Abstract
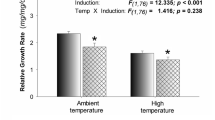
As soybean (Glycine max [L.] Merrill) is an important crop throughout the world, the action of herbivorous insects responsible for economic and productivity losses is the subject of constant research. Spodoptera frugiperda (J.E. Smith) caterpillars can cause extensive damage to soybean culture; this work investigated possible harmful effects on these caterpillars associated with the possible induced defenses of soybean plants. For this purpose, we assessed the biology of the insect (leaf consumption and performance traits) and chemical composition of the soybean leaves by ultraviolet–visible spectroscopy and chemometrics of three treatments: control, mechanically wounded soybean leaves, and S. frugiperda-damaged soybean leaves. The results reveal that both types of injuries induce changes in soybean metabolism regarding the production of phenolic substances, although only the herbivore-damaged plants provoke negative effects on insect biology. Variations in carotenoid production during the circadian cycle were also found in the control group. These results confirm that the soybean plants could endure and activate chemical defense mechanisms that impair the developmental lifecycle of the insect, suggesting possibilities for sustainable control strategies.





Similar content being viewed by others
References
Awmack CS, Leather SR (2002) Host plan quality and fecundity in herbivorous insect. Annu Rev Entomol 47:817–844
Azcarate SM, Cantarelli MA, Pellerano RG, Marchevsky EJ, Camiña M (2013) Classification of Argentinean Sauvignon blanc wines by UV spectroscopy and chemometric methods. J Food Sci 78(3):427–432
Barros EM, Torres JB, Ruberson JR, Oliveira MD (2010a) Development of Spodoptera frugiperda on different hosts and damage to reproductive structures in cotton. Entomol Exp Appl 137:237–245
Barros EM, Torres JB, Bueno AF (2010b) Oviposição, desenvolvimento e reprodução de Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae) em diferentes hospedeiros de importância econômica. Neotrop Entomol 39(6):996–1001
Beebe KR, Pell RJ, Seasholtz MB (1998) Chemometrics: a practical guide. Wiley, New York
Bernardi O, Sorgatto RJ, Barbosa AD, Domingues FA, Dourado PM, Carvalho RA et al (2014) Low suceptibility of Spodoptera cosmioides, Spodoptera eridania and Spodoptera frugiperda (Lepidoptera: Noctuidae) to genetically-modified soybean expressing Cry1Ac protein. Crop Prot 58:33–40
Blanco CA, Chiaravalle W, Dalla-Ria M, Farias JR, Garía-Degano MF, Gastaminza G, Mota-Sánchez D, Murúa MG, Omoto C, Pieralisi BK, Rodríguez J, Rodríguez-Maciel JC, Terá-Santofimio H, Terán-Vargas AP, Valencia SJ, Willink E (2016) Current situation of pests targeted by Bt crops in Latin America. Curr Opin Insect Sci 15:131–138
Boregas KGB, Mendes SM, Waquil JM, Fernandades GW (2013) Estágio de adaptação de Spodoptera frugiperda (J. E. Smith) (Lepidoptera: Noctuidae) em hospedeiros alternativos. Bragantia 77(1):61–70 Portuguese
Bortoli SA, Murata AT, Vacari AM, Bortoli CP, Ramalho DG (2012) Herbivoria em soja: efeito na composição química das folhas e na biologia da lagarta da soja e do percevejo verde pequeno. Comun Sci 3(3):192–198 Portuguese
Broch DL, Ranno SK (2012) Fertilidade do solo, adubação e nutrição da cultura da soja. In: Tecnologia e Produção: Soja e Milho 2011/2012. Fundação MS, Maracajú. [cited 2016 Nov 22], [about 37 p.]. Portuguese. Available from: http://www.fundacaoms.org.br/base/www/fundacaoms.org.br/media/attachments/15/15/5385dbdd42bd6c653952352ab872eed8dd643a607266e_02-fertilidade-do-solo-adubacao-e-nutricao-da-cultura-de-soja_543353833.pdf
Bueno AF, Panizzi AR, Corrêa-Ferreira BS, Hoffmann-Campo CB, Sosa-Gómez DR, Gazzoni DL (2012) Histórico e evolução do manejo integrado de pragas da soja no Brasil. In: Hoffmann-Campo CB, Corrêa-Ferreira BS, Moscardi F (eds) Soja: manejo integrado de insetos e outros artrópodes-praga. Embrapa, Brasília, pp 37–74
Bueno RCOF, Bueno AF, Moscardi F, Parra JRP, Hoffmann-Campo CB (2011) Lepidopteran larva consumption of soybean foliage: basis for developing multiple-species economic thresholds for pest management decisions. Pest Manag Sci 67(2):170–174
Capineira JL (2001) Handbook of vegetable pests. Academic press, San Diego
Capineira JL (2008) Encyclopedia of entomology, 2nd edn. Springer, New York
Casale M, Oliveri P, Armanino C, Lanteri S, Forina M (2010) NIR and UV-Vis spectroscopy, artificial nose and tongue: comparision of four fingerprinting techniques for the characterisation of Italian red wines. Anal Chim Acta 668:143–148
CONAB—Companhia Nacional de Abastecimento (2016) Acompanhamento da safra brasileira de grãos—safra 2015/2016. 3(10), Julho 2016. Available at http://www.conab.gov.br. Accessed 4 Aug 2016
Cruz I (1995) A lagarta-do-cartucho na cultura do milho. Circular técnica 21. Embrapa, Sete Lagoas
Fehr WR, Caviness CE (1977) Stages of soybean development [monograph] Special Report 80: Iowa State University Cooperative Extension Service.
Formentini AC, Sosa-Gómez DR, Paula-Moraes SV, Barros NM, Specht A (2015) Lepidoptera (Insecta) associated with soybeans in Argentina, Brazil,Chile and Uruguai. Cienc Rural 45(12):2113–2120
Fürstenberg-Hägg J, Sagrobelny M, Bak S (2013) Plant defense against insect herbivores. Int J Mol Sci 14(5):10242–10297
Gobbo-Neto L, Lopes N (2007) Plantas medicinais: fatores de influência no conteúdo de metabólitos secundários. Quim Nova 30:374–381 Portuguese
Greene GL, Leppla NC, Dickerson WA (1976) Velvetbean caterpillar: a rearing procedure and artificial medium. J Econ Entomol 69:487–488
Groβ N, Wasternack C, Köck M (2004) Wound-induced RNaseLE expression is jasmonate and systemin independent and occurs only locally in tomato (Lycopersicon esculentum cv. Lukullus). Phytochemistry 65:1343–1350
Hartman GL, West ED, Herman TK (2011) Crops that feed the World 2. Soybean—worldwide production and constraints caused by pathogens. Food Secur 3(1):5–17
Heidel-Fischer HM, Vogel H (2015) Molecular mechanisms of insect adaptation to plant secondary compounds. Curr Opin Insect Sci 8:8–14
Ho HM, Chen RY, Leun LK, Chan FL, Huang Y, Chen XY (2002) Difference in flavonoid and isoflavone profile between soybean and soy leaf. Biomed Pharmacother 56:289–295
Isman M (2002) Insect antifeedants. Pestic. Outlook 13:152–157. doi:10.1039/B206507J
Jahnke LS (1999) Massive carotenoid accumulation in Dunaliella bardawil induced by ultraviolet-A radiation. J Photochem Photobiol B 48:68–74
Markham KR (1982) Techniques of flavonoids identification. Academic Press, Tokyo
Martinez SS, Van Emden HF (2001) Redução do crescimento, deformidades e mortalidade de Spodoptera littoralis (Boisduval) (Lepidoptera: Noctuidae) causadas por azadiractina. Neotrop Entomol 30(1):113–125
Mithöfer A, Boland W (2012) Plant defense against herbivores: chemical aspects. Annu Rev Plant Biol 63:431–450
Mithöfer A, Maffei ME (2016) General mechanisms of plant defense and plant toxins. In: Gopalakrishnakone P, Carlini CR, Ligabue-Braun R (eds) Plant toxins. Springer, Netherlands. doi:10.1007/978-94-007-6728-7
Murakami S, Nakata R, Aboshi T, Yoshinaga N, Teraishi M, Okumoto Y et al (2014) Insect-induced daidzein, formononetin and their conjugates in soybean leaves. Metabolites 4:532–546
Oerke EC (2006) Crop losses to pests. J Agric Sci 144(1):31–43
Oerke EC, Dehne HW (2004) Safeguarding production—losses in major crops and the role of crop protection. Crop Prot 23(4):275–285
Oliveira CM, Auad AM, Mendes SM, Frizzas MR (2014) Crop losses and the economic impact of insects pest on Brazilian agriculture. Crop Prot 56:50–54
Osman SF, Fett WF (1983) Isoflavone glucoside stress metabolites of soybean leaves. Phytochemistry. 22(9):1921–1923
Parra JRP (1986) Criação de insetos para estudos com patógenos. In: Alves SB (ed) Controle microbiano de insetos. Manole, São Paulo, pp 348–373
Piubelli GC, Hoffmann-Campo CB, Moscardi F, Miyakubo SH, Oliveira CN (2005) Are chemical compounds important for soybean resistance to Anticarsia gemmatalis? J Chem Ecol 31(7):1509–1525
Pogue MG (2002) A world revision of the Genus Spodoptera Guenée (Lepidoptera: Noctuidae). Mem Am Entomol Soc 43:1–202
Porter PM, Banmiairt WL, Hassett JJ (1986) Phenolic acids and flavonoids in soybean root and leaf extracts. Environ Exp Bot 26(1):65–73
Reymond P, Weber H, Damond M, Farmer EE (2000) Differential gene expression in response to mechanical wounding and insect feeding in Arabidopsis. Plant Cell 12(5):707–720
Robards K, Prenzler PD, Tucker G, Swatsitang P, Glover W (1999) Phenolic compounds and their role in oxidative processes in fruits. Food Chem 66:401–436
Rodriguez-Amaya DB (2001) A guide to carotenoid analysis in foods. ILSI Press, Washington
Roscoe R, Gitti DC (2012) Manejo e Fertilidade do solo para a cultura da soja. In: Tecnologia e produção de soja 2013/2014. Fundação MS, Maracajú, pp 16–44. [cited 2016 Nov 22] Portuguese. Available from http://www.fundacaoms.org.br/base/www/fundacaoms.org.br/media/attachments/144/144/55ad57541853c97f65214d32ca0f45913585c9514bb71_01-manejo-e-fertilidade-do-solo-para-a-cultura-da-soja.pdf
Sá VGM, Fonseca BVC, Boregas KGB, Waquil JM (2009) Sobrevivência e Desenvolvimento Larval de Spodoptera frugiperda (J E Smith) (Lepidoptera: Noctuidae) em Hospedeiros Alternativos. Neotrop Entomol 38(1):108–115
Santos KB, Neves P, Meneguim AM, Santos RB, Santos WJ, Boas GV et al (2009) Selections and characterization of the Bacillus thuringiensis strains toxic to Spodoptera eridania (Cramer), Spodoptera coismioides (Walker) and Spodoptera frugiperda (Smith) (Lepidoptera: Noctuidae). Biol Control 50(2):157–163
Sârbu C, Naşcu-Briciu RD, Kot-Wasik A, Gorinstein S, Wasik A, Gorinstein S et al (2012) Classification and fingerprint of kiwi and pomelo fruits by multivariate analysis of chromatographic and spectroscopic data. Food Chem 130:994–1002
Schoonhoven LM, van Loon JJA, Dicke M (2005) Insect-plant biology, 2nd edn. Oxford University Press, Great Britain
Silva RA, Reis VM, Baldani Olivares FL (2008) Defesa de Plantas contra Ataque de Fitopatógenos. Embrapa Agrobiologia, Seropédica Portuguese
Silveira-Neto S, Nakano O, Barbin D, Villa-Nova NA (1976) Manual de ecologia dos insetos. Agronômica Ceres, Piracicaba Portuguese
Taiz L, Zeiger E (2009) Plant physiology, 4th edn. Sinauer, Sunderland
Vázquez-Flota F, Carrilo-Pech M, Minero-Garcia Y, Miranda-Ham ML (2004) Alkaloid metabolism in wounded Catharanthus roseus seedlings. Plant Physiol Biochem 42:623–628
Victorio CP, Kuster RM, Lage CLS (2007) Qualidade de Luz e Produção de Pigmentos Fotossintéticos em Plantas In Vitro de Phyllanthus tenellus Roxb. Rev Bras Biocienc 5(2):213–215
War AR, Paulraj MG, Ahmad T, Buhroo AA, Hussain B, Ignacimuthu S, Sharma HC (2012) Mechanisms of plant defense against insect herbivores. Plant Signal Behav 7(10):1306–1320
Acknowledgements
We thank Agência de Desenvolvimento Agrário e Extensão Rural (AGRAER) and Universidade Federal de Mato Grosso do Sul (UFMS) for their support. This work was funding by Conselho Nacional de Desenvolvimento Científico e Tecnológico [grant number 481507/2013] and Fundação de Apoio ao Desenvolvimento do Ensino, Ciência e Tecnologia do Estado de Mato Grosso do Sul [grant number TO 0165/12].
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by Joe Louis.
Rights and permissions
About this article
Cite this article
Peruca, R.D., Coelho, R.G., da Silva, G.G. et al. Impacts of soybean-induced defenses on Spodoptera frugiperda (Lepidoptera: Noctuidae) development. Arthropod-Plant Interactions 12, 257–266 (2018). https://doi.org/10.1007/s11829-017-9565-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-017-9565-x