Abstract
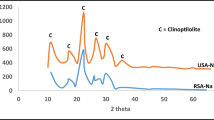
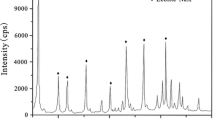
This paper presents the adsorption of Cu(II) ions from aqueous solution on Na and Fe-modified clinoptilolite. The copper adsorption experiments were performed in a batch system considering an optimum contact time of 24 h. Changes in the surfaces and structure were characterized by SEM data. According to the SEM results, it was anticipated that the removal efficiency of Fe-modified clinoptilolite was the highest compared with the natural and Na-modified clinoptilolites. Adsorption of Cu(II) ions by modified clinoptilolites was investigated as a function of the initial Cu(II) concentration, solution pH, and temperature. According to the results, the maximum adsorbed Cu amount onto Fe-modified was 19.40mg/l at the optimum operating condition with a pH value of 5.5 and temperature of 60 °C. According to the thermodynamic evaluations, positive ΔS and negative ΔG were found for the adsorption process showing that the adsorption reaction is a spontaneous process and more favorable at high temperatures. Sorption data have been interpreted in terms of Langmuir and Freundlich, Temkin and Dubinin-Radushkevich. The adsorption equilibrium was best described by the Langmuir adsorption isotherm. In addition, according to the Sips model, the sorption of Cu(II) ions on the Fe-modified clinoptilolite was found to be heterogeneous. The kinetic study showed that the Fe-modified clinoptilolite followed the pseudo-second order model. The results indicated that the clinoptilolite-rich tuff in its iron oxide form could be efficiently used for the removal of copper from aqueous solutions.
Similar content being viewed by others
References
F. Fu and Q. Wang, J. Environ. Manage., 92, 407 (2011).
K. Elaiopoulos, T. Perraki and E. Grigoropoulou, Micropor. Mesopor. Mater., 112, 441 (2008).
G. Gottardi and E. Galli, Natural Zeolites, Berlin, Springer-Verlag (1985).
F. A. Mumpton, Natural Zeolites Occurrence Properties and Use, New York, Pergamon (1976).
K. Koyama and Y. Takéuchi, J. Crystallogr., 145, 216 (1977).
W. W. Eckenfelder, Industrial Water Pollution Control, New York, McGraw-Hill (1989).
Ö. Yavuza, Y. Altunkaynakb and F. Güzel, Water Res., 37, 948 (2003).
H. Cui, L. Y. Li and J. R. Grace, Water Res., 40, 3359 (2006).
K. Payne and T. Abdel-Fattah, J. Environ. Sci. Health, Part A A 39, 2275 (2004).
T. M. Abdel-Fattah, L. K. Isaacs and K. Payne, Federal Facilities Environ. J., 14, 113 (2003).
S. Babel and T. A. Kurniawan, J. Hazard. Mater., 97, 219 (2003).
K. Payne and T. M. Abdel-Fattah, J. Environ. Sci. Health, 40, 723 (2005).
S. Çoruh, Desalination, 225, 41 (2008).
M. Panayotova and B. Velikov, J. Environ. Sci. Health A., 37, 139 (2002).
A. Arcoya, J. Gonzalez, N. Travieso and X. Seoane, Clay Minerals, 29, 123 (1994).
M. Bilici Baskan and A. Pala, Desalination, 281, 396 (2011).
S. A. Abdulkareem, E. Muzenda, A. S. Afolabi and J. Kabuba, Arabian J. Sci. Eng., 38, 2263 (2013).
A. Dimirkou, Water Res., 41, 2763 (2007).
M. K. Doula and A. Dimirkou, J. Hazard. Mater., 151, 738 (2008).
A. Dimirkou, A. Ioannou and M. Doula, Adv. Colloid Interface Sci., 97, 37 (2002).
Z. Li, J.-S. Jean, W.-T. Jiang, P.-H. Chang, C.-J. Chen and L. Liao, J. Hazard. Mater., 187, 318 (2011).
E. Erdal, J. Hazard. Mater., 165, 63 (2009).
D. K. Maria, Water Res., 40, 3167 (2006).
M. K. Doula, Chemosphere, 67, 731 (2007).
A. Dimirkou and M. K. Doula, Desalination, 224, 280 (2008).
M. Kragovic, A. Dakovic, Ž. Sekulic, M. Trgo, M. Ugrina, J. Peric and G. D. Gatta, Appl. Surface Sci., 258, 3667 (2012).
M. K. Doula, Water Res., 43, 3659 (2009).
W. S. Wan Ngah, C. S. Endud and R. Mayanar, React. Funct. Polym., 50, 181 (2002).
K. G. Bhattacharyya and S. S. Gupta, Sep. Purif. Technol., 50, 388 (2006).
B. Acemioglu, A. Samil, M. H. Alma and R. Gundogan, J. Appl. Polym. Sci., 89, 1537 (2003).
S. Larous, A. H. Meniai and M. B. Lehocine, Desalination, 185, 483 (2005).
B. Concepción-Rosabal, G. Rodríguez-Fuentes, N. Bogdanchikova, P. Bosch, M. Avalos and V. H. Lara, Micropor. Mesopor. Mater., 86, 249 (2005).
R. Petrus and J. WarchoÅ, Micropor. Mesopor. Mater., 61, 137 (2003).
N. Lihareva, L. Dimova, O. Petrov and Y. Tzvetanova, Micropor. Mesopor. Mater., 130, 32 (2010).
H. Yuh-Shan, Scientometrics, 59, 171 (2004).
S. Kocaoba, Y. Orhan and T. Akyuz, Desalination, 214, 1 (2007).
K. S. Hui, C. Y. H. Chao and S. C. Kot, J. Hazard. Mater., 127, 89 (2005).
M. Sprynskyy, B. Buszewski, A. P. Terzyk and J. Namiesnik, J. Colloid Interface Sci., 304, 21 (2006).
V. J. Inglezakis, M. D. Loizidou and H. P. Grigoropoulou, J. Colloid Interface Sci., 261, 49 (2003).
E. Álvarez-Ayuso, A. García-Sánchez and X. Querol, Water Res., 37, 4855 (2003).
A. H. Ören and A. Kaya, J. Hazard. Mater., 131, 59 (2006).
N. Moreno, X. Querol and C. Ayora, Environ. Sci. Technol., 35, 3526 (2001).
K. Gedik and I. Imamoglu, J. Hazard. Mater., 155, 385 (2008).
V. J. Inglezakis, M. D. Loizidou and H. P. Grigoropoulou, Desalination, 214, 1 (2007).
M. K. Doula, Water Res., 40, 3167 (2006).
S. K. Ouki and M. Kavannagh, Waste Manage. Res., 15, 383 (1997).
G. Eisenman, Biophysical J., 2, 259 (1962).
U. Wingenfelder, B. Nowack, G. Furrer and R. Schulin, Water Res., 39, 3287 (2005).
L. Curkovic, Š. Cerjan-Stefanovic and T. Filipan, Water Res., 31, 1379 (1997).
V. J. Inglezakis, K. J. Hadjiandreou, M. D. Loizidou and H. P. Grigoropoulou, Water Res., 35, 2161 (2001).
S. CerjanStefanovic, L. Curkovic and T. Filipan, Croat. Chem., Acta, 281 (1996).
I. Langmuir, J. Am. Chem. Soc., 40, 1361 (1918).
H. Freundlich, Z. Phys. Chem., 57, 385 (1906).
F. H. Frimmel and L. Huber, Environ. Int., 22, 507 (1996).
G. McKay, M. S. Otterburn and A. G. Sweeney, Water Res., 14, 21 (1980).
E. Gutiérrez-Segura, M. Solache-Ríos, A. Colín-Cruz and C. Fall, J. Environ. Manage., 97, 6 (2012).
M. I. Tempkin and V. Pyzhev, Acta Phys. Chim., 12, 327 (1940).
A. Dada, A. Olalekan, A. Olatunya and O. Dada, J. Appl. Chem., 3, 38 (2012).
A. Günay, E. Arslankaya and I. Tosun, J. Hazard. Mater., 146, 362 (2007).
A. Dabrowski, Adv. Colloid Interface Sci., 93, 135 (2001).
J. Peric, M. Trgo and N. Vukojevic Medvidovic, Water Res., 38, 1893 (2004).
J. Rouquerol, F. Rouquerol, P. Llewellyn, G. Maurin and K. S. Sing, Adsorption by powders and porous solids: principles, methodology and applications, Academic Press (2013).
M. M. Dubinin, Chem. Rev., 60, 235 (1960).
J. P. Hobson, The J. Phys. Chem., 73, 2720 (1969).
F. A. Pavan, S. L. P. Dias, E. C. Lima and E. V. Benvenutti, Dyes Pigm., 76, 64 (2008).
V. Vimonses, S. Lei, B. Jin, C. W. K. Chow and C. Saint, Chem. Eng. J., 148, 354 (2009).
D. Georgiev, B. Bogdanov, Y. Hristov and I. Markovska, World Academy of Sci., Eng. Technol., 6, 1098 (2012).
R. Han, W. Zou, Z. Zhang, J. Shi and J. Yang, J. Hazard. Mater., 137, 384 (2006).
K. K. Wong, C. K. Lee, K. S. Low and M. J. Haron, Process Biochemistry, 39, 437 (2003).
F. N. Acar and Z. Eren, J. Hazard. Mater., 137, 909 (2006).
M. Sciban, M. Klasnja and B. Skrbic, Wood Sci. Technol., 40, 217 (2006).
S. Shukla and R. S. Pai, Sep. Purif. Technol., 43, 1 (2005).
Q. Li, J. Zhai, W. Zhang, M. Wang and J. Zhou, J. Hazard. Mater., 141, 163 (2007).
M. Horsfall, A. A. Abia and A. I. Spiff, Bioresour. Technol., 97, 283 (2006).
S. Shukla and R. S. Pai, Bioresour. Technol., 96, 1430 (2005).
N. Chubar, J. R. Carvalho and M. J. N. Correia, Colloids Surf., A: Physicochem. Eng. Aspects., 238, 51 (2004).
B. Nasernejad, T. E. Zadeh, B. B. Pour, M. E. Bygi and A. Zamani, Process Biochem., 40, 1319 (2005).
E. Pehlivan, S. Cetin and B. Yanik, J. Hazard. Mater., 135, 193 (2006).
D. Wankasi, M. Horsfall Jnr and A. Ibuteme Spiff, Electronic J Biotechnol., 9, 587 (2006).
C. Namasivayam and K. Kadirvelu, Chemosphere, 34, 377 (1997).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Irannajad, M., Haghighi, H.K. & Safarzadeh, E. Kinetic, thermodynamic and equilibrium studies on the removal of copper ions from aqueous solutions by natural and modified clinoptilolites. Korean J. Chem. Eng. 33, 1629–1639 (2016). https://doi.org/10.1007/s11814-015-0299-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-015-0299-0