Abstract
Purpose of Review
The paper aims to discuss the global trends in gastric cancer incidence in relation to important factors involved in the pathogenesis of gastric cancer.
Recent Findings
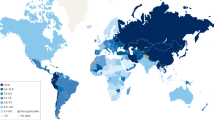
Despite a significant worldwide decline, gastric cancer remains a common cause of cancer death. The decline has been multifactorial and preceded the fall in Helicobacter pylori prevalence. The initial decline was associated with changes in food preservation and availability, especially of fresh fruits and vegetables, followed by a decline in the primary etiologic factor, H. pylori. Gastric cancer incidence remains high in East Asia, intermediate in Latin America, and low in developed countries. Significant racial/ethnic variability exists.
Summary
The rapid decline in incidence in East Asia will continue as primary and secondary prevention strategies are implemented. The incidence in Latin America is unlikely to decline significantly over the next few decades given high H. pylori prevalence in the young. Ultimately, global H. pylori eradication will be needed to largely eliminate gastric cancer.



Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: •Of importance ••Of major importance
•• Graham DY. Helicobacter pylori update: gastric cancer, reliable therapy, and possible benefits. Gastroenterology. 2015;148(4):719–31.e3. doi:10.1053/j.gastro.2015.01.040. Excellent review that discusses the effects of H. pylori infection and challenges, benefits, and potential downsides of its eradication.
•• Sugano K, Tack J, Kuipers EJ, Graham DY, El-Omar EM, Miura S, et al. Kyoto global consensus report on Helicobacter pylori gastritis. Gut. 2015;64(9):1353–67. doi:10.1136/gutjnl-2015-309252. Global consensus on gastritis which discusses classification of chronic gastritis and duodenitis and defines H. pylori infection as an infectious disease that should be cured whenever encountered.
•• Asaka M, Kato M, Sakamoto N. Roadmap to eliminate gastric cancer with Helicobacter pylori eradication and consecutive surveillance in Japan. Journal of gastroenterology. 2014;49(1):1–8. doi:10.1007/s00535-013-0897-8. Excellent review of the Japanese experience in H. pylori eradication as an approach to reducing gastric cancer deaths.
Asaka M. A new approach for elimination of gastric cancer deaths in Japan. Int J Cancer. 2013;132(6):1272–6. doi:10.1002/ijc.27965.
Feldman M.. Sleisenger & Fordtran’s gastrointestinal and liver disease: pathophysiology, diagnosis, management. 9th ed 2014 Philadelphia, PA: Saunders Elsevier; 2010. p. 887–906.
Washington K. 7th edition of the AJCC cancer staging manual: stomach. Ann Surg Oncol. 2010;17(12):3077–9. doi:10.1245/s10434-010-1362-z.
Botterweck AA, Schouten LJ, Volovics A, Dorant E, van Den Brandt PA. Trends in incidence of adenocarcinoma of the oesophagus and gastric cardia in ten European countries. Int J Epidemiol. 2000;29(4):645–54.
• Amini N, Spolverato G, Kim Y, Squires MH, Poultsides GA, Fields R, et al. Clinicopathological features and prognosis of gastric cardia adenocarcinoma: a multi-institutional US study. J Surg Oncol. 2015;111(3):285–92. doi:10.1002/jso.23799. This study compares the clinicopathological characteristics and the prognosis of gastric cardia adenocarcinoma (GCA) versus non-cardia adenocarcinoma. Key findings were that disease-free survival and overall survival were similar between patients with GCA versus non-cardia adenocarcinoma. Long term outcomes were worse with GCA and early stage disease.
Corley DA, Kubo A. Influence of site classification on cancer incidence rates: an analysis of gastric cardia carcinomas. J Natl Cancer Inst. 2004;96(18):1383–7. doi:10.1093/jnci/djh265.
Marques-Lespier JM, Gonzalez-Pons M, Cruz-Correa M. Current perspectives on gastric cancer. Gastroenterol Clin N Am. 2016;45(3):413–28. doi:10.1016/j.gtc.2016.04.002.
Lauren P. The two histological main types of gastric carcinoma: diffuse and so-called intestinal-type carcinoma. An attempt at a histo-clinical classification. Acta pathol Microbiol Scand. 1965;64:31–49.
Schistosomes, liver flukes and Helicobacter pylori. IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Lyon, 7–14 June 1994. IARC monographs on the evaluation of carcinogenic risks to humans. 1994;61:1–241.
Plummer M, Franceschi S, Vignat J, Forman D, de Martel C. Global burden of gastric cancer attributable to Helicobacter pylori. Int J Cancer. 2015;136(2):487–90. doi:10.1002/ijc.28999.
•• Graham DY, Asaka M. Eradication of gastric cancer and more efficient gastric cancer surveillance in Japan: two peas in a pod. J Gastroenterol. 2010;45(1):1–8. doi:10.1007/s00535-009-0117-8. Review of the historical relation of gastric cancer and atrophic gastritis with special emphasis on the role of acid secretion in predicting outcome. Much of this data was unknown to early H. pylori investigators causing significant delay in understanding the correlations.
Rugge M. Gastric cancer risk in patients with Helicobacter pylori infection and following its eradication. Gastroenterol Clin N Am. 2015;44(3):609–24. doi:10.1016/j.gtc.2015.05.009.
Peek RM Jr, Crabtree JE. Helicobacter infection and gastric neoplasia. J Pathol. 2006;208(2):233–48. doi:10.1002/path.1868.
Graham DY. Helicobacter pylori infection is the primary cause of gastric cancer. J Gastroenterol. 2000;35(Suppl 12):90–7.
de Vries AC, Haringsma J, Kuipers EJ. The detection, surveillance and treatment of premalignant gastric lesions related to Helicobacter pylori infection. Helicobacter. 2007;12(1):1–15. doi:10.1111/j.1523-5378.2007.00475.x.
* Fock KM. Review article: the epidemiology and prevention of gastric cancer. Aliment Pharmacol Ther. 2014;40(3):250–60. doi:10.1111/apt.12814. A comprehensive review article that covered literature since early 2000s regarding epidemiology and strategies in prevention of non cardia gastric cancer. It specifically also discusses how H. pylori eradication in high gastric cancer regions can lead to a decline in the incidence of this highly lethal disease.
•• Graham DY. History of Helicobacter pylori, duodenal ulcer, gastric ulcer and gastric cancer. World J Gastroenterol. 2014;20(18):5191–204. doi:10.3748/wjg.v20.i18.5191. Review of the natural history of H. pylori -related diseases, peptic ulcer, and gastric cancer throughout time with special emphasis on the effect of the changes in the patterns of gastritis on disease presentation and prevalence
•• Miftahussurur M, Yamaoka Y, Graham DY. Helicobacter pylori as an oncogenic pathogen, revisited. Expert reviews in molecular medicine. 2017;19:e4. doi:10.1017/erm.2017.4. Review of the role of H. pylori in gastric carcinogenesis particularly in relation to H. pylori virulence factors and inflammation.
Figueiredo C, Machado JC, Pharoah P, Seruca R, Sousa S, Carvalho R, et al. Helicobacter pylori and interleukin 1 genotyping: an opportunity to identify high-risk individuals for gastric carcinoma. J Natl Cancer Inst. 2002;94(22):1680–7.
Whelan SL. International Association of Cancer Registries—a history. Asian Pac J Cancer Prev. 2010;10
Antoni S, Soerjomataram I, Moller B, Bray F, Ferlay J. An assessment of GLOBOCAN methods for deriving national estimates of cancer incidence. Bull World Health Organ. 2016;94(3):174–84. doi:10.2471/blt.15.164384.
•• Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int J cancer. 2015;136(5):E359–E86. doi:10.1002/ijc.29210. Discussion of the worldwide incidence and mortality from 27 major cancers and for all cancers combined for 2012 and gastric cancer trends worldwide.
Ewing J. Carcinoma of the stomach. Neoplastic diseases. Philadelphia: W.B: Saunders Company; 1919. p. 605–39.
Hurst A. Schorstein lecture on the precursors of carcinoma of the stomach. Lancet. 214(5542):1023–8. doi:10.1016/S0140-6736(01)09840-3.
Sullivan PD, Christine B, Connelly R, Barrett H. Analysis of trends in age-adjusted incidence rates for 10 major sites of cancer. Am J Public Health. 1972;62(8):1065–71.
Parkin DM, Stjernsward J, Muir CS. Estimates of the worldwide frequency of twelve major cancers. Bull World Health Organ. 1984;62(2):163–82.
Wynder EL, Kmet J, Dungal N, Segi M. An epidemiological investigation of gastric cancer. Cancer. 1963;16:1461–96.
Doll R. Cancer in five continents. Proc R Soc Med. 1972;65(1):49–55.
• Ferlay J, Soerjomataram I, Ervik M , Dikshit R, Eser S , Mathers C , Rebelo M , Parkin DM , Forman D, Bray F. GLOBOCAN 2012 v1.0, Cancer incidence and mortality worldwide: IARC CancerBase No. 11 [Internet]. Lyon, France: International Agency for Research on Cancer; 2013. Available from: http://globocan.iarc.fr, accessed on 13/03/2017. Available from: http://globocan.iarc.fr, accessed on day/month/year. Accessed March 13 2017. This publication nicely summarizes the worldwide incidence and mortality from 27 major cancers and for all cancers combined up until 2012. Stomach cancer was the 5th most common cancer by incidence with the 3rd highest mortality rate.
• Ferro A, Peleteiro B, Malvezzi M, Bosetti C, Bertuccio P, Levi F, et al. Worldwide trends in gastric cancer mortality (1980–2011), with predictions to 2015, and incidence by subtype. European journal of cancer (Oxford, England: 1990). 2014;50(7):1330–44. doi:10.1016/j.ejca.2014.01.029. This paper documents mortality rates from gastric cancer decreased by 3% per year in Europe, Japan, and Korea and by 2% per year for North America and major Latin countries between 1980 and 2011.
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. doi:10.3322/caac.21262.
•• Lui FH, Tuan B, Swenson SL, Wong RJ. Ethnic disparities in gastric cancer incidence and survival in the USA: an updated analysis of 1992–2009 SEER data. Dig Dis Sci. 2014;59(12):3027–34. doi:10.1007/s10620-014-3275-3. Summary of SEER data from 1992–2009 showing ethnic/racial disparities exist in the USA with regard to gastric cancer incidence and survival.
Karimi P, Islami F, Anandasabapathy S, Freedman ND, Kamangar F. Gastric cancer: descriptive epidemiology, risk factors, screening, and prevention. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2014;23(5):700–13. doi:10.1158/1055-9965.epi-13-1057.
Soerjomataram I, Lortet-Tieulent J, Parkin DM, Ferlay J, Mathers C, Forman D, et al. Global burden of cancer in 2008: a systematic analysis of disability-adjusted life-years in 12 world regions. Lancet. 2012;380(9856):1840–50. doi:10.1016/s0140-6736(12)60919-2.
* Wang J, Sun Y, Bertagnolli MM. Comparison of gastric cancer survival between Caucasian and Asian patients treated in the United States: results from the Surveillance Epidemiology and End Results (SEER) database. Ann Surg Oncol. 2015;22(9):2965–71. doi:10.1245/s10434-015-4388-4. This paper highlights that Asians have better overall survival compared to Caucasians with respect to non cardia gastric cancer. The mean difference in 5 yr survival was 12% (range- 9% for stage IIIc to 31% for stage I). This trend was shown to persist despite adjustment for age, gender, tumor grade, and number of examined and positive lymph nodes.
* Kim GH, Liang PS, Bang SJ, Hwang JH. Screening and surveillance for gastric cancer in the United States: is it needed? Gastrointest Endosc. 2016;84(1):18–28. doi:10.1016/j.gie.2016.02.028. This review article discusses who should be screened for gastric cancer in the United States and how? Key take home points are that individuals who are immigrants to United States from regions associated with a high risk of gastric cancer (East Asia, Russia, or South America) or who have a family history of gastric cancer should be considered for gastric cancer screening. Authors suggested that those with findings of atrophic gastritis or intestinal metaplasia on screening endoscopy should undergo surveillance endoscopy every 1 to 2 years.
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9–29. doi:10.3322/caac.21208.
Steevens J, Botterweck AA, Dirx MJ, van den Brandt PA, Schouten LJ. Trends in incidence of oesophageal and stomach cancer subtypes in Europe. Eur J Gastroenterol Hepatol. 2010;22(6):669–78. doi:10.1097/MEG.0b013e32832ca091.
Tinmouth J, Green J, Ko YJ, Liu Y, Paszat L, Sutradhar R, et al. A population-based analysis of esophageal and gastric cardia adenocarcinomas in Ontario, Canada: incidence, risk factors, and regional variation. Journal of gastrointestinal surgery : official journal of the Society for Surgery of the Alimentary Tract. 2011;15(5):782–90. doi:10.1007/s11605-011-1450-9.
Powell J, McConkey CC. The rising trend in oesophageal adenocarcinoma and gastric cardia. European journal of cancer prevention : the official journal of the European Cancer Prevention Organisation (ECP). 1992;1(3):265–9.
•• Schlansky B, Sonnenberg A. Epidemiology of noncardia gastric adenocarcinoma in the United States. Am J Gastroenterol. 2011;106(11):1978–85. doi:10.1038/ajg.2011.213.0020. Analysis from three databases. The SEER registry was used for incidence, the Healthcare Costs and Utilization Project for hospitalizations, and the Compressed Mortality File for mortality showing that older age, male gender, non-White race, and residence in the Northeast region were associated with increased risk of non-cardia gastric cancer.
Wu H, Rusiecki JA, Zhu K, Potter J, Devesa SS. Stomach carcinoma incidence patterns in the United States by histologic type and anatomic site. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2009;18(7):1945–52. doi:10.1158/1055-9965.epi-09-0250.
Arnold M, Moore SP, Hassler S, Ellison-Loschmann L, Forman D, Bray F. The burden of stomach cancer in indigenous populations: a systematic review and global assessment. Gut. 2014;63(1):64–71. doi:10.1136/gutjnl-2013-305033.
Group USCSW. United States Cancer Statistics. Incidence and mortality web-based report. Atlanta: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention and National Cancer Institute; 1999–2013. www.cdc.gov/uscs
•• Dong E, Duan L, Wu BU. Racial and ethnic minorities at increased risk for gastric cancer in a regional US population study. Clinical gastroenterology and hepatology: the official clinical practice journal of the American Gastroenterological Association. 2017;15(4):511–7. doi:10.1016/j.cgh.2016.11.033. Recent study highlighting the racial/ethnic differences in the USA with respect to gastric cancer.
• SEER. SEER*Explorer: an interactive website for SEER cancer statistics [Internet]. Beta version. Surveillance Research Program, National Cancer Institute. https://seer.cancer.gov/explorer/. Accessed April 14 2014. Link to the SEER cancer statistics which includes - Recent Trends of cancers, 2000-2014, Long Term Trends, 1975-2014, Rates by Age, Stage Distribution (incidence only), 5-year Relative Survival 2007-2013, and Relative Survival Rates by Time Since Diagnosis.
•• Uthman OA, Jadidi E, Moradi T. Socioeconomic position and incidence of gastric cancer: a systematic review and meta-analysis. J Epidemiol Community Health. 2013;67(10):854–60. doi:10.1136/jech-2012-201108. A meta-analysis of 36 studies from 1966 to 2013 showing the role of socioeconomic status and gastric cancer risk.
Malaty HM, Evans DG, Evans DJ Jr, Graham DY. Helicobacter pylori in Hispanics: comparison with blacks and whites of similar age and socioeconomic class. Gastroenterology. 1992;103(3):813–6.
• Nguyen T, Ramsey D, Graham D, Shaib Y, Shiota S, Velez M, et al. The prevalence of Helicobacter pylori remains high in African American and Hispanic veterans. Helicobacter. 2015;20(4):305–15. doi:10.1111/hel.12199. The prevalence of H pylori is surrogate marker of incidence of gastric cancer in a population. This veterans based study concluded that veterans have a 30% H pylori prevalence in the US, of which African American (50-59 yr - 53.3%) and Hispanic (60-60 yr -48.1%) men had the highest prevalence, thereby highlighting the racial/ethnic differences between H pylori prevalence in the US.
•• Anderson WF, Camargo MC, Fraumeni JF Jr, Correa P, Rosenberg PS, Rabkin CS. Age-specific trends in incidence of noncardia gastric cancer in US adults. Jama. 2010;303(17):1723–8. doi:10.1001/jama.2010.496. This article suggests that between 1977 and 2006, the rates of non-cardia gastric cancer increased in Whites 25–39 years of age, the reason for which were unclear.
• Camargo MC, Anderson WF, King JB, Correa P, Thomas CC, Rosenberg PS, et al. Divergent trends for gastric cancer incidence by anatomical subsite in US adults. Gut. 2011;60(12):1644–9. doi:10.1136/gut.2010.236737. The paper discusses key findings regarding epidemiology of non cardia gastric cancer by anatomical site, race and age group. In this data analysis between 1976-2007, non cardia gastric cancer decreased in both blacks and whites, and for all anatomical subsites except increased incidence in corpus cancer in younger and middle age white men as well as blacks.
•• Peleteiro B, Bastos A, Ferro A, Lunet N. Prevalence of Helicobacter pylori infection worldwide: a systematic review of studies with national coverage. Dig Dis Sci. 2014;59(8):1698–709. doi:10.1007/s10620-014-3063-0. A systematic review of 37 studies through September, 2013 from 22 countries summarizing global trends in H. pylori prevalence.
• Inoue M. Changing epidemiology of Helicobacter pylori in Japan. Gastric cancer: official journal of the International Gastric Cancer Association and the Japanese Gastric Cancer Association. 2017;20(Suppl 1):3–7. doi:10.1007/s10120-016-0658-5. This paper highlights the concept birth cohort effect of H pylori prevalence in Japan which is directly related to its rapid economic development. Less than 2% of subjects born after 2000 have H pylori infection in Japan. This has been the key reason for decline in gastric cancer in Japan, in addition to their primary and secondary prevention strategies.
•• Porras C, Nodora J, Sexton R, Ferreccio C, Jimenez S, Dominguez RL, et al. Epidemiology of Helicobacter pylori infection in six Latin American countries (SWOG Trial S0701). Cancer causes & control: CCC. 2013;24(2):209–15. doi:10.1007/s10552-012-0117-5. Data from major Latin American countries showing a high H. pylori prevalence with no significant difference in prevalence between young and old.
Shu L, Wang XQ, Wang SF, Wang S, Mu M, Zhao Y, et al. Dietary patterns and stomach cancer: a meta-analysis. Nutr Cancer. 2013;65(8):1105–15. doi:10.1080/01635581.2013.828086.
•• Lunet N, Valbuena C, Vieira AL, Lopes C, Lopes C, David L, et al. Fruit and vegetable consumption and gastric cancer by location and histological type: case-control and meta-analysis. European journal of cancer prevention: the official journal of the European Cancer Prevention Organisation (ECP). 2007;16(4):312–27. doi:10.1097/01.cej.0000236255.95769.22. Case control and meta-analysis confirming fruit or vegetable intake was associated with a decreased risk of gastric cancer regardless of the anatomical location and the histological type.
•• Kamada T, Haruma K, Ito M, Inoue K, Manabe N, Matsumoto H, et al. Time trends in Helicobacter pylori infection and atrophic gastritis over 40 years in Japan. Helicobacter. 2015;20(3):192–8. doi:10.1111/hel.12193. A 40-year study showing that the precursor lesions for gastric cancer declined markedly despite no change in H. pylori prevalence but associated with westernization of the diet and reduced salt intake and smoking.
Shiotani A, Cen P, Graham DY. Eradication of gastric cancer is now both possible and practical. Semin Cancer Biol. 2013;23(6 Pt B):492–501. doi:10.1016/j.semcancer.2013.07.004.
•• Wang C, Weber A, Graham DY. Age, period, and cohort effects on gastric cancer mortality. Dig Dis Sci. 2015;60(2):514–23. doi:10.1007/s10620-014-3359-0. First study examining the combined effects of age, period, and birth cohort on the incidence of gastric cancer in a defined population.
•• Peleteiro B, Castro C, Morais S, Ferro A, Lunet N. Worldwide burden of gastric cancer attributable to tobacco smoking in 2012 and predictions for 2020. Dig Dis Sci. 2015;60(8):2470–6. doi:10.1007/s10620-015-3624-x. This study analyzed population attributable fractions with respect to smoking for 118 countries and gender and regional differences in gastric cancer incidence attributable to smoking
•• Zeng M, Mao XH, Li JX, Tong WD, Wang B, Zhang YJ, et al. Efficacy, safety, and immunogenicity of an oral recombinant Helicobacter pylori vaccine in children in China: a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2015;386(10002):1457–64. doi:10.1016/s0140-6736(15)60310-5. RCT from China showing that an oral recombinant H. pylori vaccine provided significant protection from H. pylori in children.
• Yeh JM, Hur C, Schrag D, Kuntz KM, Ezzati M, Stout N, et al. Contribution of H. pylori and smoking trends to US incidence of intestinal-type noncardia gastric adenocarcinoma: a microsimulation model. PLoS medicine. 2013;10(5):e1001451. doi:10.1371/journal.pmed.1001451.
•• Lee YC, Chiang TH, Chou CK, Tu YK, Liao WC, Wu MS, et al. Association between Helicobacter pylori eradication and gastric cancer incidence: a systematic review and meta-analysis. Gastroenterology. 2016;150(5):1113–24.e5. doi:10.1053/j.gastro.2016.01.028. Meta-analysis of 24 studies through May, 2015 confirming a 47% reduction in gastric cancer with H. pylori eradication. Benefits accrued in all populations studied.
Lee YC, Chiang TH, Liou JM, Chen HH, Wu MS, Graham DY. Mass eradication of Helicobacter pylori to prevent gastric cancer: theoretical and practical considerations. Gut and Liver. 2016;10(1):12–26. doi:10.5009/gnl15091.
Graham DY, Malaty HM, Evans DG, Evans DJ Jr, Klein PD, Adam E. Epidemiology of Helicobacter pylori in an asymptomatic population in the United States. Effect of age, race, and socioeconomic status. Gastroenterology. 1991;100(6):1495–501.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Ashish Sharma, Maya Balakrishnan, and Rollin George declare no conflict of interest.
David Graham is a consultant for RedHill Biopharma regarding novel H. pylori therapies and has received research support for culture of H. pylori and is the PI of an international study of the use of antimycobacterial therapy for Crohn’s disease. He is also a consultant for BioGaia in relation to probiotic therapy for H. pylori infection and for Takeda in relation to H. pylori therapies.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Support
Dr. Graham is supported in part by the Office of Research and Development Medical Research Service Department of Veterans Affairs, Public Health Service grant DK56338, which funds the Texas Medical Center Digestive Diseases Center. Dr. Balakrishnan is supported in part by a prevention grant from the Cancer Prevention and Research Institute of Texas–CPRIT (PP160089).
Additional information
This article is part of the Topical Collection on Stomach and Duodenum
Key Points
(1) Gastric cancer incidence has declined worldwide but remains the fifth cause of cancer and the third cause of cancer deaths globally. However, there are significant differences in its incidence across the world and within ethnic/racial subgroups in developed countries.
(2) East Asia currently carries the highest gastric cancer burden; however, the risk is declining as the societies become westernized. Latin America is an outlier in that it has intermediate rates of gastric cancer but a decline is not predicted given the persistence of risk factors.
(3) Reductions in gastric cancer are largely the consequence of improvements in sanitation, food preservation, and availability as well as a decline in H. pylori infections. Reduction in tobacco smoking remains an important modifiable risk factor in gastric and other cancers.
Rights and permissions
About this article
Cite this article
Balakrishnan, M., George, R., Sharma, A. et al. Changing Trends in Stomach Cancer Throughout the World. Curr Gastroenterol Rep 19, 36 (2017). https://doi.org/10.1007/s11894-017-0575-8
Published:
DOI: https://doi.org/10.1007/s11894-017-0575-8