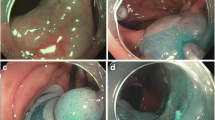
Opinion statement
Polypectomy reduces the incidence and mortality of colorectal cancer (CRC). The widespread adoption of CRC screening, more rigorous colonoscopy techniques, and advancements in endoscopic imaging have led to a greater awareness of complex polyps. Whereas surgery was once considered necessary for many large sessile or laterally spreading lesions (LSLs) in the colorectum, the majority can now be removed endoscopically. Endoscopic mucosal resection (EMR) is an established technique for treatment of colorectal LSLs. When performed by experts, EMR is highly effective and safe and can be completed in an outpatient or day-stay setting. Advancements in EMR effectiveness encompass a better understanding of the factors leading to post-EMR recurrence, protocols to recognize and treat it, and interventions that prevent recurrent or residual adenoma. New techniques for treating intra-procedural bleeding and a novel classification system to identify and inform proactive management of deep mural injury enhance the safety profile of EMR. However, each of these incremental advancements necessitates a meticulous and systematic approach that only committed and properly trained endoscopists can master. While alternative interventions such as endoscopic submucosal dissection (ESD) offer potential advantages over EMR, the added procedural complexity, risks, and costs limit the relevance of ESD to a minority of lesions in the colorectum. This article reviews the expanding body of evidence supporting EMR as the first-line treatment of colorectal LSLs ≥20 mm.





Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Zauber AG, Winawer SJ, O'Brien MJ, et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012;366:687–96.
Nishihara R, Wu K, Lochhead P, et al. Long-term colorectal-cancer incidence and mortality after lower endoscopy. N Engl J Med. 2013;369:1095–105.
Jensen CD, Corley DA, Quinn VP, et al. Fecal immunochemical test program performance over 4 rounds of annual screening: a retrospective cohort study. Ann Intern Med. 2016;164:456–63.
Jayanna M, Burgess NG, Singh R, et al. Cost analysis of endoscopic mucosal resection vs surgery for large laterally spreading colorectal lesions. Clin Gastroenterol Hepatol. 2016;14:271–8. e1-2
Ahlenstiel G, Hourigan LF, Brown G, et al. Actual endoscopic versus predicted surgical mortality for treatment of advanced mucosal neoplasia of the colon. Gastrointest Endosc. 2014;80:668–76.
Keswani RN, Law R, Ciolino JD, et al. Adverse events after surgery for nonmalignant colon polyps are common and associated with increased length of stay and costs. Gastrointest Endosc 2016;84:296–303 e1.
Moss A, Nalankilli K. Completing the circle of informed consent for EMR versus surgery for nonmalignant large or complex colorectal polyps. Gastrointest Endosc. 2016;84:304–6.
Friedland S, Banerjee S, Kochar R, et al. Outcomes of repeat colonoscopy in patients with polyps referred for surgery without biopsy-proven cancer. Gastrointest Endosc. 2014;79:101–7.
Moss A. From gastroenterologist to surgeon to gastroenterologist for management of large sessile colonic polyps: something new under the sun? Gastrointest Endosc. 2014;79:108–10.
Burgess NG, Bahin FF, Bourke MJ. Colonic polypectomy (with videos). Gastrointest Endosc. 2015;81:813–35.
Klein A, Bourke MJ. Advanced polypectomy and resection techniques. Gastrointest Endosc Clin N Am. 2015;25:303–33.
Hassan C, Repici A, Sharma P, et al. Efficacy and safety of endoscopic resection of large colorectal polyps: a systematic review and meta-analysis. Gut. 2016;65:806–20.
Moss A, Bourke MJ, Williams SJ, et al. Endoscopic mucosal resection outcomes and prediction of submucosal cancer from advanced colonic mucosal neoplasia. Gastroenterology. 2011;140:1909–18.
•• Moss A, Williams SJ, Hourigan LF, et al. Long-term adenoma recurrence following wide-field endoscopic mucosal resection (WF-EMR) for advanced colonic mucosal neoplasia is infrequent: results and risk factors in 1000 cases from the Australian Colonic EMR (ACE) study. Gut. 2015;64:57–65. This article demonstrates the long-term effectiveness of EMR based on a prospective analysis of 1000 cases. Among those who had successful initial EMR with or without endoscopic treatment of minor recurrence at 4 and 16 months, over 98% were adenoma free without sugery at the 16-month follow-up. Risk factors for recurrence were also identified and include lesion size >40 mm, use of argon plasma coagulation to ablate adenoma, and intra-procedural bleeding.
Khashab M, Eid E, Rusche M, et al. Incidence and predictors of “late” recurrences after endoscopic piecemeal resection of large sessile adenomas. Gastrointest Endosc. 2009;70:344–9.
Pellise M, Burgess NG, Tutticci N, et al. Endoscopic mucosal resection for large serrated lesions in comparison with adenomas: a prospective multicentre study of 2000 lesions. Gut 2016. doi:10.1136/gutjnl-2015-310249.
Rao AK, Soetikno R, Raju GS, et al. Large sessile serrated polyps can be safely and effectively removed by endoscopic mucosal resection. Clin Gastroenterol Hepatol. 2016;14:568–74.
Bahin FF, Rasouli KN, Byth K, et al. Prediction of clinically significant bleeding following wide-field endoscopic resection of large sessile and laterally spreading colorectal lesions: a clinical risk score. Am J Gastroenterol. 2016;111:1115–22.
• Tate DJ, Desomer L, Klein A, et al. Adenoma recurrence after piecemeal colonic EMR is predictable; the Sydney EMR Recurrence Tool. Gastrointest Endosc. 2016. doi:10.1016/j.gie.2016.11.027.This study evaluates the temporal profile of adenoma recurrence and combines established risk factors into a clinical score predicting low-risk patients that may avoid early surveillance at 4-6 months.
Pellise M, Desomer L, Burgess NG, et al. The influence of clips on scars after EMR: clip artifact. Gastrointest Endosc. 2016;83:608–16.
Desomer L, Tutticci N, Tate DJ, et al. A standardized imaging protocol is accurate in detecting recurrence after EMR. Gastrointest Endosc. 2016. doi:10.1016/j.gie.2016.06.031.
Knabe M, Pohl J, Gerges C, et al. Standardized long-term follow-up after endoscopic resection of large, nonpedunculated colorectal lesions: a prospective two-center study. Am J Gastroenterol. 2014;109:183–9.
Fahrtash-Bahin F, Holt BA, Jayasekeran V, et al. Snare tip soft coagulation achieves effective and safe endoscopic hemostasis during wide-field endoscopic resection of large colonic lesions (with videos). Gastrointest Endosc. 2013;78:158–63. e1
Moss A, Bourke MJ, Pathmanathan N. Safety of colonic tattoo with sterile carbon particle suspension: a proposed guideline with illustrative cases. Gastrointest Endosc. 2011;74:214–8.
Pohl H, Srivastava A, Bensen SP, et al. Incomplete polyp resection during colonoscopy-results of the complete adenoma resection (CARE) study. Gastroenterology. 2013;144:74–80. e1
Holt BA, Bourke MJ. Wide field endoscopic resection for advanced colonic mucosal neoplasia: current status and future directions. Clin Gastroenterol Hepatol. 2012;10:969–79.
Fujiya M, Tanaka K, Dokoshi T, et al. Efficacy and adverse events of EMR and endoscopic submucosal dissection for the treatment of colon neoplasms: a meta-analysis of studies comparing EMR and endoscopic submucosal dissection. Gastrointest Endosc. 2015;81:583–95.
Brooker JC, Saunders BP, Shah SG, et al. Treatment with argon plasma coagulation reduces recurrence after piecemeal resection of large sessile colonic polyps: a randomized trial and recommendations. Gastrointest Endosc. 2002;55:371–5.
Bahin FF, Pellise M, Williams SJ, et al. Extended endoscopic mucosal resection does not reduce recurrence compared with standard endoscopic mucosal resection of large laterally spreading colorectal lesions. Gastrointest Endosc 2016;84(6:)997–1006. e1
Klein A, Jayasekeran V, Hourigan L, et al. A multi-center randomized trial of thermal ablation of the margin of the post endoscopic mucosal resection (EMR) mucosal defect in the prevention of adenoma recurrence following EMR: preliminary results from the “SCAR” study. Gastroenterology. 2016;150:S1266.
Burgess NG, Metz AJ, Williams SJ, et al. Risk factors for intraprocedural and clinically significant delayed bleeding after wide-field endoscopic mucosal resection of large colonic lesions. Clin Gastroenterol Hepatol. 2014;12:651–61. e1-3
Burgess NG, Williams SJ, Hourigan LF, et al. A management algorithm based on delayed bleeding after wide-field endoscopic mucosal resection of large colonic lesions. Clin Gastroenterol Hepatol. 2014;12(9):1525–33.
Albeniz E, Fraile M, Ibanez B, et al. A scoring system to determine risk of delayed bleeding after endoscopic mucosal resection of large colorectal lesions. Clin Gastroenterol Hepatol. 2016;14:1140–7.
Liaquat H, Rohn E, Rex DK. Prophylactic clip closure reduced the risk of delayed postpolypectomy hemorrhage: experience in 277 clipped large sessile or flat colorectal lesions and 247 control lesions. Gastrointest Endosc. 2013;77:401–7.
Metz AJ, Bourke MJ, Moss A, et al. Factors that predict bleeding following endoscopic mucosal resection of large colonic lesions. Endoscopy. 2011;43:506–11.
Park CH, Jung YS, Nam E, et al. Comparison of efficacy of prophylactic endoscopic therapies for postpolypectomy bleeding in the colorectum: a systematic review and network meta-analysis. Am J Gastroenterol. 2016;111:1230–43.
Bahin FF, Naidoo M, Williams SJ, et al. Prophylactic endoscopic coagulation to prevent bleeding after wide-field endoscopic mucosal resection of large sessile colon polyps. Clin Gastroenterol Hepatol. 2015;13:724–30. e1-2
Boumitri C, Mir FA, Ashraf I, et al. Prophylactic clipping and post-polypectomy bleeding: a meta-analysis and systematic review. Ann Gastroenterol. 2016;29:502–8.
Bahin FF, Rasouli KN, Williams SJ, et al. Prophylactic clipping for the prevention of bleeding following wide-field endoscopic mucosal resection of laterally spreading colorectal lesions: an economic modeling study. Endoscopy. 2016;48:754–61.
Raju GS, Saito Y, Matsuda T, et al. Endoscopic management of colonoscopic perforations (with videos). Gastrointest Endosc. 2011;74:1380–8.
Swan MP, Bourke MJ, Moss A, et al. The target sign: an endoscopic marker for the resection of the muscularis propria and potential perforation during colonic endoscopic mucosal resection. Gastrointest Endosc. 2011;73:79–85.
•• Burgess NG, Bassan MS, McLeod D, et al. Deep mural injury and perforation after colonic endoscopic mucosal resection: a new classification and analysis of risk factors. Gut 2016. doi:10.1136/gutjnl-2015-309848.A novel classification system for deep mural injury following piecemeal EMR is proposed in this study. Meticulous examination of the post-EMR defect permits early identification and proactive management of higher grades of injury with low rates of clinically significant sequelae.
Holt BA, Jayasekeran V, Sonson R, et al. Topical submucosal chromoendoscopy defines the level of resection in colonic EMR and may improve procedural safety (with video). Gastrointest Endosc. 2013;77:949–53.
Tutticci N, Klein A, Sonson R, et al. Endoscopic resection of subtotal or completely circumferential laterally spreading colonic adenomas: technique, caveats, and outcomes. Endoscopy. 2016;48:465–71.
Nanda KS, Tutticci N, Burgess NG, et al. Endoscopic mucosal resection of laterally spreading lesions involving the ileocecal valve: technique, risk factors for failure, and outcomes. Endoscopy. 2015;47:710–8.
Tate DJ, Desomer L, Hourigan LF, et al. Endoscopic mucosal resection of laterally spreading lesions around or involving the appendiceal orifice (PA LSLs): techniques, risk factors for failure and outcomes of a tertiary referral cohort. Gastrointest Endosc. 2016;83:AB144.
Jacob H, Toyonaga T, Ohara Y, et al. Endoscopic submucosal dissection of cecal lesions in proximity to the appendiceal orifice. Endoscopy. 2016;48:829–36.
Al-Bawardy B, Rajan E, WongKeeSong LM. Over-the-scope clip-assisted endoscopic full-thickness resection of epithelial and subepithelial GI lesions. Gastrointest Endosc 2016.
Schmidt A, Bauerfeind P, Gubler C, et al. Endoscopic full-thickness resection in the colorectum with a novel over-the-scope device: first experience. Endoscopy. 2015;47:719–25.
Pimentel-Nunes P, Dinis-Ribeiro M, Ponchon T, et al. Endoscopic submucosal dissection: European Society of Gastrointestinal Endoscopy (ESGE) guideline. Endoscopy. 2015;47:829–54.
Tanaka S, Kashida H, Saito Y, et al. JGES guidelines for colorectal endoscopic submucosal dissection/endoscopic mucosal resection. Dig Endosc. 2015;27:417–34.
Nakajima T, Saito Y, Tanaka S, et al. Current status of endoscopic resection strategy for large, early colorectal neoplasia in Japan. Surg Endosc. 2013;27:3262–70.
Saito Y, Uraoka T, Yamaguchi Y, et al. A prospective, multicenter study of 1111 colorectal endoscopic submucosal dissections (with video). Gastrointest Endosc. 2010;72:1217–25.
Lee EJ, Lee JB, Lee SH, et al. Endoscopic submucosal dissection for colorectal tumors—1,000 colorectal ESD cases: one specialized institute’s experiences. Surg Endosc. 2013;27:31–9.
Niimi K, Fujishiro M, Kodashima S, et al. Long-term outcomes of endoscopic submucosal dissection for colorectal epithelial neoplasms. Endoscopy. 2010;42:723–9.
Nishiyama H, Isomoto H, Yamaguchi N, et al. Endoscopic submucosal dissection for laterally spreading tumours of the colorectum in 200 consecutive cases. Surg Endosc. 2010;24:2881–7.
Rahmi G, Hotayt B, Chaussade S, et al. Endoscopic submucosal dissection for superficial rectal tumors: prospective evaluation in France. Endoscopy. 2014;46:670–6.
Rolny P. The need for supplementary surgery after endoscopic treatment of colorectal neoplasms: comparing endoscopic mucosal resection and endoscopic submucosal dissection. Gastrointest Endosc. 2016;83:1299.
Lee EY, Bourke MJ. EMR should be the first-line treatment for large laterally spreading colorectal lesions. Gastrointest Endosc. 2016;84:326–8.
Rutter MD, Chattree A, Barbour JA, et al. British Society of Gastroenterology/Association of Coloproctologists of Great Britain and Ireland guidelines for the management of large non-pedunculated colorectal polyps. Gut. 2015;64:1847–73.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Steven J. Heitman, David J. Tate, and Michael J. Bourke declare that they have no conflict of interest.
Human and animal rights and informed consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Colon
Summary box: recommendations for endoscopic mucosal resection (EMR)
Summary box: recommendations for endoscopic mucosal resection (EMR)
Fundamentals of resection:
-
Practitioners of EMR must methodically characterize all lesions and assess for features of submucosal invasion prior to commencing the resection.
-
Submucosal injection is done in a dynamic fashion using a crystalloid/colloid solution containing a dye (indigo carmine or methylene blue) and dilute epinephrine (1:100,000).
-
Snare selection (size, shape, wire thickness) is tailored to the lesion.
-
A systematic inject-and-resect technique is performed. This includes meticulous snare placement and tissue capture, snare closure by the endoscopist with a check for tissue mobility, and finally application of thermal energy controlled by a microprocessor electrosurgical unit.
-
Use of CO2 insufflation throughout the procedure is mandatory.
Prevention of recurrence:
-
All visible adenoma should be resected using a snare.
-
The resection margin should include a 1–2 mm circumferential rim of endoscopically normal tissue.
-
Following removal of all endoscopically apparent adenoma, we routinely treat the post-EMR margin with snare tip soft coagulation (ERBE Effect 4, 80 W) to treat invisible potential residual disease in an adjuvant setting.
Managing intra-procedural bleeding and addressing deep mural injury:
-
Patients should be positioned such that dependent fluid (including blood) pools away from the resection field.
-
Minor intra-procedural bleeding should be treated with snare tip soft coagulation.
-
Coagulating forceps can be used to treat persistent or significant bleeding and are preferred over endoscopic clips, which can interfere with the resection.
-
The Sydney Classification of Deep Mural Injury categorizes the full spectrum of mural injury and we use it to evaluate all post-EMR defects.
-
Topical application of the chromoinjectate using the injection catheter with the needle retracted can be used to focally interrogate areas of potential concern.
-
In 2017, tissue resection-related perforation can be reliably treated endoscopically with clip closure in >95% of cases. The key aspect is to recognize it during the procedure.
Follow-up:
-
Surveillance should occur at 4–6 months and 18 months post EMR.
-
Endoscopists must carefully and systematically evaluate all post-EMR scars using high-definition white light and virtual chromoendoscopy.
-
For endoscopically evident recurrence, we attempt to resect it using a stiff thin-wired snare. When snare excision is not feasible, we employ the technique of cold avulsion using standard biopsy forceps followed by snare tip soft coagulation.
-
Any inconclusive nodularity should be biopsied and then destroyed.
-
At present, routine biopsy of the post-EMR scar during surveillance is standard of care, but our recent evidence suggests that it may not be necessary and can be better targeted using high definition endoscopic imaging.
Rights and permissions
About this article
Cite this article
Heitman, S.J., Tate, D.J. & Bourke, M.J. Optimizing Resection of Large Colorectal Polyps. Curr Treat Options Gastro 15, 213–229 (2017). https://doi.org/10.1007/s11938-017-0131-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11938-017-0131-5