Abstract
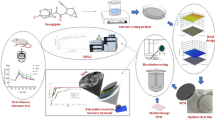
Quercetin (QCN) is a plant polyphenol with a variety of medicinal effects. Poor water solubility, on the other hand, restricts its therapeutic effectiveness. The purpose of this study was to develop mixed micellar systems using two biocompatible amphiphilic PEO-PPO-PEO triblock copolymers, Pluronic P123 (EO20-PO70-EO20) and Pluronic F88 (EO104-PO39-EO104), in order to enhance the aqueous solubility and oral bioavailability of QCN drug. The critical micelle concentrations (CMCs) of mixed P123/F88 micellar solutions were investigated using UV–visible spectroscopy with pyrene as a probe. Mixed P123/F88 micelles have low CMCs, indicating that they have a stable micelle structure even when diluted. The solubility of QCN in aqueous mixed P123/F88 micellar solutions at different temperatures was investigated to better understand drug entrapment. The QCN solubility increased with increasing temperature in the mixed P123/F88 micellar system. The QCN-incorporated mixed P123/F88 micelles were prepared using the thin-film hydration method and were well characterized in terms of size and morphology, compatibility, in vitro release and antioxidant profile. In addition, the cell proliferation activity of the mixed micelles was evaluated in the MCF-7 cell line. The QCN-incorporated mixed P123/F88 micelles had a small particle size (< 25 nm) and a negative zeta potential with a spherical shape. The in vitro release behaviour of QCN from a mixed P123/F88 micellar system was slower and more sustained at physiological conditions. The oxidation resistance of QCN-incorporating mixed P123/F88 micelles was shown to be considerably higher than that of pure QCN. An in vitro cell proliferation study revealed that QCN-incorporated mixed micells were effective in inhibiting tumour cell growth. In conclusion, the QCN-incorporated mixed P123/F88 micelle may be a promising approach to increase QCN oral bioavailability, antioxidant activity, and cell viability.
Graphical Abstract









Similar content being viewed by others
Availability of Data and Materials
The authors confirm that all data underlying the findings are fully available without restriction. All relevant data are within the paper.
References
Date, A. A., Nagarsenker, M. S., Patere, S., Dhawan, V., Gude, R. P., Hassan, P. A., & Fahr, A. (2011). Lecithin-based novel cationic nanocarriers (Leciplex) II: Improving therapeutic efficacy of quercetin on oral administration. Molecular Pharmaceutics, 8(3), 716–726.
Mukhopadhyay, P., & Prajapati, A. K. (2015). Quercetin in anti-diabetic research and strategies for improved quercetin bioavailability using polymer-based carriers–a review. RSC Advances, 5(118), 97547–97562.
Chen, C., Zhou, J., & Ji, C. (2010). Quercetin: A potential drug to reverse multidrug resistance. Life Sciences, 87(11–12), 333–338.
Rice-evans, C. A., Miller, N. J., Bolwell, P. G., Bramley, P. M., & Pridham, J. B. (1995). The relative antioxidant activities of plant-derived polyphenolic flavonoids. Free Radical Research, 22(4), 375–383.
Mangels, A. R., Holden, J. M., Beecher, G. R., Forman, M. R., & Lanza, E. (1993). Carotenoid content of fruits and vegetables: an evaluation of analytic data. Journal of the American Dietetic Association, 93(3), 284–296.
Bronner, C., & Landry, Y. (1985). Kinetics of the inhibitory effect of flavonoids on histamine secretion from mast cells. Agents and Actions., 16(3), 147–151.
Kaul, T. N., Middleton, E., Jr., & Ogra, P. L. (1985). Antiviral effect of flavonoids on human viruses. Journal of Medical Virology, 15(1), 71–79.
Hanasaki, Y., Ogawa, S., & Fukui, S. (1994). The correlation between active oxygens scavenging and antioxidative effects of flavonoids. Free Radical Biology & Medicine, 16(6), 845–850.
Morand, C., Crespy, V., Manach, C., Besson, C., Demigne, C., & Remesy, C. (1998). Plasma metabolites of quercetin and their antioxidant properties. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 275(1), R212–R219.
Plumb, G. W., Price, K. R., & Williamson, G. (1999). Antioxidant properties of flavonol glycosides from green beans. Redox Report, 4(3), 123–127.
Fiorani, M., de Sanctis, R., Menghinello, P., Cucchiarini, L., Cellini, B., & Dachà, M. (2001). Quercetin prevents glutathione depletion induced by dehydroascorbic acid in rabbit red blood cells. Free Radical Research, 34(6), 639–648.
Pralhad, T., & Rajendrakumar, K. (2004). Study of freeze-dried quercetin–cyclodextrin binary systems by DSC, FT-IR, X-ray diffraction and SEM analysis. Journal of Pharmaceutical and Biomedical Analysis, 34(2), 333–339.
Movassaghian, S., Merkel, O. M., & Torchilin, V. P. (2015). Applications of polymer micelles for imaging and drug delivery. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology, 7(5), 691–707.
Chiappetta, D. A., & Sosnik, A. (2007). Poly (ethylene oxide)–poly (propylene oxide) block copolymer micelles as drug delivery agents: improved hydrosolubility, stability and bioavailability of drugs. European Journal of Pharmaceutics and Biopharmaceutics, 66(3), 303–317.
Gaucher, G., Dufresne, M. H., Sant, V. P., Kang, N., Maysinger, D., & Leroux, J. C. (2005). Block copolymer micelles: preparation, characterization and application in drug delivery. Journal of Controlled Release, 109(1–3), 169–188.
Bae, Y. H., & Yin, H. (2008). Stability issues of polymeric micelles. Journal of Controlled Release, 131(1), 2–4.
Huang, C. K., Lo, C. L., Chen, H. H., & Hsiue, G. H. (2007). Multifunctional micelles for cancer cell targeting, distribution imaging, and anticancer drug delivery. Advanced Functional Materials, 17(14), 2291–2297.
Oerlemans, C., Bult, W., Bos, M., Storm, G., Nijsen, J. F. W., & Hennink, W. E. (2010). Polymeric micelles in anticancer therapy: targeting, imaging and triggered release. Pharmaceutical Research, 27(12), 2569–2589.
Pitto-Barry, A., & Barry, N. P. (2014). Pluronic® block-copolymers in medicine: from chemical and biological versatility to rationalisation and clinical advances. Polymer Chemistry, 5(10), 3291–3297.
Almeida, M., Magalhães, M., Veiga, F., & Figueiras, A. (2018). Poloxamers, poloxamines and polymeric micelles: Definition, structure and therapeutic applications in cancer. Journal of Polymer Research, 25(1), 1–14.
Gente, G., Iovino, A., & La Mesa, C. (2004). Supramolecular association of a triblock copolymer in water. Journal of Colloid and Interface Science, 274(2), 458–464.
Alvarez-Lorenzo, C., Sosnik, A., & Concheiro, A. (2011). PEO-PPO block copolymers for passive micellar targeting and overcoming multidrug resistance in cancer therapy. Current Drug Targets, 12(8), 1112–1130.
Chen, Y., Khan, A. R., Yu, D., Zhai, Y., Ji, J., Shi, Y., & Zhai, G. (2019). Pluronic F127-functionalized molybdenum oxide nanosheets with pH-dependent degradability for chemo-photothermal cancer therapy. Journal of Colloid and Interface Science, 553, 567–580.
Hu, H., Petrosyan, A., Osna, N. A., Liu, T., Olou, A. A., Alakhova, D. Y., & Bronich, T. K. (2019). Pluronic block copolymers enhance the anti-myeloma activity of proteasome inhibitors. Journal of Controlled Release, 306, 149–164.
Alexandridis, P., Holzwarth, J. F., & Hatton, T. A. (1994). Micellization of poly (ethylene oxide)-poly (propylene oxide)-poly (ethylene oxide) triblock copolymers in aqueous solutions: thermodynamics of copolymer association. Macromolecules, 27(9), 2414–2425.
Wakaskar, R. R., Bathena, S. P. R., Tallapaka, S. B., Ambardekar, V. V., Gautam, N., Thakare, R., & Vetro, J. A. (2015). Peripherally cross-linking the shell of core-shell polymer micelles decreases premature release of physically loaded combretastatin A4 in whole blood and increases its mean residence time and subsequent potency against primary murine breast tumors after IV administration. Pharmaceutical Research, 32(3), 1028–1044.
Sezgin, Z., Yüksel, N., & Baykara, T. (2006). Preparation and characterization of polymeric micelles for solubilization of poorly soluble anticancer drugs. European Journal of Pharmaceutics and Biopharmaceutics, 64(3), 261–268.
Batrakova, E. V., & Kabanov, A. V. (2008). Pluronic block copolymers: evolution of drug delivery concept from inert nanocarriers to biological response modifiers. Journal of Controlled Release, 130(2), 98–106.
Tiwari, S., Kansara, V., & Bahadur, P. (2020). Targeting anticancer drugs with pluronic aggregates: Recent updates. International Journal of Pharmaceutics., 586, 119544.
Xiao, B., Zhang, M., Viennois, E., Zhang, Y., Wei, N., Baker, M. T., & Merlin, D. (2015). Inhibition of MDR1 gene expression and enhancing cellular uptake for effective colon cancer treatment using dual-surface-functionalized nanoparticles. Biomaterials, 48, 147–160.
Zhang, Y., Song, W., Geng, J., Chitgupi, U., Unsal, H., Federizon, J., & Lovell, J. F. (2016). Two-dimensional antimonene single crystals grown by van der Waals epitaxy. Nature Communications, 7(1), 1–9.
Aydin, F., Chu, X., Uppaladadium, G., Devore, D., Goyal, R., Murthy, N. S., & Dutt, M. (2016). Self-assembly and critical aggregation concentration measurements of ABA triblock copolymers with varying B block types: model development, prediction, and validation. The Journal of Physical Chemistry B, 120(15), 3666–3676.
Jackson, J. K., Springate, C. M., Hunter, W. L., & Burt, H. M. (2000). Neutrophil activation by plasma opsonized polymeric microspheres: inhibitory effect of Pluronic F127. Biomaterials, 21(14), 1483–1491.
Lee, E. S., Oh, Y. T., Youn, Y. S., Nam, M., Park, B., Yun, J., & Oh, K. T. (2011). Binary mixing of micelles using Pluronics for a nano-sized drug delivery system. Colloids and Surfaces B: Biointerfaces, 82(1), 190–195.
Sharma, R. K., Shaikh, S., Ray, D., & Aswal, V. K. (2018). Binary mixed micellar systems of PEO-PPO-PEO block copolymers for lamotrigine solubilization: a comparative study with hydrophobic and hydrophilic copolymer. Journal of Polymer Research, 25(3), 1–11.
Nandni, D., Vohra, K. K., & Mahajan, R. K. (2009). Study of micellar and phase separation behavior of mixed systems of triblock polymers. Journal of Colloid and Interface Science, 338(2), 420–427.
Aswal, V. K., & Goyal, P. S. (2000). Small-angle neutron scattering diffractometer at Dhruva reactor. Current Science, 79(7), 947–953.
Gabriele, B., Fazio, A., Carchedi, M., & Plastina, P. (2012). In vitro antioxidant activity of extracts of Sybaris liquorice roots from Southern Italy. Natural Product Research, 26(23), 2176–2181.
Pursuwani, B. H., Bhatt, B. S., Vaidya, F. U., Pathak, C., & Patel, M. N. (2021). etrazolo [1, 5-a] quinoline moiety-based Os (IV) complexes: DNA binding/cleavage, bacteriostatic and photocytotoxicity assay. Journal of Biomolecular Structure & Dynamics, 39(8), 2894–2903.
Liu, Y., Yang, J., Wang, X., Liu, J., Wang, Z., Liu, H., & Chen, L. (2016). In vitro and in vivo evaluation of redoxresponsive sorafenib carrier nanomicelles synthesized from poly (acryic acid)-cystamine hydrochloride-D-α-tocopherol succinate. Journal of Biomaterials Science, Polymer Edition, 27(17), 1729–1747.
Cagel, M., Tesan, F. C., Bernabeu, E., Salgueiro, M. J., Zubillaga, M. B., Moretton, M. A., & Chiappetta, D. A. (2017). Polymeric mixed micelles as nanomedicines: Achievements and perspectives. European Journal of Pharmaceutics and Biopharmaceutics, 113, 211–228.
da Silva, R. C., & Loh, W. (1998). Effect of additives on the cloud points of aqueous solutions of ethylene oxide–propylene oxide–ethylene oxide block copolymers. Journal of Colloid and Interface Science, 202(2), 385–390.
Span, K., Verhoef, J. J., Hunt, H., van Nostrum, C. F., Brinks, V., Schellekens, H., & Hennink, W. E. (2016). A novel oral iron-complex formulation: Encapsulation of hemin in polymeric micelles and its in vitro absorption. European Journal of Pharmaceutics and Biopharmaceutics, 108, 226–234.
Zhao, L., Du, J., Duan, Y., Zhang, H., Yang, C., Cao, F., & Zhai, G. (2012). Curcumin loaded mixed micelles composed of Pluronic P123 and F68: preparation, optimization and in vitro characterization. Colloids and Surfaces B: Biointerfaces, 97, 101–108.
Alexandridis, P., Nivaggioli, T., & Hatton, T. A. (1995). Temperature effects on structural properties of Pluronic P104 and F108 PEO-PPO-PEO block copolymer solutions. Langmuir, 11(5), 1468–1476.
Zhao, L., Shi, Y., Zou, S., Sun, M., Li, L., & Zhai, G. (2011). Formulation and in vitro evaluation of quercetin loaded polymeric micelles composed of pluronic P123 and Da-tocopheryl polyethylene glycol succinate. Journal of Biomedical Nanotechnology, 7(3), 358–365.
Fraile, M., Buratto, R., Gomez, B., Martin, A., & Cocero, M. J. (2014). Enhanced delivery of quercetin by encapsulation in poloxamers by supercritical antisolvent process. Industrial and Engineering Chemistry Research, 53(11), 4318–4327.
Su, Y. L., Wang, J., & Liu, H. Z. (2002). FTIR spectroscopic study on effects of temperature and polymer composition on the structural properties of PEO− PPO− PEO block copolymer micelles. Langmuir, 18(14), 5370–5374.
Cai, X., Liu, M., Zhang, C., Sun, D., & Zhai, G. (2016). pH-responsive copolymers based on pluronic P123-poly (β-amino ester): synthesis, characterization and application of copolymer micelles. Colloids and Surfaces B: Biointerfaces, 142, 114–122.
Lu, Z., Bu, C., Hu, W., Zhang, H., Liu, M., Lu, M., & Zhai, G. (2018). Preparation and in vitro and in vivo evaluation of quercetin-loaded mixed micelles for oral delivery. Bioscience, Biotechnology, and Biochemistry, 82(2), 238–246.
Mu, L., Teo, M. M., Ning, H. Z., Tan, C. S., & Feng, S. S. (2005). Novel powder formulations for controlled delivery of poorly soluble anticancer drug: Application and investigation of TPGS and PEG in spray-dried particulate system. Journal of Controlled Release, 103(3), 565–575.
Boots, A. W., Li, H., Schins, R. P., Duffin, R., Heemskerk, J. W., Bast, A., & Haenen, G. R. (2007). The quercetin paradox. Toxicology and Applied Pharmacology, 222(1), 89–96.
Acknowledgements
The corresponding author acknowledged Center of Excellence in Polymers (CoEP), Applied Chemistry Department, Faculty of Technology and Engineering for necessary lab facilities.
Funding
This work was supported by the UGC-DAE Research Project (CRS-M-205), Bhabha Atomic Research Centre, Mumbai, Maharashtra, India.
Author information
Authors and Affiliations
Contributions
Hemil S Patel, Sofiya J Shaikhand Rakesh K Sharma designed the study, analysed the data, and wrote the final manuscript. Debes Ray andVinod K Aswal contributed to the SANS measurments. Foram Vaidya and Chandramani Pathak carried out the molecular biology studies. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
I confirm that the final manuscript has been seen and approved by all the authors. The undersigned author transfers all copyright ownership of the manuscript to the International Journal of Applied Biochemistry and Biotechnology in the event the work is published.
Consent for Publication
The authors declare that they consent for publication.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Patel, H.S., Shaikh, S.J., Ray, D. et al. Formulation, Solubilization, and In Vitro Characterization of Quercetin-Incorporated Mixed Micelles of PEO-PPO-PEO Block Copolymers. Appl Biochem Biotechnol 194, 445–463 (2022). https://doi.org/10.1007/s12010-021-03691-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-021-03691-w