Abstract
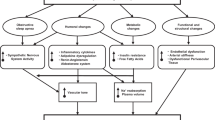
Obesity-related hypertension is commonly characterized by increased sympathetic nerve activity and is therefore acknowledged as a predominantly neurogenic form of hypertension. The sustained sympatho-excitation not only contributes to the rise in blood pressure but also elicits a vicious cycle which facilitates further weight gain and progression of associated co-morbidities. While weight loss and exercise remain at the forefront of therapy for obesity and obesity-related hypertension, the difficulties in achieving and maintaining long-term weight loss with lifestyle measures and the variable blood pressure response to weight loss often necessitate prescription of antihypertensive drug therapy. Remarkably, there are no specific recommendations for pharmacologic treatment for obese patients with arterial hypertension in any of the current guidelines and general principles of antihypertensive treatment are applied. The use of β-blockers and diuretics is commonly discouraged as first- or second-line therapy due to their unfavorable metabolic effects. This review explores evolving therapeutic strategies which based on their interference with pathophysiologic mechanism relevant in the context of obesity may guide optimized treatment of obesity-related hypertension.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Alwan A. Global status report on non-communicable diseases 2010. Geneva: World Health Organization; 2011.
WHO. Obesity—preventing and managing the global epidemic. WHO consultation on obesity, Geneva, 3–5 June 1997. Geneva: WHO; 1997.
Must A, et al. The disease burden associated with overweight and obesity. JAMA. 1999;282:1523–9.
Wilson PW, et al. Overweight and obesity as determinants of cardiovascular risk: the Framingham experience. Arch Intern Med. 2002;162:1867–72.
Doll S, et al. Body mass index, abdominal adiposity and blood pressure: consistency of their association across developing and developed countries. Int J Obes. 2002;26:48–57.
Droyvold WB, et al. Change in body mass index and its impact on blood pressure: a prospective population study. Int J Obes. 2005;29:650–5.
Neter JE, et al. Influence of weight reduction on blood pressure: a meta-analysis of randomized controlled trials. Hypertension. 2003;42:878–84.
Appel LJ, et al. Dietary approaches to prevent and treat hypertension: a scientific statement from the American Heart Association. Hypertension. 2006;47:296–308.
Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med. 1998;15:539–53.
Alberti KG, et al. Harmonizing the metabolic syndrome. a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–5.
Schlaich M, et al. Metabolic syndrome: a sympathetic disease? Lancet Diabetes Endocrinol. 2015;3:148–57.
Straznicky NE, et al. Effects of dietary weight loss on sympathetic activity and cardiac risk factors associated with the metabolic syndrome. J Clin Endocrinol Metab. 2005;90:5998–6005.
Stelfox HT, et al. Hemodynamic monitoring in obese patients: the impact of body mass index on cardiac output and stroke volume. Crit Care Med. 2006;34:1243–6.
Strazzullo P, et al. Abnormalities of renal sodium handling in the metabolic syndrome. Results of the Olivetti Heart Study. J Hypertens. 2006;24:1633–9.
Rumantir MS, et al. Neural mechanisms in human obesity-related hypertension. J Hypertens. 1999;17:1125–33.
Grassi G, et al. Adrenergic and reflex abnormalities in obesity-related hypertension. Hypertension. 2000;36:538–42.
Narkiewicz K, et al. Sympathetic activity in obese subjects with and without obstructive sleep apnea. Circulation. 1998;98:772–6.
Wofford MR, et al. Antihypertensive effect of alpha- and beta-adrenergic blockade in obese and lean hypertensive subjects. Am J Hypertens. 2001;14:694–8.
Shibao C, et al. Autonomic contribution to blood pressure and metabolism in obesity. Hypertension. 2007;49:27–33.
Lambert E, et al. Differing pattern of sympathoexcitation in normal-weight and obesity-related hypertension. Hypertension. 2007;50:862–8.
WHO. Obesity and overweight: fact sheet number 311. 2011 [updated 2011; cited]; Available from: http://www.who.int/mediacentre/factsheets/fs311/en/index.html. Accessed 8 Nov 2009.
Frohlich ED. Clinical management of the obese hypertensive patient. Cardiol Rev. 2002;10:127–38.
Hall JE. Pathophysiology of obesity hypertension. Curr Hypertens Rep. 2000;2:139–47.
Hall JE, et al. Mechanisms of obesity-associated cardiovascular and renal disease. Am J Med Sci. 2002;324:127–37.
Wofford MR, Hall JE. Pathophysiology and treatment of obesity hypertension. Curr Pharm Des. 2004;10:3621–37.
Victor RG, Shafiq MM. Sympathetic neural mechanisms in human hypertension. Curr Hypertens Rep. 2008;10:241–7.
Sarzani R, et al. Renin-angiotensin system, natriuretic peptides, obesity, metabolic syndrome, and hypertension: an integrated view in humans. J Hypertens. 2008;26:831–43.
Bloomgarden ZT. Obesity, hypertension, and insulin resistance. Diabetes Care. 2002;25:2088–97.
Re RN. The clinical implication of tissue renin angiotensin systems. Curr Opin Cardiol. 2001;16:317–27.
Greenfield JR, et al. Modulation of blood pressure by central melanocortinergic pathways. N Engl J Med. 2009;360:44–52.
Dzau VJ, Re R. Tissue angiotensin system in cardiovascular medicine: a paradigm shift? Circulation. 1994;89:493–8.
Engeli S. Dysregulation of the endocannabinoid system in obesity. J Neuroendocrinol. 2008;20:110–5.
Sarzani R. Endocannabinoids, blood pressure and the human heart. J Neuroendocrinol. 2008;20:58–62.
Grassi G, et al. Blood pressure lowering effects of rimonabant in obesity-related hypertension. J Neuroendocrinol. 2008;20:63–8.
• Straznicky NE, et al. The effects of weight loss versus weight loss maintenance on sympathetic nervous system activity and metabolic syndrome components. J Clin Endocrinol Metab. 2011;96:E503–8. A prospective lifestyle intervention trial that identified divergent effects of successful weight loss maintenance on whole-body norepinephrine spillover rate and MSNA suggesting organ-specific differentiation in SNS adaptation to weight loss under conditions of negative vs. stable energy balance.
Uretsky S, et al. Obesity paradox in patients with hypertension and coronary artery disease. Am J Med. 2007;120:863–70.
Dentali F, et al. Management of hypertension in overweight and obese patients: a practical guide for clinicians. Curr Hypertens Rep. 2005;7:330–6.
Schmieder RE, et al. Obesity as a determinant for response to antihypertensive treatment. Br Med J. 1993;307:537–40.
Gress TW, et al. Hypertension and antihypertensive therapy as risk factors for type 2 diabetes mellitus. Atherosclerosis Risk in Communities Study. N Engl J Med. 2000;342:905–12.
Sharma AM, et al. Hypothesis: beta-adrenergic receptor blockers and weight gain: a systematic analysis. Hypertension. 2001;37:250–4.
Jordan J, et al. Hemodynamic and metabolic responses to valsartan and atenolol in obese hypertensive patients. J Hypertens. 2005;23:2313–8.
Jordan J, et al. Antihypertensive treatment in patients with class 3 obesity. Ther Adv Endocrinol Metab. 2012;3:93–8.
Chazova I, et al. Moxonidine improves glycaemic control in mildly hypertensive, overweight patients: a comparison with metformin. Diabetes Obes Metab. 2006;8:456–65.
Haenni A, Lithell H. Moxonidine improves insulin sensitivity in insulin-resistant hypertensives. J Hypertens. 1999;17:S29–35.
Strojek K, et al. Lowering of microalbuminuria in diabetic patients by a sympathicoplegic agent: novel approach to prevent progression of diabetic nephropathy? J Am Soc Nephrol. 2001;12:602–5.
Vonend O, et al. Moxonidine treatment of hypertensive patients with advanced renal failure. J Hypertens. 2003;21:1709–17.
Prichard BN, et al. Moxonidine: a new antiadrenergic antihypertensive agent. J Hypertens. 1999;17:S41–54.
Topal E, et al. The effect of moxonidine on endothelial dysfunction in metabolic syndrome. Am J Cardiovasc Drugs. 2006;6:343–8.
• Matthews VB, et al. Role of the sympathetic nervous system in regulation of the sodium glucose cotransporter 2. J Hypertens. 2017;35:2059–68. This is the first study to identify the importance of SNS-SGLT2 cross talk that accounts for SNS-induced alterations in glucose metabolism and SGLT2 inhibition with dapagliflozin resulted in cardiovascular and renal protection.
• Zinman B, et al. EMPA-REG OUTCOME investigators. 27 version 2, dated 25 June 2016 Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117–28. A randomized control trial where type 2 diabetics at high risk for cardiovascular events received empagliflozin along with standard care had a lower rate of the primary composite cardiovascular outcome and death as compared with placebo.
• Chazova I, Schlaich MP. Improved hypertension control with the imidazoline agonist moxonidine in a multinational metabolic syndrome population: principal results of the MERSY study. Int J Hypertens. 2013; https://doi.org/10.1155/2013/541689. A large, multinational study that appraised the effects of moxonidine on blood pressure, anthropometric, lipid, and metabolic parameters of the metabolic syndrome in routine clinical practice.
Schlaich MP, et al. European Society of Hypertension Working Group on Obesity; Australian and New Zealand Obesity Society. European Society of Hypertension Working Group on Obesity. Obesity-induced hypertension and target organ damage: current knowledge and future directions. J Hypertens. 2009;27:207–11.
Sanjuliani AF, et al. Selective imidazoline agonist moxonidine in obese hypertensive patients. Int J ClinPract. 2006;60:621–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Schlaich is supported by an NHMRC Research Fellowship and has received consulting fees, and/or research support from Abbott. Dr. Schultz reports grants from Abbott vascular, personal fees from Abbot vascular, outside the submitted work. None of the other authors declare any conflict of interest relevant to this manuscript.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Antihypertensive Agents: Mechanisms of Drug Action
Rights and permissions
About this article
Cite this article
Carnagarin, R., Gregory, C., Azzam, O. et al. The Role of Sympatho-Inhibition in Combination Treatment of Obesity-Related Hypertension. Curr Hypertens Rep 19, 99 (2017). https://doi.org/10.1007/s11906-017-0795-1
Published:
DOI: https://doi.org/10.1007/s11906-017-0795-1