Abstract
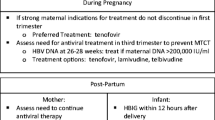
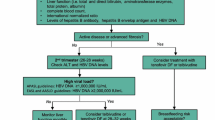
Hepatitis B remains a global health issue, despite an effective vaccine. Mother to child transmission (MTCT) of hepatitis B has become increasingly recognized, especially in those patients with high maternal viremia. In a woman of childbearing age, carefully assessing disease status and urgency of treatment is key, along with discussion of family planning, timing and choice of therapy. In a pregnant patient with chronic hepatitis B, treatment in the third trimester may be considered in those with HBV DNA levels >107 copies (2 × 106 IU/ml) and may reduce the perinatal transmission rate, but a thorough discussion of the risks and benefits is essential. Duration of antiviral therapy depends upon the indication for treatment as those individuals treated for active disease from the hepatitis B would need long term therapy, while those treated in the third trimester only to prevent MTCT may have therapy discontinued.



Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
World Health Organization: Hepatitis B. World Health Organization fact sheet 204 (revised Oct. 2000). Available at: http://who.int/inf-fs/en/fact204.html.
Kowdley KV, Wang CC, Welch S, et al. Prevalence of chronic hepatitis B among foreign born persons living in the United States by country of origin. Hepatology. 2012;56(2):422–33.
Stevens CE, Beasley RP, Tsui J, et al. Vertical transmission of hepatitis B antigen in Taiwan. N Engl J Med. 1975;292:771–4.
Lee SD, Lo KJ, Tsai YT, et al. Maternal hepatitis B virus DNA in mother-infant transmission. Lancet. 1989;1:719.
Ngui SL, O’Connell S, Eglin RP, et al. Low detection rate and maternal provenance of hepatitis B virus S gene mutants in cases of failed postnatal immunoprophylaxis in England and Wales. J Infect Dis. 1997;176:1360–5.
Karthigesu VD, Allison LM, Ferguson M, et al. A hepatitis B virus variant found in the sera of immunised children induces a conformational change in the HBsAg “a” determinant. J Med Virol. 1999;58:346–52.
Lin HH, Lee TY, Chen DS, et al. Transplacental leakage of HBeAg-positive maternal blood as the most likely route in causing intrauterine infection with hepatitis B virus. J Pediatr. 1987;111:877–81.
Lazizi Y, Badur S, Perk Y. Selective unresponsiveness to HBsAg vaccine in newborns related with an in utero passage of hepatitis B virus DNA. Vaccine. 1997;15:1095–100.
Xu DZ, Yan YP, Choi BC, et al. Risk factors and mechanism of transplacental transmission of hepatitis B virus: a case-control study. J Med Virol. 2002;67:20–6.
Towers CV, Asrat T, Rumney P. The presence of hepatitis B surface antigen and deoxyribonucleic acid in amniotic fluid and cord blood. Am J Obstet Gynecol. 2001;184:1514–8.
Burk RD, Hwang LY, Ho GY, et al. Outcome of perinatal hepatitis B virus exposure is dependent on maternal virus load. J Infect Dis. 1994;170:1418–23.
•• Wiseman E, Fraser MA, Holden S, et al. Perinatal transmission of hepatitis B virus: an Australian experience. MJA. 2009;109:489–92. Large, well designed study in Australia confirming transmission rates of HBV in mothers with high viremia, despite adequate vaccination.
Zou H, Chen Y, Duan Z, et al. A retrospective study for clinical outcome of caesarean section on perinatal transmission of hepatitis B virus in infants born to HBeAg positive mothers with chronic hepatitis. Hepatology. 2010;52:235A.
Lee SD, Lo KJ, Tsai YT, et al. Role of caesarean section in prevention of mother-infant transmission of hepatitis B. Lancet. 1988;2:833–4.
Hill JB, Sheffield JS, Kim MJ, et al. Risk of hepatitis B transmission in breast-fed infants of chronic hepatitis B carriers. Obstet Gynecol. 2002;99:1049–52.
Lok AS, McMahon BJ. Chronic hepatitis B: update 2009. Hepatology. 2009;50:1–36.
•• Xu WM, Cui YT, Wang L, et al. Lamivudine in late pregnancy to prevent perinatal transmission of hepatitis B virus infection: a multicentre, randomized, double-blind, placebo-controlled study. J Viral Hepat. 2009;16:94–103. The only prospective randomized controlled trial of antiviral therapy, but this study conclusion was marred by many lost to follow-up.
Han L, Zhang H-W, Xie J-X, et al. A meta-analysis of lamivudine for interruption of mother-to-child transmission of hepatitis B virus. World J Gastroenterol. 2011;17:4321–33.
•• Han G-R, Cao M-K, Zhao W, et al. A prospective and open label study for the efficacy and safety of telbivudine in pregnancy for the prevention of perinatal transmission of hepatitis B virus infection. J Hepatol. 2011;55:1215–21. A large recent study confirmed risk of perinatal transmission with high maternal viremia and use of telbivudine in this group showing reduced transmission.
•• The Antiretroviral Pregancy Registry Interim Report for 1 January 1989 through 31 July 2011. Available at http://www.apregistry.com/forms/interim_report.pdf. The largest database of exposure to antiviral therapy, although mainly in HIV patients.
ter Borg MJ, Leemans WF, de Man RA, Janssen HLA. Exacerbation of chronic hepatitis B infection after delivery. J Vir Hepat. 2008;15:37–41.
Singhal A, Kanagala R, Jalil S, et al. Chronic HBV with pregnancy: reactivation flare causing fulminant hepatic failure. Annals Hepatol. 2011;10(2):233–6.
Tran TT. Understanding cultural barriers in hepatitis B virus infection. Cleveland Clinic J Med. 2009;76(3):S25–9.
Disclosure
Tran TT is a consultant for Gilead Sciences, BMS, Novartis and receives honoraria from BMS and Gilead.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tran, T.T. Hepatitis B in Pregnancy. Curr Hepatitis Rep 12, 1–6 (2013). https://doi.org/10.1007/s11901-012-0154-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11901-012-0154-2