritarwan, K. (2019). Hepatoprotective activity and nephroprotective activity of peel extract from three varieties of the passion fruit (Passiflora Sp) in the albino rat. Open Access Macedonian Journal of Medical Sciences, 7(4), 536–542. https://doi.org/10.3889/oamjms.2019.153
Prandoni, P. (2016). Venous and arterial thrombosis: Is there a link? In Md. S. Islam (Ed.), Thrombosis and embolism: From research to clinical practice (Vol. 906, pp. 273–283). Cham: Springer International Publishing. https://doi.org/10.1007/5584_2016_121
Sannaningaiah, D., Shivaiah, A., Kengaiah, J., Srinivasa, C., Nandish, S. K. M., Ramachandraiah, C., Hanumegowda, S., & Shinde, M. (2021). Sorghum protein extract protects RBC from sodium nitrite-induced oxidative stress and exhibits anticoagulant and antiplatelet activity. Folia Medica, 63(6), 884–894. https://doi.org/10.3897/folmed.63.e57713
Singh, R., Devi, S., & Gollen, R. (2015). Role of free radicals in atherosclerosis, diabetes and dyslipidaemia: Larger-than-life: Role of free radicals in atherosclerosis, diabetes and dyslipidaemia. Diabetes/Metabolism Research and Reviews, 31(2), 113–126. https://doi.org/10.1002/dmrr.2558
Simon, J. P., & Evan Prince, S. (2018). Aqueous leaves extract of Madhuca longifolia attenuate diclofenac-induced hepatotoxicity: Impact on oxidative stress, inflammation, and cytokines. Journal of Cellular Biochemistry, 119(7), 6125–6135. https://doi.org/10.1002/jcb.26812
Veenith, T., Martin, H., Le Breuilly, M., Whitehouse, T., Gao-Smith, F., Duggal, N., & Moss, P. (2022). High generation of reactive oxygen species from neutrophils in patients with severe COVID-19. Scientific Reports, 12(1), 10484. https://doi.org/10.1038/s41598-022-13825-7
Vilesgonzalez, J. (2004). Atherothrombosis: A widespread disease with unpredictable and life-threatening consequences*1. European Heart Journal, 25(14), 1197–1207. https://doi.org/10.1016/j.ehj.2004.03.011
Acknowledgements
The authors thank Vision Group of Science and Technology, Government of Karnataka, India, for financial support and Liveon Biolabs Private Limited, Tumkur, Karnataka, India, for extending animal studies. The authors also thank the Department of Science and Technology (DST), Govt. of India, for providing the instrumentation facility under the scheme ‘Fund for Improvement of Science and Technology’ (Grant No. FIST-No. SR/FST/LS1/2018/175(C)) to the Department of Biochemistry, Kuvempu University Shankaraghatta, Karnataka, to achieve this piece of work and for awarding inspire fellowship to carry out the Ph.D. degree of the candidate.
Funding
This work is supported by Vision Group of Science and Technology, Government of Karnataka, India (VGST/CISEE/2012–13/282, dated March 16th, 2013). DST, New Delhi 2018/IF_180400.
Author information
Authors and Affiliations
Contributions
SH and DS together planned and designed the research work. CR, AS, and MV assisted in the laboratory work. SSM, RR, and SG reviewed the article.
Corresponding authors
Ethics declarations
Ethics Approval
The research work involving only Sprague Dawley Female rats weighing 150–180 g was procured from Liveon Biolabs, limited. Institutional Animal ethical committee (TUMKUR, INDIA). All the experimentations were conducted under ethical guidelines and were approved by the Institutional Human Ethical Committee, Tumkur University, and Tumkur. Conducting animal experiments was permitted by the Institutional Animal Ethical Committee, Liveon Biolabs Private Limited, Tumkur. The animal handling was preceded following the guidelines of the Committee for Monitoring and Supervision of Experiments on Animals (CPCSEA). Ethical clearance was taken for this study from Liveon Biolabs, limited. Institutional Animal ethical committee (LBLIAEC) TUMKUR, with a protocol number LBPL-IAEC-47–05-2019.
Consent to Participate
All authors have their consent to participate.
Consent for Publication
All authors have their consent to publish their work.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hanumegowda, S.M., Srinivasa, C., Shivaiah, A. et al. Antioxidant and Antithrombotic Activities of Kenaf Seed (Hibiscus cannabinus) Coat Ethanol Extract in Sprague Dawley Rats. Appl Biochem Biotechnol 195, 772–800 (2023). https://doi.org/10.1007/s12010-022-04144-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-04144-8