Abstract
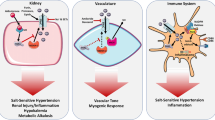
Collectrin (Tmem27) is a transmembrane glycoprotein that is highly expressed in the kidney and vascular endothelium. It is a homologue of the angiotensin-converting enzyme 2 (ACE2) but harbors no catalytic domain. In the extravascular tissues of the kidney, collectrin is localized to the proximal tubule and collecting duct. Collectrin-deficient mice are featured with hypertension and exaggerated salt sensitivity. These phenotypes are associated with impaired uptake of the nitric oxide precursor l-arginine and the expression of its amino acid transporters, CAT-1 and y+LAT1, in endothelial cells. In addition, collectrin-deficient mice display decreased dimerization of nitric oxide synthase and decreased nitric oxide synthesis, but enhanced superoxide generation, suggesting that deletion of collectrin leads to a state of nitric oxide synthase uncoupling. These findings suggest that collectrin plays a protective role against hypertension. The collectrin knockout mouse represents a unique model for hypertension research. Furthermore, collectrin may serve as a novel therapeutic target in the treatment of hypertension.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Chow CK, Teo KK, Rangarajan S, Islam S, Gupta R, Avezum A, et al. Prevalence, awareness, treatment, and control of hypertension in rural and urban communities in high-, middle-, and low-income countries. JAMA. 2013;310(9):959–68. doi:10.1001/jama.2013.184182.
Zhang H, Wada J, Hida K, Tsuchiyama Y, Hiragushi K, Shikata K, et al. Collectrin, a collecting duct-specific transmembrane glycoprotein, is a novel homolog of ACE2 and is developmentally regulated in embryonic kidneys. J Biol Chem. 2001;276(20):17132–9. doi:10.1074/jbc.M006723200.
Mount DB. Collectrin and the kidney. Curr Opin Nephrol Hypertens. 2007;16(5):427–9. doi:10.1097/MNH.0b013e3282e9acc5.
Yasuhara A, Wada J, Malakauskas SM, Zhang Y, Eguchi J, Nakatsuka A, et al. Collectrin is involved in the development of salt-sensitive hypertension by facilitating the membrane trafficking of apical membrane proteins via interaction with soluble N-ethylmaleiamide-sensitive factor attachment protein receptor complex. Circulation. 2008;118(21):2146–55. doi:10.1161/CIRCULATIONAHA.108.787259.
Zhang H, Wada J, Kanwar YS, Tsuchiyama Y, Hiragushi K, Hida K, et al. Screening for genes up-regulated in 5/6 nephrectomized mouse kidney. Kidney Int. 1999;56(2):549–58. doi:10.1046/j.1523-1755.1999.00561.x.
Malakauskas SM, Quan H, Fields TA, McCall SJ, Yu MJ, Kourany WM, et al. Aminoaciduria and altered renal expression of luminal amino acid transporters in mice lacking novel gene collectrin. Am J Physiol Renal Physiol. 2007;292(2):F533–44. doi:10.1152/ajprenal.00325.2006.
McCoy KE, Zhou X, Vize PD. Collectrin/tmem27 is expressed at high levels in all segments of the developing Xenopus pronephric nephron and in the Wolffian duct. Gene Expr Patterns : GEP. 2008;8(4):271–4. doi:10.1016/j.gep.2007.12.002.
Fukui K, Yang Q, Cao Y, Takahashi N, Hatakeyama H, Wang H, et al. The HNF-1 target collectrin controls insulin exocytosis by SNARE complex formation. Cell Metab. 2005;2(6):373–84. doi:10.1016/j.cmet.2005.11.003.
Akpinar P, Kuwajima S, Krutzfeldt J, Stoffel M. Tmem27: a cleaved and shed plasma membrane protein that stimulates pancreatic beta cell proliferation. Cell Metab. 2005;2(6):385–97. doi:10.1016/j.cmet.2005.11.001.
Danilczyk U, Sarao R, Remy C, Benabbas C, Stange G, Richter A, et al. Essential role for collectrin in renal amino acid transport. Nature. 2006;444(7122):1088–91. doi:10.1038/nature05475.
Cechova S, Zeng Q, Billaud M, Mutchler S, Rudy CK, Straub AC, et al. Loss of collectrin, an angiotensin-converting enzyme 2 homolog, uncouples endothelial nitric oxide synthase and causes hypertension and vascular dysfunction. Circulation. 2013;128(16):1770–80. doi:10.1161/CIRCULATIONAHA.113.003301. This study established the role of collectrin in blood pressure regulation. Furthermore, the study links amino acid transport defect with “L-arginine paradox,” endothelial dysfunction, and hypertension.
Yamagata K, Oda N, Kaisaki PJ, Menzel S, Furuta H, Vaxillaire M, et al. Mutations in the hepatocyte nuclear factor-1alpha gene in maturity-onset diabetes of the young (MODY3). Nature. 1996;384(6608):455–8. doi:10.1038/384455a0.
Shih DQ, Stoffel M. Molecular etiologies of MODY and other early-onset forms of diabetes. Curr Diabetes Rep. 2002;2(2):125–34.
Pontoglio M, Barra J, Hadchouel M, Doyen A, Kress C, Bach JP, et al. Hepatocyte nuclear factor 1 inactivation results in hepatic dysfunction, phenylketonuria, and renal Fanconi syndrome. Cell. 1996;84(4):575–85.
Esterhazy D, Stutzer I, Wang H, Rechsteiner MP, Beauchamp J, Dobeli H, et al. Bace2 is a beta cell-enriched protease that regulates pancreatic beta cell function and mass. Cell Metab. 2011;14(3):365–77. doi:10.1016/j.cmet.2011.06.018. This report identified the beta-site amyloid precursor protein cleaving enzyme 2 (Bace2) as the sheddase of collectrin, suggesting that Bace2 may be a therapeutic target in conditions in which increased expression of collectrin may be desirable for potential beneficial effects such as salt-sensitive hypertension.
Bennett BD, Babu-Khan S, Loeloff R, Louis JC, Curran E, Citron M, et al. Expression analysis of BACE2 in brain and peripheral tissues. J Biol Chem. 2000;275(27):20647–51. doi:10.1074/jbc.M002688200.
Gurley SB, Allred A, Le TH, Griffiths R, Mao L, Philip N, et al. Altered blood pressure responses and normal cardiac phenotype in ACE2-null mice. J Clin Invest. 2006;116(8):2218–25. doi:10.1172/JCI16980.
Hartner A, Cordasic N, Klanke B, Veelken R, Hilgers KF. Strain differences in the development of hypertension and glomerular lesions induced by deoxycorticosterone acetate salt in mice. Nephrol Dial Transplant. 2003;18(10):1999–2004. doi:10.1093/ndt/gfg299.
Salzler HR, Griffiths R, Ruiz P, Chi L, Frey C, Marchuk DA, et al. Hypertension and albuminuria in chronic kidney disease mapped to a mouse chromosome 11 locus. Kidney Int. 2007;72(10):1226–32.
Francois H, Athirakul K, Howell D, Dash R, Mao L, Kim HS, et al. Prostacyclin protects against elevated blood pressure and cardiac fibrosis. Cell Metab. 2005;2(3):201–7. doi:10.1016/j.cmet.2005.08.005.
Ryan MJ, Didion SP, Davis DR, Faraci FM, Sigmund CD. Endothelial dysfunction and blood pressure variability in selected inbred mouse strains. Arterioscler Thromb Vasc Biol. 2002;22(1):42–8.
Wang Q, Hummler E, Nussberger J, Clement S, Gabbiani G, Brunner HR, et al. Blood pressure, cardiac, and renal responses to salt and deoxycorticosterone acetate in mice: role of Renin genes. J Am Soc Nephrol. 2002;13(6):1509–16.
Knowles RG, Moncada S. Nitric oxide synthases in mammals. Biochem J. 1994;298(Pt 2):249–58.
Takimoto E, Champion HC, Li M, Ren S, Rodriguez ER, Tavazzi B, et al. Oxidant stress from nitric oxide synthase-3 uncoupling stimulates cardiac pathologic remodeling from chronic pressure load. J Clin Invest. 2005;115(5):1221–31.
Balligand JL, Feron O, Dessy C. eNOS activation by physical forces: from short-term regulation of contraction to chronic remodeling of cardiovascular tissues. Physiol Rev. 2009;89(2):481–534. doi:10.1152/physrev.00042.2007.
Verhaar MC, Westerweel PE, van Zonneveld AJ, Rabelink TJ. Free radical production by dysfunctional eNOS. Heart. 2004;90(5):494–5.
Closs EI, Scheld JS, Sharafi M, Forstermann U. Substrate supply for nitric-oxide synthase in macrophages and endothelial cells: role of cationic amino acid transporters. Mol Pharmacol. 2000;57(1):68–74.
Hecker M, Sessa WC, Harris HJ, Anggard EE, Vane JR. The metabolism of L-arginine and its significance for the biosynthesis of endothelium-derived relaxing factor: cultured endothelial cells recycle L-citrulline to L-arginine. Proc Natl Acad Sci U S A. 1990;87(21):8612–6.
Simon A, Plies L, Habermeier A, Martine U, Reining M, Closs EI. Role of neutral amino acid transport and protein breakdown for substrate supply of nitric oxide synthase in human endothelial cells. Circ Res. 2003;93(9):813–20. doi:10.1161/01.res.0000097761.19223.0d.
Arancibia-Garavilla Y, Toledo F, Casanello P, Sobrevia L. Nitric oxide synthesis requires activity of the cationic and neutral amino acid transport system y+L in human umbilical vein endothelium. Exp Physiol. 2003;88(6):699–710.
Kakoki M, Kim HS, Arendshorst WJ, Mattson DL. L-Arginine uptake affects nitric oxide production and blood flow in the renal medulla. Am J Physiol Regul Integr Comp Physiol. 2004;287(6):R1478–85. doi:10.1152/ajpregu.00386.2004.
Kakoki M, Kim HS, Edgell CJ, Maeda N, Smithies O, Mattson DL. Amino acids as modulators of endothelium-derived nitric oxide. Am J Physiol Renal Physiol. 2006;291(2):F297–304. doi:10.1152/ajprenal.00417.2005.
Rajapakse NW, Mattson DL. Role of L-arginine in nitric oxide production in health and hypertension. Clin Exp Pharmacol Physiol. 2009;36(3):249–55. doi:10.1111/j.1440-1681.2008.05123.x.
Yang Z, Kaye DM. Endothelial dysfunction and impaired L-arginine transport in hypertension and genetically predisposed normotensive subjects. Trends Cardiovasc Med. 2006;16(4):118–24. doi:10.1016/j.tcm.2006.02.003.
Schlaich MP, Parnell MM, Ahlers BA, Finch S, Marshall T, Zhang WZ, et al. Impaired L-arginine transport and endothelial function in hypertensive and genetically predisposed normotensive subjects. Circulation. 2004;110(24):3680–6. doi:10.1161/01.CIR.0000149748.79945.52.
Chen PY, Sanders PW. Role of nitric oxide synthesis in salt-sensitive hypertension in Dahl/Rapp rats. Hypertension. 1993;22(6):812–8.
Siani A, Pagano E, Iacone R, Iacoviello L, Scopacasa F, Strazzullo P. Blood pressure and metabolic changes during dietary L-arginine supplementation in humans. Am J Hypertens. 2000;13(5 Pt 1):547–51.
Pallone TL, Mattson DL. Role of nitric oxide in regulation of the renal medulla in normal and hypertensive kidneys. Curr Opin Nephrol Hypertens. 2002;11(1):93–8.
Dye JF, Vause S, Johnston T, Clark P, Firth JA, D‘Souza SW, et al. Characterization of cationic amino acid transporters and expression of endothelial nitric oxide synthase in human placental microvascular endothelial cells. FASEB J. 2004;18(1):125–7. doi:10.1096/fj.02-0916fje.
Greene B, Pacitti AJ, Souba WW. Characterization of L-arginine transport by pulmonary artery endothelial cells. Am J Physiol. 1993;264(4 Pt 1):L351–6.
Kilberg MS, Stevens BR, Novak DA. Recent advances in mammalian amino acid transport. Annu Rev Nutr. 1993;13:137–65. doi:10.1146/annurev.nu.13.070193.001033.
Verrey F, Jack DL, Paulsen IT, Saier Jr MH, Pfeiffer R. New glycoprotein-associated amino acid transporters. J Membr Biol. 1999;172(3):181–92.
Kakoki M, Wang W, Mattson DL. Cationic amino acid transport in the renal medulla and blood pressure regulation. Hypertension. 2002;39(2):287–92.
Mattson DL, Meister CJ. Renal cortical and medullary blood flow responses to L-NAME and ANG II in wild-type, nNOS null mutant, and eNOS null mutant mice. Am J Physiol Regul Integr Comp Physiol. 2005;289(4):R991–7. doi:10.1152/ajpregu.00207.2005.
Verrey F, Ristic Z, Romeo E, Ramadan T, Makrides V, Dave MH, et al. Novel renal amino acid transporters. Annu Rev Physiol. 2005;67:557–72.
Broer S. Apical transporters for neutral amino acids: physiology and pathophysiology. Physiology. 2008;23:95–103.
Dall‘Asta V, Bussolati O, Sala R, Rotoli BM, Sebastio G, Sperandeo MP, et al. Arginine transport through system y(+)L in cultured human fibroblasts: normal phenotype of cells from LPI subjects. Am J Physiol Cell Physiol. 2000;279(6):C1829–37.
Deves R, Boyd CA. Transporters for cationic amino acids in animal cells, discovery, structure, and function. Physiol Rev. 1998;78:487–545.
Kamada Y, Nagaretani H, Tamura S, Ohama T, Maruyama T, Hiraoka H, et al. Vascular endothelial dysfunction resulting from l-arginine deficiency in a patient with lysinuric protein intolerance. J Clin Invest. 2001;108(5):717–24.
Yang Z, Venardos K, Jones E, Morris BJ, Chin-Dusting J, Kaye DM. Identification of a novel polymorphism in the 3′UTR of the L-arginine transporter gene SLC7A1: contribution to hypertension and endothelial dysfunction. Circulation. 2007;115(10):1269–74. doi:10.1161/CIRCULATIONAHA.106.665836.
Schnitzer JE, Liu J, Oh P. Endothelial caveolae have the molecular transport machinery for vesicle budding, docking, and fusion including VAMP, NSF, SNAP, annexins, and GTPases. J Biol Chem. 1995;270(24):14399–404.
Schneider MP, Ge Y, Pollock DM, Pollock JS, Kohan DE. Collecting duct-derived endothelin regulates arterial pressure and Na excretion via nitric oxide. Hypertension. 2008;51(6):1605–10. doi:10.1161/hypertensionaha.107.108126.
Hyndman KA, Boesen EI, Elmarakby AA, Brands MW, Huang P, Kohan DE, et al. Renal collecting duct NOS1 maintains fluid-electrolyte homeostasis and blood pressure. Hypertension. 2013;62(1):91–8. doi:10.1161/HYPERTENSIONAHA.113.01291.
Yagil C, Sapojnikov M, Kreutz R, Zurcher H, Ganten D, Yagil Y. Role of chromosome X in the Sabra rat model of salt-sensitive hypertension. Hypertension. 1999;33(1):261–5.
Altirriba J, Gasa R, Casas S, Ramirez-Bajo MJ, Ros S, Gutierrez-Dalmau A, et al. The role of transmembrane protein 27 (TMEM27) in islet physiology and its potential use as a beta cell mass biomarker. Diabetologia. 2010;53(7):1406–14. doi:10.1007/s00125-010-1728-6.
Esterhazy D, Akpinar P, Stoffel M. Tmem27 dimerization, deglycosylation, plasma membrane depletion, and the extracellular Phe-Phe motif are negative regulators of cleavage by Bace2. Biol Chem. 2012;393(6):473–84. doi:10.1515/hsz-2012-0104. This study identified sites of collectrin protein that determine its susceptibility to cleavage by Bace2 and may have implication for the manipulation of the stability of collectrin in certain disease states.
Acknowledgment
This work was supported by NIH/NIDDK T32 DK072922 Training Grant, Division of Nephrology, University of Virginia to P.L.C.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Pei-Lun Chu and Thu H. Le declare no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Hypertension and the Kidney
Rights and permissions
About this article
Cite this article
Chu, PL., Le, T.H. Role of Collectrin, an ACE2 Homologue, in Blood Pressure Homeostasis. Curr Hypertens Rep 16, 490 (2014). https://doi.org/10.1007/s11906-014-0490-4
Published:
DOI: https://doi.org/10.1007/s11906-014-0490-4