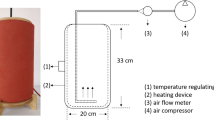
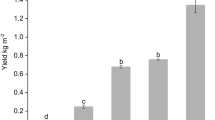
Abstract
Previously, elicitation using microbe consortium inside Ragi Tape (RT) has given some insight with better results than mono-starter elicitation. However, the exact microbe species involved during elicitation remain unknown. This research aimed to identify the microbe community structure before and after elicitation using RT. Soybean (SB) and Mung Bean (MB) were used as an elicitation substrate to evaluate whether different beans affected the microbial community structure. To understand the effect of elicitation on the phytochemical profile, phytochemical content was analyzed using Liquid Chromatography-High Resolution Mass Spectrometry (LC-HRMS) analysis. Fungal and bacterial species were identified using Illumina MiSeq platform and analyzed using the QIIME2 pipeline. As a result, at the species level, Saccharomycopsis fibuligera was found to be dominant fungi after elicitation occurred in both SB and MB, while Bacillus sp. and Enterobacter sp. in SB and Pantoea sp., Enterobacter sp., and Acinetobacter radioresistens in MB become the most abundant taxa from the bacterial group. Alpha diversity showed that bacteria were more abundant than fungi, while RT had greater diversity than the microbial community in post-elicitation (both SB and MB). However, beta diversity analysis revealed that both SB and MB have similar community structures. There was no significant effect of different substrates on the microbial community structure after elicitation using RT. On the other hand, the phytochemical profile was differed between the original and elicited SB and MB. In conclusion, RT has good potential as a starter of elicitation with its feasibility and availability in increasing the secondary metabolites content for general application purposes.




Similar content being viewed by others
Abbreviations
- RT:
-
Ragi Tape, pre-elicitation
- SB:
-
Soybean, post-elicitation
- MB:
-
Mungbean, post-elicitation
- LC-HRMS:
-
Liquid Chromatography-High Resolution Mass Spectrometry
- NGS:
-
Next-Generation Sequencing
- OTUs:
-
Operational Taxonomic Units
- PCoA:
-
Principal Coordinates Analysis
- OMB:
-
Original Mungbean
- EMB:
-
Elicited Mungbean
- OSB:
-
Original Soybean
- ESB:
-
Elicited Soybean
References
Aisyah S, Gruppen H, Andini S et al (2016) Variation in accumulation of isoflavonoids in Phaseoleae seedlings elicited by Rhizopus. Food Chem 196:694–701. https://doi.org/10.1016/j.foodchem.2015.09.110
Algar E, Ramos-Solano B, García-Villaraco A et al (2013) Bacterial bioeffectors modify bioactive profile and increase Isoflavone content in soybean sprouts (Glycine max var Osumi). Plant Foods Hum Nutr 68:299–305. https://doi.org/10.1007/s11130-013-0373-x
Atho’illah MF, Safitri YD, Nur’aini FD, et al (2019) Evaluation of Glyceollin accumulation and antioxidant properties on soybean (glycine max L.) through combination of different biotic elicitor and light. Scientific Study & Research Chemistry & chemical engineering, biotechnology, food industry; Bacau 20:199–208. Doi: 10.13140/RG.2.2.19614.28483
Beck JM, Young VB, Huffnagle GB (2012) The microbiome of the lung. Transl Res 160:258–266. https://doi.org/10.1016/j.trsl.2012.02.005
Bertrand S, Bohni N, Schnee S et al (2014) Metabolite induction via microorganism co-culture: a potential way to enhance chemical diversity for drug discovery. Biotechnol Adv 32:1180–1204. https://doi.org/10.1016/j.biotechadv.2014.03.001
Bhutani N, Maheshwari R, Kumar P, Suneja P (2021) Bioprospecting of endophytic bacteria from nodules and roots of Vigna radiata. Vigna unguiculata and Cajanus cajan for their potential use as bioinoculants Plant Gene 100326. https://doi.org/10.1016/j.plgene.2021.100326
Bokulich NA, Kaehler BD, Rideout JR et al (2018) Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2’s q2-feature-classifier plugin. Microbiome 6:90. https://doi.org/10.1186/s40168-018-0470-z
Bolyen E, Rideout JR, Dillon MR et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Callahan BJ, McMurdie PJ, Rosen MJ et al (2016) DADA2: high-resolution sample inference from Illumina amplicon data. Nat Methods 13:581–583. https://doi.org/10.1038/nmeth.3869
Cao Y, Fanning S, Proos S, et al (2017) A Review on the Applications of Next Generation Sequencing Technologies as Applied to Food-Related Microbiome Studies Front Microbiol 8:. doi: 10.3389/fmicb.2017.01829
Caporaso JG, Lauber CL, Walters WA et al (2012) Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J 6:1621–1624. https://doi.org/10.1038/ismej.2012.8
Chi Z, Chi Z, Liu G et al (2009) Saccharomycopsis fibuligera and its applications in biotechnology. Biotechnol Adv 27:423–431. https://doi.org/10.1016/j.biotechadv.2009.03.003
Costa EM da, Carvalho F de, Nóbrega RSA, et al (2016) Bacterial strains from floodplain soils perform different plant-growth promoting processes and enhance cowpea growth. Sci agric (Piracicaba, Braz) 73:301–310. doi: 10.1590/0103-9016-2015-0294
Dwidjoseputro D, Wolf FT (1970) Microbiological studies of Indonesian fermented foodstuffs. Mycopathol Mycol Appl 41:211–222. https://doi.org/10.1007/BF02051099
Farrell K, Jahan MA, Kovinich N (2017) Distinct mechanisms of biotic and chemical elicitors enable additive elicitation of the anticancer Phytoalexin Glyceollin I. Molecules 22. https://doi.org/10.3390/molecules22081261
Halder M, Sarkar S, Jha S (2019) Elicitation: a biotechnological tool for enhanced production of secondary metabolites in hairy root cultures. Eng Life Sci 19:880–895. https://doi.org/10.1002/elsc.201900058
Hunter JD (2007) Matplotlib: a 2D graphics environment. Comput Sci Eng 9:90–95. https://doi.org/10.1109/MCSE.2007.55
Hussain M, Fareed S, Ansari S et al (2012) Current approaches toward production of secondary plant metabolites. J Pharm Bioallied Sci 4:10–20. https://doi.org/10.4103/0975-7406.92725
Isah T (2019) Stress and defense responses in plant secondary metabolites production. Biol Res 52:39. https://doi.org/10.1186/s40659-019-0246-3
Jain S, Vaishnav A, Kumari S et al (2017) Chitinolytic Bacillus-mediated induction of Jasmonic acid and defense-related proteins in soybean (Glycine max L. Merrill) plant against Rhizoctonia solani and fusarium oxysporum. J Plant Growth Regul 36:200–214. https://doi.org/10.1007/s00344-016-9630-1
Jeandet P (2015) Phytoalexins: current Progress and future prospects. Molecules 20:2770–2774. https://doi.org/10.3390/molecules20022770
Kim HJ, Suh H-J, Kim JH et al (2010a) Estrogenic activity of glyceollins isolated from soybean elicited with aspergillus sojae. J Med Food 13:382–390. https://doi.org/10.1089/jmf.2009.1281
Kim HJ, Suh H-J, Lee CH et al (2010b) Antifungal activity of Glyceollins isolated from soybean elicited with aspergillus sojae. J Agric Food Chem 58:9483–9487. https://doi.org/10.1021/jf101694t
Li JH, Wang ET, Chen WF, Chen WX (2008) Genetic diversity and potential for promotion of plant growth detected in nodule endophytic bacteria of soybean grown in Heilongjiang province of China. Soil Biol Biochem 40:238–246. https://doi.org/10.1016/j.soilbio.2007.08.014
Namdeo A (2007) Plant cell elicitation for production of secondary metabolites: a review. Pharmacogn Rev 1:69–79
Pereira M, Morais L, Marques E et al (2019) Humic substances and efficient microorganisms: elicitation of medicinal plants—a review. J Agric Sci 11:268. https://doi.org/10.5539/jas.v11n7p268
Pluskal T, Castillo S, Villar-Briones A, Orešič M (2010) MZmine 2: modular framework for processing, visualizing, and analyzing mass spectrometry-based molecular profile data. BMC Bioinformatics 11:395. https://doi.org/10.1186/1471-2105-11-395
Radman R, Saez T, Bucke C, Keshavarz T (2003) Elicitation of plants and microbial cell systems. Biotechnol Appl Biochem 37:91–102. https://doi.org/10.1042/ba20020118
Ramirez-Estrada K, Vidal-Limon H, Hidalgo D, et al (2016) Elicitation, an effective strategy for the biotechnological production of bioactive high-added value compounds in plant cell factories. Molecules 21:. doi: 10.3390/molecules21020182
Ranf S (2016) Immune sensing of lipopolysaccharide in plants and animals: same but different. PLoS Pathog 12. https://doi.org/10.1371/journal.ppat.1005596
Seca AML, Pinto DCGA (2019) Biological potential and medical use of secondary metabolites. Medicines (Basel) 6. https://doi.org/10.3390/medicines6020066
Shakya P, Marslin G, Siram K et al (2019) Elicitation as a tool to improve the profiles of high-value secondary metabolites and pharmacological properties of Hypericum perforatum. J Pharm Pharmacol 71:70–82. https://doi.org/10.1111/jphp.12743
Sieuwerts S, de Bok FAM, Hugenholtz J, van Hylckama Vlieg JET (2008) Unraveling microbial interactions in food fermentations: from classical to genomics approaches. Appl Environ Microbiol 74:4997–5007. https://doi.org/10.1128/AEM.00113-08
Smid EJ, Lacroix C (2013) Microbe-microbe interactions in mixed culture food fermentations. Curr Opin Biotechnol 24:148–154. https://doi.org/10.1016/j.copbio.2012.11.007
Soliman T, Yang S-Y, Yamazaki T, Jenke-Kodama H (2017) Profiling soil microbial communities with next-generation sequencing: the influence of DNA kit selection and technician technical expertise. PeerJ 5. https://doi.org/10.7717/peerj.4178
Sujaya IN, Amachi S, Yokota A et al (2001) Identification and characterization of lactic acid bacteria in ragi tape. World J Microbiol Biotechnol 17:349–357. https://doi.org/10.1023/A:1016642315022
Świeca M (2015) Elicitation with abiotic stresses improves pro-health constituents, antioxidant potential and nutritional quality of lentil sprouts. Saudi J Biol Sci 22:409. https://doi.org/10.1016/j.sjbs.2014.12.007
Thakur M, Bhattacharya S, Khosla PK, Puri S (2019) Improving production of plant secondary metabolites through biotic and abiotic elicitation. J Appl Res Med Aromat Plants 12:1–12. https://doi.org/10.1016/j.jarmap.2018.11.004
Vaishnav P, Demain AL (2011) Unexpected applications of secondary metabolites. Biotechnol Adv 29:223–229. https://doi.org/10.1016/j.biotechadv.2010.11.006
Vasconsuelo A, Boland R (2007) Molecular aspects of the early stages of elicitation of secondary metabolites in plants. Plant Sci 172:861–875. https://doi.org/10.1016/j.plantsci.2007.01.006
Walterson AM, Stavrinides J (2015) Pantoea: insights into a highly versatile and diverse genus within the Enterobacteriaceae. FEMS Microbiol Rev 39:968–984. https://doi.org/10.1093/femsre/fuv027
Zhao L, Xu Y, Lai X (2018) Antagonistic endophytic bacteria associated with nodules of soybean (Glycine max L.) and plant growth-promoting properties. Braz J Microbiol 49:269–278. https://doi.org/10.1016/j.bjm.2017.06.007
Acknowledgments
We would like to thank all of qiime2 developers, particularly Michael Scott Robeson (University of Arkansas for Medical Sciences), for providing trained SILVA classifiers in the qiime2 forum.
Authors’ Contributions.
FEH = performing experiment, original draft preparation; WW = performing LC-HRMS analysis, writing-review and editing; MR = data analysis, writing-review and editing; NW = funding acquisition, supervision; YDJ = conceptualization, writing-review and editing.
Funding
This research was funded by the Ministry of Research and Technology, the Republic of Indonesia, as research funding scheme of PMDSU scholarship program with contract no. 1243.6/UN10.C10/PN/2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no potential conflict of interest.
Ethical approval
No ethical clearance needed due to no animal involvement in this study.
Informed consent
This study does not include human as a subject.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 71 kb)
Rights and permissions
About this article
Cite this article
Hermanto, F.E., Warsito, W., Rifa’i, M. et al. Unveiling microbial community structure in Ragi tape as elicitors to increase secondary metabolites contents in Glycine max and Vigna radiata. Biologia 76, 3901–3907 (2021). https://doi.org/10.1007/s11756-021-00917-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-021-00917-4