Abstract
Purpose of Review
PET scanner design and performance evaluation has been driven historically by the imaging requirements for whole-body imaging in oncology. Cardiac PET imaging for accurate quantification of myocardial blood flow (MBF) using short-lived tracers such as rubidium-82 imposes additional requirements for wide dynamic range and high count-rate accuracy. This paper examines the technical challenges encountered in cardiac imaging of myocardial perfusion and blood flow quantification.
Recent Findings
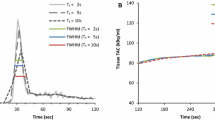
The newest PET-CT scanners using digital silicon photomultiplier technology have high absolute sensitivity (4–20%) and time-of-flight resolution (3–7 cm) which further improves image quality. The concept of “integral” noise equivalent counts (iNEC) is introduced to compare scanner count-rate performance over the wide dynamic range encountered in MBF imaging with rubidium-82.
Summary
The latest-generation digital PET scanners with wide axial field-of-view and enhanced time-of-flight resolution should enable accurate quantification of MBF, without any compromise in the quality of conventional ECG-gated myocardial perfusion images.






Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Yoshinaga K, Katoh C, Manabe O, Klein R, Naya M, Sakakibara M, et al. Incremental diagnostic value of regional myocardial blood flow quantification over relative perfusion imaging with generator-produced rubidium-82 PET. Circ J. 2011;75:2628–34.
Ziadi MC, deKemp RA, Williams KA, Guo A, Chow BJW, Renaud JM, et al. Impaired myocardial flow reserve on rubidium-82 positron emission tomography imaging predicts adverse outcomes in patients assessed for myocardial ischemia. J Am Coll Cardiol. 2011;58:740–8.
Ziadi MC, Dekemp RA, Williams K, et al. Does quantification of myocardial flow reserve using rubidium-82 positron emission tomography facilitate detection of multivessel coronary artery disease? J Nucl Cardiol. 2012;19:670–80.
Murthy VL, Naya M, Foster CR, Hainer J, Gaber M, di Carli G, et al. Improved cardiac risk assessment with non-invasive measures of coronary flow reserve. Circulation. 2011;124:2215–24.
Klein R, Beanlands RSB, deKemp RA. Quantification of myocardial blood flow and flow reserve: technical aspects. J Nucl Cardiol. 2010;17:555–70.
Murthy VL, Bateman TM, Beanlands RS, Berman DS, Borges-Neto S, Chareonthaitawee P, et al. Clinical quantification of myocardial blood flow using PET: joint position paper of the SNMMI Cardiovascular Council and the ASNC. J Nucl Cardiol. 2018;25:269–97.
Bateman T. American Society of Nuclear Cardiology and Society of Nuclear Medicine and Molecular Imaging joint position statement on the clinical indications for myocardial perfusion PET. J Nucl Cardiol. 2016;23:1227–31.
Tahari AK, Lee A, Rajaram M, Fukushima K, Lodge MA, Lee BC, et al. Absolute myocardial flow quantification with 82Rb PET/CT: comparison of different software packages and methods. Eur J Nucl Med Mol Imaging. 2014;41:126–35.
Nesterov SV, Deshayes E, Sciagrà R, Settimo L, Declerck JM, Pan XB, et al. Quantification of myocardial blood flow in absolute terms using 82Rb PET imaging. JACC Cardiovasc Imaging. 2014;7:1119–27.
• Renaud JM, Yip K, Guimond J, et al. Characterization of 3-dimensional PET systems for accurate quantification of myocardial blood flow. J Nucl Med. 2017;58:103–9 This study developed a method to evaluate the high count-rate performance of PET scanners, as observed during first-pass dynamic imaging for accurate quantification of myocardial blood flow.
Klein R, Ocneanu A, Renaud JM, Ziadi MC, Beanlands RSB, deKemp RA. Consistent tracer administration profile improves test–retest repeatability of myocardial blood flow quantification with 82Rb dynamic PET imaging. J Nucl Cardiol. 2018;25:929–41.
Cherry SR, Jones T, Karp JS, Qi J, Moses WW, Badawi RD. Total-body PET: maximizing sensitivity to create new opportunities for clinical research and patient care. J Nucl Med. 2018;59:3–12.
Badawi RD, Shi H, Hu P, Chen S, Xu T, Price PM, et al. First human imaging studies with the EXPLORER total-body PET scanner. J Nucl Med. 2019;60:299–303.
Vandenberghe S, Mikhaylova E, D’Hoe E, Mollet P, Karp JS. Recent developments in time-of-flight PET. EJNMMI Phys. 2016;3:3.
Surti S. Update on time-of-flight PET imaging. J Nucl Med. 2015;56:98–105.
NEMA NU 2-2018. Performance measurements of positron emission tomographs (PET). 2018. Available at https://www.nema.org/Standards/Pages/Performance-Measurements-of-Positron-Emission-Tomographs.aspx.
Budinger TF. Time-of-flight positron emission tomography: status relative to conventional PET. J Nucl Med. 1983;24:73–8.
Zhang J, Maniawski P, Knopp MV. Performance evaluation of the next generation solid-state digital photon counting PET/CT system. EJNMMI Res. 2018;8:97.
van Sluis J, de Jong J, Schaar J, Noordzij W, van Snick P, Dierckx R, et al. Performance characteristics of the digital biograph vision PET/CT system. J Nucl Med. 2019;60:1031–6.
Pan T, Einstein SA, Kappadath SC, Grogg KS, Lois Gomez C, Alessio AM, et al. Performance evaluation of the 5-ring GE Discovery MI PET/CT system using the national electrical manufacturers association NU 2-2012 standard. Med Phys. 2019;46:3025–33.
Li X, Qi W, Miyahara M, Kolthammer J. Performance characterization of an SiPM-based time-of-flight Canon PET/CT scanner. J Nucl Med. 2020;61(S1):14 [abstract].
Klein R, Celiker-Guler E, Rotstein BH, deKemp RA. PET and SPECT tracers for myocardial perfusion imaging. Semin Nucl Med. 2020;50:208–18.
Wangerin K, Ahn S, Ross SG, Kinahan PE, Manjeshwar RM (2015) Improving lesion detectability in PET imaging with a penalized likelihood reconstruction algorithm. In: Mello-Thoms CR, Kupinski MA (eds). Orlando, Florida, United States, p 94160W.
Kesner AL, Chung JH, Lind KE, Kwak JJ, Lynch D, Burckhardt D, et al. Validation of software gating: a practical technology for respiratory motion correction in PET. Radiology. 2016;281:239–48.
Manwell S, Klein R, Xu T, deKemp RA. Clinical comparison of the positron emission tracking (PeTrack) algorithm with the real-time position management system for respiratory gating in cardiac positron emission tomography. Med Phys. 2020;47:1713–26.
Feng T, Wang J, Fung G, Tsui B. Non-rigid dual respiratory and cardiac motion correction methods after, during, and before image reconstruction for 4D cardiac PET. Phys Med Biol. 2016;61:151–68.
•• Armstrong IS, Hayden C, Memmot MJ, Arumugam P (2020) A preliminary evaluation of a high temporal resolution data-driven motion correction algorithm for rubidium-82 on a SiPM PET-CT system. J Nucl Cardiol. Online First. Myocardial perfusion imaging (MPI) quality can be improved dramatically using a second-by-second correction of patient body motion within the PET image reconstruction process.
Presotto L, Busnardo E, Perani D, Gianolli L, Gilardi MC, Bettinardi V. Simultaneous reconstruction of attenuation and activity in cardiac PET can remove CT misalignment artifacts. J Nucl Cardiol. 2015;23:1086–97.
Hunter C, Presotto L, Klein R, Pelletier-Galarneau M, Ruddy TR, Dekemp R. P303-maximum likelihood reconstruction of activity and attenuation (MLAA) for CO2 stress in Rb-82 PET/CT respiratory gated imaging. Eur Heart J - Cardiovasc Imaging. 2019;20(3):jez148.031. https://doi.org/10.1093/ehjci/jez148.031.
Petibon Y, Rakvongthai Y, El Fakhri G, Ouyang J. Direct parametric reconstruction in dynamic PET myocardial perfusion imaging: in vivo studies. Phys Med Biol. 2017;62:3539–65.
Lodge MA, Carson RE, Carrasquillo JA, Whatley M, Libutti SK, Bacharach SL. Parametric images of blood flow in oncology PET studies using [15O]water. J Nucl Med. 2000;41:1784–92.
•• Germino M, Carson RE. Cardiac-gated parametric images from 82 Rb PET from dynamic frames and direct 4D reconstruction. Med Phys. 2018;45:639–54 This study demonstrated the ability reconstruct ECG-gated images of myocardial blood flow (ml/min/g) directly from the raw coincidence PET data, including compensation for respiratory-motion.
van Dijk JD, Jager PL, van Osch JAC, Khodaverdi M, van Dalen JA. Comparison of maximal Rubidium-82 activities for myocardial blood flow quantification between digital and conventional PET systems. J Nucl Cardiol. 2019;26:1286–91.
deKemp RA, Genoud P, Pappon M, Kamani C, Gnesi S, Prior JO. Dynamic range of the newest digital PET-CT scanner for myocardial blood flow quantification using rubidium-82. J Nucl Med. 2020;61:334.
Reddin JS, Scheuermann JS, Bharkhada D, Smith AM, Casey ME, Conti M, Karp J (2018) Performance evaluation of the SiPM-based Siemens Biograph Vision PET/CT System. 2018 IEEE Nuclear Science Symposium and Medical Imaging Conference Proceedings (NSS/MIC), Sydney, Australia, 2018, pp. 1–5. https://doi.org/10.1109/NSSMIC.2018.8824710.
United Imaging uMI 780 Data Sheet. Available at https://usa.united-imaging.com/products/molecular-imaging/umi-780/.
Rausch I, Ruiz A, Valverde-Pascual I, Cal-González J, Beyer T, Carrio I. Performance evaluation of the Vereos PET/CT system according to the NEMA NU2-2012 standard. J Nucl Med. 2019;60:561–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Ran Klein and Robert deKemp are consultants with Jubilant DRAXimage and have received grant funding from university/industry partnership programs including GE Healthcare, Jubilant DRAXimage, Shelley Medical Solutions, and Hermes Medical Solutions. They receive revenues from rubidium generator technologies licensed to Jubilant DRAXimage and revenue shares from the sale of FlowQuant®.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Nuclear Cardiology
Rights and permissions
About this article
Cite this article
Klein, R., deKemp, R.A. Selection of PET Camera and Implications on the Reliability and Accuracy of Absolute Myocardial Blood Flow Quantification. Curr Cardiol Rep 22, 109 (2020). https://doi.org/10.1007/s11886-020-01376-0
Published:
DOI: https://doi.org/10.1007/s11886-020-01376-0