Abstract
Background
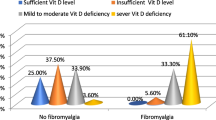
Fibromyalgia patients who are exposed to extreme oxidative stress may face more severe clinical features or oxidative stress may be increased by the severity of the disease.
Aim
The purpose of these investigation were to determine serum paraoxonase activities (PON-1) and nitric oxide (NO) activities and malondialdehyde (MDA) level in fibromyalgia and whether there were any associations between these enzymes activities, MDA level, and clinical parameters.
Methods
The study groups were consisted of 30 primer fibromyalgia patients and 30 healthy subjects. Clinical findings, pain severity, functional disability, general health status, anxiety, and depression assessed, and serum PON-1 activity, MDA, and NO levels were measured.
Results
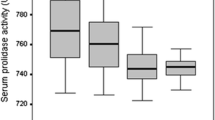
The primer fibromyalgia group had significantly higher MDA, low density lipoprotein-cholesterol (LDL-C), and decreased PON-1 activity, NO, and high density lipoprotein-cholesterol (HDL-C) with respect to controls. The paraoxonase activity was negatively correlated with MDA, LDL-C, Visual Analog Scale (VAS), Fibromyalgia Impact Questionnaire score (FIQ score), tender point score, age, and BDI score, while positively correlated with NO and HDL-C. MDA level was positively correlated with VAS, FIQ score, tender point score, age, and negatively correlated with NO level.
Conclusion
These results suggest that FMS patients have an alteration in levels of MDA, NO, and PON-1 activities. We think that impaired oxidant/antioxidant status may affect the symptoms of the disease. Also, they may be of importance in the complex physiopathologic mechanism behind the development of FMS.


Similar content being viewed by others
Data Availability
Availability of data and material: Data is available from the corresponding author upon request
References
Siracusa R, Paola RD, Cuzzocrea S et al (2021) Fibromyalgia: pathogenesis, mechanisms, diagnosis and treatment options update. Int J Mol Sci 22(8):3891. https://doi.org/10.3390/ijms22083891
Maffei ME (2020) Fibromyalgia: recent advances in diagnosis, classification, pharmacotherapy and alternative remedies. Int J Mol Sci 21(21):7877. https://doi.org/10.3390/ijms21217877
Katz RS, Wolfe F, Michaud K (2006) Fibromyalgia diagnosis: a comparison of clinical, survey, and American College of Rheumatology criteria. Arthritis Rheum 54(1):169–176
Topbas M, Cakirbay H, Gulec H et al (2005) The prevalence of fibromyalgia in women aged 20–64 in Turkey. Scand J Rheumatol 34(2):140–144
Ahmadnezhad M, Ghazizadeh H, Bijari M et al (2021) Serum pro-oxidant-antioxidant balance in subjects with type 2 diabetes mellitus. Comb Chem High Throughput Screen 24(9):1476–1481. https://doi.org/10.2174/1386207323999201103203153
Bagherifard A, AminiKadijani A, Yahyazadeh H et al (2020) The value of serum total oxidant to the antioxidant ratio as a biomarker of knee osteoarthritis. Clin Nutr ESPEN 38:118–123. https://doi.org/10.1016/j.clnesp.2020.05.019
Baskol G, Baskol M, Yurci A et al (2006) Serum paraoxonase 1 activity and malondialdehyde levels in patients with ulcerative colitis. Cell Biochem Func 24:283–286
Akkuş S, Naziroğlu M, Eriş S et al (2009) Levels of lipid peroxidation, nitric oxide, and antioxidant vitamins in plasma of patients with fibromyalgia. Cell Biochem Funct 27(4):181–185. https://doi.org/10.1002/cbf.15487
McIver KL, Evans C, Kraus RM et al (2006) NO-mediated alterations in skeletal muscle nutritive blood flow and lactate metabolism in fibromyalgia. Pain 120:161–169
Pall ML (2001) Common etiology of posttraumatic stress disorder, fibromyalgia, chronic fatigue syndrome and multiple chemical sensitivity via elevated nitric oxide/peroxynitrite. Med Hypotheses 57(2):139–145
Ozgocmen S, Ozyurt H, Sogut S et al (2006) Antioxidant status, lipid peroxidation and nitric oxide in fibromyalgia: etiologic and therapeutic concerns. Rheumatol Int 26:598–603
Fatima G, Das SK, Mahdi AA (2013) Oxidative stress and antioxidative parameters and metal ion content in patients with fibromyalgia syndrome: implications in the pathogenesis of the disease. Clin Exp Rheumatol 31(6 Suppl 79):S128–S133
Altindag O, Celik H (2006) Total antioxidant capacity and the severity of the pain in patients with fibromyalgia. Redox Rep 11:131–135. https://doi.org/10.1179/135100006X116628
Lioudaki S, Verikokos C, Kouraklis G et al (2019) Paraoxonase-1: characteristics and role in atherosclerosis and carotid artery disease. Curr Vasc Pharmacol 17(2):141–146. https://doi.org/10.2174/1570161115666171129212359
Taler-Verčič A, Goličnik M, Bavec A (2020) The structure and function of paraoxonase-1 and its comparison to paraoxonase-2 and -3. Molecules 25(24):5980. https://doi.org/10.3390/molecules25245980
Zuin M, Trentini A, Marsillach J et al (2022) Paraoxonase-1 (PON-1) arylesterase activity levels in patients with coronary artery disease: a meta-analysis. Dis Markers 2022:4264314. https://doi.org/10.1155/2022/4264314
Altindag O, Gur A, Calgan N et al (2007) Paraoxonase and arylesterase activities in fibromyalgia. Redox Rep 12(3):134–138. https://doi.org/10.1179/rer.2007.12.3.134
Sendur OF, Turan Y, Tastaban E et al (2008) Serum antioxidants and nitric oxide levels in fibromyalgia: a controlled study. Rheumatol Int 29(6):629–633. https://doi.org/10.1007/s00296-008-0738-x
Barrera G, Pizzimenti S, Daga M et al (2018) Lipid peroxidation-derived aldehydes, 4-hydroxynonenal and malondialdehyde in aging-related disorders. Antioxidants 7(8):102
Toker A, Kucuksen S, Kucuk A et al (2014) Serum ischemia-modified albumin and malondialdehyde levels and superoxide dismutase activity in patients with fibromyalgia. Clin Lab 60(10):1609–1615. https://doi.org/10.7754/clin.lab.2014.131017
Hassan FE, Sakr HI, Mohie PM et al (2021) Pioglitazone improves skeletal muscle functions in reserpine-induced fibromyalgia rat model. Ann Med 53(1):1032–1040. https://doi.org/10.1080/07853890.2021.1916069
Koca TT, Seyithanoglu M, Sagiroglu S et al (2018) Frequency of audiological complaints in patients with fibromyalgia syndrome and its relationship with oxidative stress. Niger J Clin Pract 21(10):1271–1277. https://doi.org/10.4103/njcp.njcp_95_18
Wolfe F, Smythe HA, Yunus MB et al (1990) The American College of Rheumatology 1990 criteria for the classification of fibromyalgia. Report of the multicenter criteria committee. Arthritis Rheum 33:160–172
Hisli N (1989) Reliability and validity of Beck Depression Inventory among university students. J Turk Psychol 7:3–13
Sarmer S, Ergin S, Yavuzer G (2000) The validity and reliability of the Turkish version of the Fibromyalgia Impact Questionnaire. Rheumatol Int 20(1):9–12. https://doi.org/10.1007/s002960000077
Asakawa T, Matsushita S (1980) Coloring conditions of thiabarbituric acid test for detectinf lipid hydroperoxides. Lipids 15:137–140
Furlong CE, Richter RJ, Seidel SL et al (1989) Spectrophotometric assays for the enzymatic hydrolysis of the active metabolites of chlorpyrifos and parathion by plasma paraoxonase/arylesterase. Anal Biochem 180(2):242–247. https://doi.org/10.1016/0003-2697(89)90424-7
Bories PN, Bories C (1995) Nitrate determination in biological fluids by an enzymatic one-step assay with nitrate reductase. Clin Chem 41(6 Pt 1):904–907
Bagis S, Tamer L, Sahin G et al (2005) Free radicals and antioxidants in primary fibromyalgia: an oxidative stress disorder? Rheumatol Int 25:188–190
Gur M, Aslan M, Yildiz A et al (2006) Paraoxonase and arylesterase activities in coronary artery disease. Eur J of Clin Invest 36:779–787
Larson AA, Giovengo SL, Russel IJ et al (2000) Changes in the concentrations of aminoacids in the cerebrospinal fluid that correlate with pain in patients with fibromyalgia : implications for nitric oxide pathways. Pain 87:201–211
Cordero MD (2011) Oxidative stress in fibromyalgia: pathophysiology and clinical implications. Reumatol Clin 7(5):281–283
Cordero MD, de Miguel M, Moreno-Fernandez AM (2011) Mitochondrial dysfunction in fibromyalgia and its implication in the pathogenesis of disease. Med Clin (Barc) 136(6):252–256
Inanici F, Yunus MB (2004) History of fibromyalgia: past to present. Curr Pain Headache Rep 8(5):369–378
De Caterina R, Libby P, Peng HB et al (1995) Nitric oxide decreases cytokine-induced endothelial activation. Nitric oxide selectively reduces endothelial expression of adhesion molecules and proinflammatory cytokines. J Clin Invest 96:60–68
Scandalios JG (2005) Oxidative stress: molecular perception and transduction of signals triggering antioxidant gene defenses. Braz J Med Biol Res 38:995–1014
Alasehirli B, Demiryurek S, Arica E et al (2007) No evidence for an association between the Glu298Asp polymorphism of the endothelial nitric oxide synthase gene and fibromyalgia syndrome. Rheumatol Int 27:275–280
Shukla V, Kumar DS, Ali MA et al (2020) Nitric oxide, lipid peroxidation products, and antioxidants in primary fibromyalgia and correlation with disease severity. J Med Biochem 39(2):165–170. https://doi.org/10.2478/jomb-2019-0033
Author information
Authors and Affiliations
Contributions
All authors contributed to data analysis, drafting, and critical revision of the publication, and they agree to be responsible for all elements of it.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Atamer, Y., Sarac, S., Asık, H.K. et al. Serum paraoxonase activities, nitric oxide, and malondialdehyde levels are altered in patients with primary fibromyalgia syndrome. Ir J Med Sci 192, 2541–2547 (2023). https://doi.org/10.1007/s11845-023-03280-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-023-03280-2