Abstract
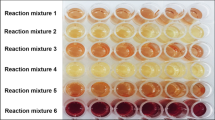
For several years, researchers have studied the formation of zinc protoporphyrin IX in meat, as it is considered to be an important natural colouring agent in dry cured or fermented meat products in the absence of nitrite and/ or nitrate. Until now, however, mainly pork meat is used for these investigations. The goal of this research was to relate in vitro zinc protoporphyrin IX and protoporphyrin IX formation in eight meat sources (chicken, turkey, pork, lamb, beef, veal, horse and porcine liver) to eight intrinsic parameters using partial least squares regression (PLS) analysis. Significant differences in pH, initial metmyoglobin formation, metmyoglobin reduction ability, total heme, zinc chelatase activity, and total iron and zinc concentration between meat sources were found. Water activity, however, was not significantly different between meat sources. Liver tissue and horse meat showed the best ability to form zinc protoporphyrin IX. Formation of protoporphyrin IX was limited in all meat sources. PLS analysis revealed that mainly zinc chelatase activity, followed by total heme, total iron and zinc content, were predominant intrinsic parameters to explain variations in zinc protoporphyrin IX formation. These findings could be important for meat industry in order to establish the production of red coloured nitrite-free meat products.




Similar content being viewed by others
References
Adamsen, C. E., Møller, J. K. S., Laursen, K., Olsen, K., & Skibsted, L. H. (2006). Zn-porphyrin formation in cured meat products: effect of added salt and nitrite. Meat Science, 72(4), 672–679.
Ajioka, R. S., Phillips, J. D., & Kushner, J. P. (2006). Biosynthesis of heme in mammals. Biochimica et Biophysica Acta, 1763(7), 723–736.
AMSA. (2012). AMSA Meat Color Measurement Guidelines Measurement.
Becker, E. M., Westermann, S., Hansson, M., & Skibsted, L. H. (2012). Parallel enzymatic and non-enzymatic formation of zinc protoporphyrin IX in pork. Food Chemistry, 130(4), 832–840.
Belgische voedingsmiddelentabel, Nubel vzw, 5e editie (2010).
Chan, J. T. Y., Omana, D. A., & Betti, M. (2011). Effect of ultimate pH and freezing on the biochemical properties of proteins in Turkey breast meat. Food Chemistry, 127(1), 109–117.
Chau, T. T., Ishigaki, M., Kataoka, T., & Taketani, S. (2010). Porcine ferrochelatase: the relationship between iron-removal reaction and the conversion of heme to Zn-protoporphyrin. Bioscience, Biotechnology, and Biochemistry, 74(7), 1415–1420.
Chau, T. T., Ishigaki, M., Kataoka, T., & Taketani, S. (2011). Ferrochelatase catalyzes the formation of Zn-protoporphyrin of dry-cured ham via the conversion reaction from heme in meat. Journal of Agricultural and Food Chemistry, 59(22), 12238–12245.
Cross, A. A. J., Harnly, J. J. M., Ferrucci, L. M. L., Risch, A., Mayne, S. T., & Sinha, R. (2012). Developing a heme iron database for meats according to meat type, cooking method and doneness level. Food and Nutrition Sciences, 3(7), 905–913.
Dailey, H. A., Dailey, T. A., Wu, C., Medlock, A. E., Wang, K., Rose, J. P., & Wang, B. (2000). Ferrochelatase at the millennium: structures , mechanisms and [ 2Fe-2S ] clusters. Cellular and Molecular Life Sciences, 57, 1909–1926.
De Maere, H., Fraeye, I., De Mey, E., Dewulf, L., Michiels, C., Paelinck, H., & Chollet, S. (2016). Formation of naturally occurring pigments during the production of nitrite-free dry fermented sausages. Meat Science, 114, 1–7.
Devine, C., & Dikeman, M. (2004). Encyclopedia of Meat Sciences ed. Elsevier Acad: Press, Oxford.
Durek, J., Bolling, J. S., Knorr, D., Schwägele, F., & Schlüter, O. (2012). Effects of different storage conditions on quality related porphyrin fluorescence signatures of pork slices. Meat Science, 90(1), 252–258.
Elroy, N. N., Rogers, J., Mafi, G. G., Van Overbeke, D. L., Hartson, S. D., & Ramanathan, R. (2015). Species-specific effects on non-enzymatic metmyoglobin reduction in vitro. Meat Science, 105, 108–113.
Estévez, M., & Cava, R. (2004). Lipid and protein oxidation, release of iron from heme molecule and colour deterioration during refrigerated storage of liver pâté. Meat Science, 68(4), 551–558.
Ferreira, G. C. (1999). Ferrochelatase. The International Journal of Biochemistry & Cell Biology, 31(10), 995–1000.
Gill, C. O. (2005). Safety and storage stability of horse meat for human consumption. Meat Science, 71(3), 506–513.
Grossi, A. B., do Nascimento, E. S. P., Cardoso, D. R., & Skibsted, L. H. (2014). Proteolysis involvement in zinc–protoporphyrin IX formation during Parma ham maturation. Food Research International, 56, 252–259.
Gurtler, J. B., Doyle, M. P., & Kornacki, J. L. (2014). The microbiological safety of spices and low-water activity foods and spices. Food microbiology and food safety. New York: Springer Science.
Hunter, G. A., Sampson, M. P., & Ferreira, G. C. (2008). Metal ion substrate inhibition of ferrochelatase. The Journal of Biological Chemistry, 283(35), 23685–23691.
International Organization for Standardization (1997). Determination of moisture content, ISO 1442:1997 standard.
Ishikawa, H., Yoshihara, M., Baba, A., Kawabuchi, T., Sato, M., Numata, M., & Matsumoto, K. (2006). Formation of zinc protoporphyrin IX from myoglobin with pork loin extract. Journal of the Faculty of Agriculture, 51(1), 93–97.
Ishikawa, H., Kawabuchi, T., Kawakami, Y., Sato, M., Numata, M., & Matsumoto, K. (2007). Formation of zinc protoporphyrin IX and protoporphyrin IX from oxymyoglobin in porcine heart mitochondria. Food Science and Technology Research, 13(1), 85–88.
Jacobs, J. M., Sinclair, P. R., Sinclair, J. F., Gorman, N., Walton, H. S., Wood, S. G., & Nichols, C. (1998). Formation of zinc protoporphyrin in cultured hepatocytes: effects of ferrochelatase inhibition, iron chelation or lead. Toxicology, 125(2–3), 95–105.
Jay, J. M., Loessner, M. J., & Golden, D. A. (2005). Modern food microbiology (7th ed.). New York: Springer Science.
Klont, R. E., Barnier, V. M. H., Van Dijk, A., Smulders, F. J. M., Hoving-Bolink, A. H., Hulsegge, B., & Eikelenboom, G. (2000). Effects of rate of pH fall, time of deboning, aging period, and their interaction on veal quality characteristics. Journal of Animal Science, 78(7), 1845–1851.
Labbé, R. F., Vreman, H. J., & Stevenson, D. K. (1999). Zinc protoporphyrin: a metabolite with a mission. Clinical Chemistry, 45(12), 2060–2072.
Lammens, V., Peeters, E., De Maere, H., De Mey, E., Paelinck, H., Leyten, J., & Geers, R. (2007). A survey of pork quality in relation to pre-slaughter conditions, slaughterhouse facilities, and quality assurance. Meat Science, 75(3), 381–387.
Lindahl, G. (2005). Colour Characteristics of Fresh Pork. Doctor’s dissertation.
Litwinczuk, A., Florek, M., Skalecki, P., & Litwinczuk, Z. (2008). Chemical composition and physicochemical properties of horse meat from the longissimus. Journal of Muscle Foods, 19(3), 223–236.
Lombardi-Boccia, G., Martinez-Dominguez, B., & Aguzzi, A. (2002). Total heme and non-heme iron in raw and cooked meats. Journal of Food Chemistry and Toxicology, 67(5), 1738–1741.
Mancini, R. A., Seyfert, M., & Hunt, M. C. (2008). Effects of data expression, sample location, and oxygen partial pressure on initial nitric oxide metmyoglobin formation and metmyoglobin-reducing-activity measurement in beef muscle. Meat Science, 79(2), 244–251.
Mikkelsen, A., Juncher, D., & Skibsted, L. H. (1999). Metmyoglobin reductase activity in porcine M. longissimus dorsi muscle. Meat Science, 51(2), 155–161.
Parolari, G., Benedini, R., & Toscani, T. (2009). Color formation in nitrite-free dried hams as related to Zn-protoporphyrin IX and Zn-chelatase activity. Journal of Food Science, 74(6), 413–418.
Samel, L. M., Hunt, M. C., Kropf, D. H., Hachmeister, K. A., & Johnson, D. E. (2002). Comparison of assays for metmyoglobin reducing ability in beef inside and outside. Food Chemistry and Toxicology, 67(3), 978–984.
Schneider, J., Wulf, J., Surowsky, B., Schmidt, H., Schwägele, F., & Schlüter, O. (2008). Fluorimetric detection of protoporphyrins as an indicator for quality monitoring of fresh intact pork meat. Meat Science, 80(4), 1320–1325.
Wakamatsu, J., Nishimura, T., & Hattori, A. (2004a). A Zn-porphyrin complex contributes to bright red color in Parma ham. Meat Science, 67(1), 95–100.
Wakamatsu, J., Okui, J., Ikeda, Y., Nishimura, T., & Hattori, A. (2004b). Establishment of a model experiment system to elucidate the mechanism by which Zn-protoporphyrin IX is formed in nitrite-free dry-cured ham. Meat Science, 68(2), 313–317.
Wakamatsu, J., Okui, J., Hayashi, N., Nishimura, T., & Hattori, A. (2007). Zn protoporphyrin IX is formed not from heme but from protoporphyrin IX. Meat Science, 77(4), 580–586.
Wakamatsu, J., Odagiri, H., Nishimura, T., & Hattori, A. (2009). Quantitative determination of Zn protoporphyrin IX, heme and protoporphyrin IX in Parma ham by HPLC. Meat Science, 82(1), 139–142.
Wakamatsu, J., Murakami, N., & Nishimura, T. (2015). A comparative study of zinc protoporphyrin IX-forming properties of animal by-products as sources for improving the color of meat products. Animal Science Journal, 86, 547–552.
Warriss, P. D., Brown, S. N., Adams, S. J., & Lowe, D. B. (1990). Variation in haem pigment concentration and colour in meat from British pigs. Meat Science, 28(4), 321–329.
Zhu, L. G., & Brewer, M. S. (1998). Metmyoglobin reducing capacity of fresh normal, PSE, and DFD pork during retail display. Journal of Food Science, 63(3), 390–393.
Acknowledgments
This work was performed with financial support of internal funding of KU Leuven.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
De Maere, H., Chollet, S., Claeys, E. et al. In Vitro Zinc Protoporphyrin IX Formation in Different Meat Sources Related to Potentially Important Intrinsic Parameters. Food Bioprocess Technol 10, 131–142 (2017). https://doi.org/10.1007/s11947-016-1804-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-016-1804-0