Abstract
Key message
Cocksfoot shows intra-specific variability in its response to constant temperature during germination.
Abstract
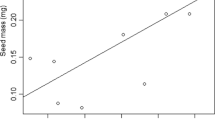
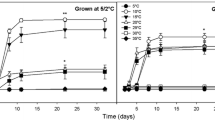
Temperature, in conjunction with other environmental factors, controls plant development, including seed germination and its timing, which directly affects seedling survival. Indirectly, seed germination affects recruitment of individuals and hence the genetic dynamics of grasslands communities. Cocksfoot (Dactylis glomerata L.) is a major species growing in temperate and Mediterranean grasslands. Six accessions of D. glomerata, including four natural populations and two commercial varieties were evaluated for their responses to eight constant temperatures, from 5 to 40 ℃, with increases by 5 ℃. Four replicates of one-hundred seeds per accession were put in the dark and counted for germination at variable frequencies and duration that depended on temperature treatments. We estimated maximum germination percentage, maximum germination rate as well as apparent initial time and time when 50% of seed germinate. The F test showed significant difference (P < 0.01) between the response curves of various accessions. At least four types of responses can be distinguished. Temperature for maximum germination ranged from 5 to 25 ℃, depending on accessions. The results of this study reveal the existence of intraspecific genetic variability during germination in D. glomerata for the response to temperature. Such variability could be used to select new varieties adapted to thermal conditions induced by ongoing climate change.





Similar content being viewed by others
References
Ahmed LQ, Escobar-Gutérrez AJ (2022a) Unexpected intraspecific variability of perennial ryegrass (Lolium perenne L.) in response to constant temperature during germination and initial heterotrophic growth. Front Plant Sci 13:856099. https://doi.org/10.3389/fpls.2022a.856099
Ahmed LQ, Escobar-Gutérrez AJ (2022b) Tall fescue (Festuca arundinacea Schreb.) shows intraspecific variability in response to temperature during germination. Agronomy. https://doi.org/10.3390/agronomy12051245
Ahmed LQ, Durand JL, Escobar-Gutérrez AJ (2019) Genetic diversity of alfalfa (Medicago sativa) in response to temperature during germination. Seed Sci Technol. 47(3):351–356. https://doi.org/10.15258/sst.2019.47.3.10
Baskin CC, Baskin JM (2014) Seeds: ecology, biogeography, and evolution of dormancy and germination, 2nd edn. Academic Press, San Diego (eBook ISBN: 9780124166837)
Bewley JD (1997) Seed germination and dormancy. Plant Cell 9(7):1055–1066. https://doi.org/10.1105/tpc.9.7.1055
Bewley JD, Black M (1994) Seeds physiology of development and germination, 2nd edn. Plenum Press, New York London. https://doi.org/10.1007/978-1-4899-1002-8 (ISBN 978-1-4899-1002-8)
Black AD, Moot DJ, Lucas RJ (2006) Development and growth characteristics of Caucasian and white clover seedlings, compared with perennial ryegrass. Grass Forage Sci 61:442–453. https://doi.org/10.1111/j.1365-2494.2006.00553.x
Boller B, Greene SL (2010) Genetic resources. Fodder Crop and Amenity Grasses. Hand book of Plant Breeding 5edn by: Boller B, Posselt UK, Veronesi F. New York, USA. Pp 13–37. https://link.springer.com/book/10.1007%2F978-1-4419-0760-8. https://doi.org/10.1007/978-1-4419-0760-8
Bretagnolle F, Thompson JD, Lumaret R (1995) The influence of seed size variation on seed germination and seedling vigour in diploid and tetraploid Dactylis glomerata L. Ann Bot 76:607–615. https://doi.org/10.1006/anbo.1995.1138
Charlton JFL, Hampton JG, Scott DJ (1986) Temperature effects on germination of New Zealand herbage grasses. In: Proceedings of the New Zealand Grassland Association. 47: 165–167.
Finch-Savage WE, Steckel JRA, Phelps K (1998) Germination and post-germination growth to carrot seedling emergence: predictive threshold models and sources of variation between sowing occasions. New Phytol 139:505–516. https://doi.org/10.1046/j.1469-8137.1998.00208.x
Ghaleb W, Ahmed L, Escobar-Gutiérrez AJ, Julier B (2021) The history of domestication and selection of lucerne: a new perspective from the genetic diversity for seed germination in response to temperature and scarification. Front Plant Sci 11:578121. https://doi.org/10.3389/fpls.2020.578121
Last L, Widmer F, Fjellstad W, Stoyanova S, Kölliker R (2013) Genetic diversity of natural orchardgrass (Dactylis glomerata L.) populations in three regions in Europe. BioMed Central Genetics 14:1471–2156
Lu H, Shen J, Jin X, Hannaway DB, Daly C, Halbleib MD (2008) Determining optimal seeding times for tall fescue using germination studies and spatial climate analysis. Agric for Meteorol 148:931–941. https://doi.org/10.1016/j.agrformet.2008.01.004
Mara FPO (2012) The role of grassland in food security and climate change. Ann Bot 110:1263–1270. https://doi.org/10.1093/aob/mcs209
Monks DP, SadatAsilan K, Moot DJ (2009) Cardinal temperatures and thermal time requirements for germination of annual and perennial temperate pasture species. Agronomy New Zealand. 39:95–109
Moot DJ, Scott WR, Roy AM, Nicholls AC (2000) Base temperature and thermal time requirements for germination and emergence of temperature pasture species. N Z J Agric Res 43:15–25. https://doi.org/10.1080/00288233.2000.9513404
Mousset C (2000) Rassemblement, utilisation et gestion des ressources génétiques de dactyle a l’INRA de Lusignan. Fourrages. 162:121–139
Norton MR (2015) Increasing the seed production of summer-dormant, drought-tolerant cocksfoot. Agronomy. Proceedings of the 17th ASA Conference, 20–24 September 2015. pp 1034.
Palazzo AJ, Brar G (1997) The effects of temperature on germination of eleven Festuca cultivars. USA cold region research and engineering laboratory. Special Report. 97–19.
Pannangpetch K, Bean EW (1984) Effects of temperature on germination in populations of Dcatylis glomerata from NW Spain and Central Italy. Ann Bot 53:633–639. https://doi.org/10.3390/agronomy9090533
Probert RJ, Smith RD, Birch P (1985a) Germination responses to light and alternating temperature in European populations of Dactylis glomerata L. II. the genetic and environmental components of germination. New Phytol 99(2):317–322. https://doi.org/10.1111/j.1469-8137.1985a.tb03659.x
Probert RJ, Smith RD, Birch P (1985b) Germination responses to light and alternating temperature in European populations of Dactylis glomerata L. I. variability in relation to origin. The New Phytologist. 99: 317–322. https://www.jstor.org/stable/2434404
Probert RJ, Smith RD, Birch P (1986) Germination responses to light and alternating temperatures in European populations of Dactylis glomerata L. V. the principle components of the alternating temperature requirement. The New Phytologist. 1(1):133–142. https://www.jstor.org/stable/2484988
Qiu J (2005) Quantifying the effects of temperature on dormancy change and germination in orchardgrass (Dactylis glomerata L.) and western wheatgrass (Pascopyrum smihii Rydb. L.). Master Thesis of Science, University of Saskatchewan, Canada. Pp 123.
Ranal M, Santana DG (2006) How and why to measure the germination process? Braz J Botany 29:1–11. https://doi.org/10.1590/S0100-84042006000100002
Roundy BA, Biedenbender SH (1996) Germination of warm season grasses under constant and dynamic temperatures. Journal Range Manage. 49: 42–431. https://www.jstor.org/stable/4002924
Sachs J (1860) Die Abhängigkeit de Keimgung von Der Temperatur. Jahrb Für Wiss Bot 2:338–377
Shafii BS, Price WJ (2001) Estimation of cardinal temperatures in germination data analysis. J Agric Biol Environ Stat 6:356–366. https://doi.org/10.1198/108571101317096569
Shen JB, Xu LY, Jin XQ, Chen JH, Lu HF (2008) Effect of temperature regime on germinations of seed of perennial ryegrass (Lolium perenne). Grass Forage Sci 63:249–256. https://doi.org/10.1111/j.1365-2494.2008.00630.x
Stanisavljevic R, Ðjokic D, Milenkovic J, Ðukanovic L, Stevovic V, Simic A, Dodig D (2011) Seed germination and seedling vigour of Italian ryegrass, cocksfoot and timothy following harvest and storage. Ciência e Agrotecnologia 35:1141–1148. https://doi.org/10.1590/S1413-70542011000600014
Stewart AV, Ellison NW (2011) The genus Dactylis. In: Kole C (ed) Wild crop relatives: genomic and breeding resources, millets and grasses. Springer, New York, pp 73–87
Thornley JHM, Sutcliffe JF, Mahlberg P (1976) Mathematical models in plant physiology. Academic Press, London
Tribouillois H (2014) Caractérisation fonctionnelle d'espèces utilisées en cultures intermédiaires et analyse de leurs performances en mélanges bi-spécifiques pour produire des services écosystémiques de gestion de l'azote. PhD, Thèse, Université de Toulouse, France. pp 219.
Trudgill DL, Squire GR, Thompson K (2000) A thermal time basis for comparing the germination requirements of some British herbaceous plants. New Phytol 145:107–114. https://doi.org/10.1046/j.1469-8137.2000.00554.x
Volaire F, Lelièvre F (2001) Drought survival in Dactylis glomerata and Festuca arundinacea under similar rooting conditions in tubes. Plant Soil 229:225–234. https://doi.org/10.1023/A:1004835116453
Wang H, Cutforth H, McCaig T, McLeod G, Brandt K, Lemke R, Goddard T, Sprout C (2009) Predicting the time to 50% seedling emergence in wheat using a beta model NJAS-Wageningen. J Life Sci 57:65–71. https://doi.org/10.1016/j.njas.2009.07.003
Yin X, Kropff MJ, McLaren G, Visperas RM (1995) A nonlinear model for crop development as a function of temperature. Agric for Meteorol 77:1–16. https://doi.org/10.1016/0168-1923(95)02236-Q
Zeng B, Zhang XQ, Lan Y, Yang WY (2008) Evaluation of genetic diversity and relationships in orchardgrass (Dactylis glomerata L.) germplasm based on SRAP markers. Can J Plant Sci 88(1):53–60. https://doi.org/10.4141/CJPS07017
Zhang H, McGill CR, Irving LJ, Kemp PD, Zhou D (2013) A modified thermal time model to predict germination rate of ryegrass and tall fescue at constant temperature. Crop Sci 53:240–249. https://doi.org/10.2135/cropsci2012.02.0085
Acknowledgements
The authors thank the reviewers and the editor for their constructive comments and suggestions.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by A. Gniazdowska-Piekarska.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ahmed, L.Q., Escobar-Gutiérrez, A.J. Analysis of intra-specific variability of cocksfoot (Dactylis glomerata L.) in response to temperature during germination. Acta Physiol Plant 44, 117 (2022). https://doi.org/10.1007/s11738-022-03439-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-022-03439-x