Abstract
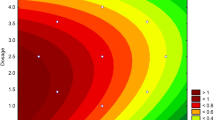
Al-Fe (hydr)oxides with different Al/Fe molar ratios (4:1, 1:1, 1:4, 0:1) were prepared using a coprecipitat method and were then employed for simultaneous removal of arsenate and fluoride. The 4Al: Fe was superior to other adsorbents for removal of arsenate and fluoride in the pH range of 5.0–9.0. The adsorption capacity of the Al-Fe (hydr)oxides for arsenate and fluoride at pH 6.5±0.3 increased with increasing Al content in the adsorbents. The linear relationship between the amount of OH− released from the adsorbent and the amount of arsenate or fluoride adsorbent by 4Al: Fe indicated that the adsorption of arsenate and fluoride by Al-Fe (hydr)oxides was realized primarily through quantitative ligand exchange. Moreover, there was a very good correlation between the surface hydroxyl group densities of Al-Fe (hydr)oxides and their adsorption capacities for arsenate or fluoride. The highest adsorption capacity for arsenate and fluoride by 4Al : Fe is mainly ascribed to its highest surface hydroxyl group density besides its largest pHpzc. The dosage of adsorbent necessary to remove arsenate and fluoride to meet the drinking water standard was mainly determined by the presence of fluoride since fluoride was generally present in groundwater at much higher concentration than arsenate.
Similar content being viewed by others
References
Chouhan S, Flora S J S. Arsenic and fluoride: two major ground water pollutants. Indian Journal of Experimental Biology, 2010, 48 (7): 666–678
Wu C X, Gu X L, Ge Y M, Zhang J H, Wang J D. Effects of high fluoride and arsenic on brain biochemical indexes and learning-memory in rats. Fluoride, 2006, 39(4): 274–279
Amini M, Abbaspour K C, Berg M, Winkel L, Hug S J, Hoehn E, Yang H, Johnson C A. Statistical modeling of global geogenic arsenic contamination in groundwater. Environmental Science & Technology, 2008, 42(10): 3669–3675
Amini M, Mueller K, Abbaspour K C, Rosenberg T, Afyuni M, Møller K N, Sarr M, Johnson C A. Statistical modeling of global geogenic fluoride contamination in groundwaters. Environmental Science & Technology, 2008, 42(10): 3662–3668
Armienta M A, Segovia N. Arsenic and fluoride in the groundwater of Mexico. Environmental Geochemistry and Health, 2008, 30(4): 345–353
Tang Y L, Guan X H, Su T Z, Gao N Y, Wang J M. Fluoride adsorption onto activated alumina: modeling the effects of pH and some competing ions. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2009, 337(1–3): 33–38
Warren C, Burgess W G, Garcia M G. Hydrochemical associations and depth profiles of arsenic and fluoride in Quaternary loess aquifers of northern Argentina. Mineralogical Magazine, 2005, 69 (5): 877–886
Wang Y, Shvartsev S L, Su C. Genesis of arsenic/fluoride-enriched soda water: a case study at Datong, northern China. Applied Geochemistry, 2009, 24(4): 641–649
Farooqi A, Masuda H, Firdous N. Toxic fluoride and arsenic contaminated groundwater in the Lahore and Kasur districts, Punjab, Pakistan and possible contaminant sources. Environmental Pollution, 2007, 145(3): 839–849
Farooqi A, Masuda H, Kusakabe M, Naseem M, Firdous N. Distribution of highly arsenic and fluoride contaminated groundwater from east Punjab, Pakistan, and the controlling role of anthropogenic pollutants in the natural hydrological cycle. Geochemical Journal, 2007, 41(4): 213–234
Farooqi A, Masuda H, Siddiqui R, Naseem M. Sources of arsenic and fluoride in highly contaminated soils causing groundwater contamination in Punjab, Pakistan. Archives of Environmental Contamination and Toxicology, 2009, 56(4): 693–706
Gomez M L, Blarasin M T, Martinez D E. Arsenic and fluoride in a loess aquifer in the central area of Argentina. Environmental Geology, 2009, 57(1): 143–155
Smedley P L, Zhang M, Zhang G, Luo Z. Mobilisation of arsenic and other trace elements in fluviolacustrine aquifers of the Huhhot Basin, Inner Mongolia. Applied Geochemistry, 2003, 18(9): 1453–1477
Wang G Q, Huang Y Z, Xiao B Y, Qian X C, Yao H, Hu Y, Gu Y L, Zhang C, Liu K T. Toxicity from water containing arsenic and fluoride in Xinjiang. Fluoride, 1997, 30(2): 81–84
Zhu C, Bai G, Liu X, Li Y. Screening high-fluoride and high-arsenic drinking waters and surveying endemic fluorosis and arsenism in Shaanxi Province in western China. Water Research, 2006, 40(16): 3015–3022
MHPRC. Standards for Dinking Water Quality. Beijing: Ministry of Health of the People’s Republic of China, 2007. Available online at http://www.moh.gov.cn/open/db_query/new/20070628143525.pdf. (accessed April 5, 2013)
Devi R, Alemayehu E, Singh V, Kumar A, Mengistie E. Removal of fluoride, arsenic and coliform bacteria by modified homemade filter media from drinking water. Bioresource Technology, 2008, 99(7): 2269–2274
Mlilo T B, Brunson L R, Sabatini D A. Arsenic and fluoride removal using simple materials. Journal of Environmental Engineering, 2010, 136(4): 391–398
Pinon-Miramontes M, Bautista-Margulis R G, Perez-Hernandez A. Removal of arsenic and fluoride from drinking water with cake alum and a polymeric anionic flocculent. Fluoride, 2003, 36(2): 122–128
Padilla A P, Saitua H. Performance of simultaneous arsenic, fluoride and alkalinity (bicarbonate) rejection by pilot-scale nanofiltration. Desalination, 2010, 257(1–3): 16–21
Zhao X, Zhang B, Liu H, Qu J. Simultaneous removal of arsenite and fluoride via an integrated electro-oxidation and electrocoagulation process. Chemosphere, 2011, 83(5): 726–729
Ingallinella A M, Pacini V A, Fernández R G, Vidoni R M, Sanguinetti G. Simultaneous removal of arsenic and fluoride from groundwater by coagulation-adsorption with polyaluminum chloride. Journal of Environmental Science and Health Part A, 2011, 46 (11): 1288–1296
Deng S, Liu H, Zhou W, Huang J, Yu G. Mn-Ce oxide as a high-capacity adsorbent for fluoride removal from water. Journal of Hazardous Materials, 2011, 186(2–3): 1360–1366
Pan Y F, Chiou C T, Lin T F. Adsorption of arsenic(V) by iron-oxide-coated diatomite (IOCD). Environmental Science and Pollution Research International, 2010, 17(8): 1401–1410
Ren Z, Zhang G, Chen J P. Adsorptive removal of arsenic from water by an iron-zirconium binary oxide adsorbent. Journal of Colloid and Interface Science, 2011, 358(1): 230–237
Biswas K, Saha S K, Ghosh U C. Adsorption of fluoride from aqueous solution by a synthetic Iron(III)-Aluminum(III) mixed oxide. Industrial & Engineering Chemistry Research, 2007, 46(16): 5346–5356
Hong H J, Farooq W, Yang J S, Yang J W. Preparation and evaluation of Fe-Al binary oxide for arsenic removal: comparative study with single metal oxides. Separation Science and Technology, 2010, 45(12–13): 1975–1981
Masue Y, Loeppert R H, Kramer T A. Arsenate and arsenite adsorption and desorption behavior on coprecipitated aluminum: iron hydroxides. Environmental Science & Technology, 2007, 41(3): 837–842
Sujana MG, Anand S. Iron and aluminium based mixed hydroxides: a novel sorbent for fluoride removal from aqueous solutions. Applied Surface Science, 2010, 256(23): 6956–6962
Sujana M G, Soma G, Vasumathi N, Anand S. Studies on fluoride adsorption capacities of amorphous Fe/Al mixed hydroxides from aqueous solutions. Journal of Fluorine Chemistry, 2009, 130(8): 749–754
Tamura H, Tanaka A, Mita K, Furuichi R. Surface hydroxyl site densities on metal oxides as a measure for the ion-exchange capacity. Journal of Colloid and Interface Science, 1999, 209(1): 225–231
Jain A, Raven K P, Loeppert R H. Arsenite and arsenate adsorption on ferrihydrite: surface charge reduction and net OH− release stoichiometry. Environmental Science & Technology, 1999, 33(8): 1179–1184
Schwertmann U, Taylor RM. Iron Oxides. In: Dixon J B, Weed S B, Dinauer R C, eds. Minerals in Soil Environments. Madison WI: Soil Science Society of America, 1989
Krokidis X, Raybaud P, Gobichon A E, Rebours B, Euzen P, Toulhoat H. Theoretical study of the dehydration process of boehmite to gamma-alumina. Journal of Physical Chemistry B, 2001, 105(22): 5121–5130
Liu Y T, Hesterberg D. Phosphate bonding on noncrystalline Al/Fe-hydroxide coprecipitates. Environmental Science & Technology, 2011, 45(15): 6283–6289
Guan X H, Wang J M, Chusuei C C. Removal of arsenic from water using granular ferric hydroxide: macroscopic and microscopic studies. Journal of Hazardous Materials, 2008, 156(1–3): 178–185
Li Y H, Wang S G, Cao A Y, Zhao D, Zhang X F, Xu C L, Luan Z K, Ruan D B, Liang J, Wu D H, Wei B Q. Adsorption of fluoride from water by amorphous alumina supported on carbon nanotubes. Chemical Physics Letters, 2001, 350(5–6): 412–416
George S, Pandit P, Gupta A B. Residual aluminium in water defluoridated using activated alumina adsorption—modeling and simulation studies. Water Research, 2010, 44(10): 3055–3064
Mohapatra M, Rout K, Singh P, Anand S, Layek S, Verma H C, Mishra B K. Fluoride adsorption studies on mixed-phase nano iron oxides prepared by surfactant mediation-precipitation technique. Journal of Hazardous Materials, 2011, 186(2–3): 1751–1757
Stumm W, Morgan J J. Aquatic Chemistry: Chemical Equilibria and Rates in NaturalWaters, 3rd ed. New York: JohnWiley & Sons, Inc, 1996
Kumar V, Talreja N, Deva D, Sankararamakrishnan N, Sharma A, Verma N. Development of bi-metal doped micro- and nano multi-functional polymeric adsorbents for the removal of fluoride and arsenic(V) from wastewater. Desalination, 2011, 282SI: 27–38
Guan X H. Adsorption of phosphates and organic acids on aluminum hydroxide in aquatic environment-mechanisms and interactions. Dissertation for the Doctoral Degree. Hong Kong: Hong Kong University of Science and Technology, 2005
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qiao, J., Cui, Z., Sun, Y. et al. Simultaneous removal of arsenate and fluoride from water by Al-Fe (hydr)oxides. Front. Environ. Sci. Eng. 8, 169–179 (2014). https://doi.org/10.1007/s11783-013-0533-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11783-013-0533-0