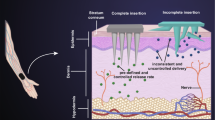
Abstract
A glucose-mediated drug delivery system would be highly satisfactory for diabetes diagnosis since it can intelligently release drug based on blood glucose levels. Herein, a glucose-responsive drug delivery system by integrating glucose-responsive poly(3-acrylamidophenylboronic acid) (PAPBA) functionalized hollow mesoporous silica nanoparticles (HMSNs) with transcutaneous microneedles (MNs) has been designed. The grafted PAPBA serves as gatekeeper to prevent drug release from HMSNs at normoglycemic levels. In contrast, faster drug release is detected at a typical hyperglycemic level, which is due to the change of hydrophilicity of PAPBA at high glucose concentration. After transdermal administration to diabetic rats, an effective hypoglycemic effect is achieved compared with that of subcutaneous injection. These observations indicate that the designed glucose-responsive drug delivery system has a potential application in diabetes treatment.
Similar content being viewed by others
References
Ogurtsova K, da Rocha Fernandes J D, Huang Y, et al. IDF Diabetes Atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Research and Clinical Practice, 2017, 128: 40–50
Tuomi T, Santoro N, Caprio S, et al. The many faces of diabetes: a disease with increasing heterogeneity. Lancet, 2014, 383(9922): 1084–1094
Owens D R, Zinman B, Bolli G B. Insulins today and beyond. Lancet, 2001, 358(9283): 739–746
Al Hayek A A, Robert A A, Al Dawish M A. Skin-related complications among adolescents with type 1 diabetes using insulin pump therapy. Clinical Medicine Insights: Endocrinology and Diabetes, 2018, 11: 1–5
Chantelau E, Spraul M, Mühlhauser I, et al. Long-term safety, efficacy and side-effects of continuous subcutaneous insulin infusion treatment for type 1 (insulin-dependent) diabetes mellitus: a one center experience. Diabetologia, 1989, 32(7): 421–426
Asche C V, Shane-McWhorter L, Raparla S. Health economics and compliance of vials/syringes versus pen devices: a review of the evidence. Diabetes Technology & Therapeutics, 2010, 12(S1): S101–S108
Derraik J G B, Rademaker M, Cutfield W S, et al. Poorer glycaemic control is associated with increased skin thickness at injection sites in children with type 1 diabetes. International Journal of Pediatric Endocrinology, 2014, 2014(1): 2
Fuchs J, Hovorka R. Closed-loop control in insulin pumps for type-1 diabetes mellitus: safety and efficacy. Expert Review of Medical Devices, 2020, 17(7): 707–720
Musolino G, Dovc K, Boughton C K, et al. Reduced burden of diabetes and improved quality of life: Experiences from unrestricted day-and-night hybrid closed-loop use in very young children with type 1 diabetes. Pediatric Diabetes, 2019, 20(6): 794–799
Croissant J G, Fatieiev Y, Khashab N M. Degradability and clearance of silicon, organosilica, silsesquioxane, silica mixed oxide, and mesoporous silica nanoparticles. Advanced Materials, 2017, 29(9): 1604634
Li Z, Barnes J C, Bosoy A, et al. Mesoporous silica nanoparticles in biomedical applications. Chemical Society Reviews, 2012, 41(7): 2590–2605
Yang K Y, Glemza R, Jarowski C I. Effects of amorphous silicon dioxides on drug dissolution. Journal of Pharmaceutical Sciences, 1979, 68(5): 560–565
Zhu J, Niu Y, Li Y, et al. Stimuli-responsive delivery vehicles based on mesoporous silica nanoparticles: recent advances and challenges. Journal of Materials Chemistry B: Materials for Biology and Medicine, 2017, 5(7): 1339–1352
Choi Y, Lee J E, Lee J H, et al. A biodegradation study of SBA-15 microparticles in simulated body fluid and in vivo. Langmuir, 2015, 31(23): 6457–6462
Chen Y, Chen H R, Shi J L. Construction of homogenous/heterogeneous hollow mesoporous silica nanostructures by silicaetching chemistry: Principles, synthesis, and applications. Accounts of Chemical Research, 2014, 47(1): 125–137
Jia X, He D G, Zhang A M, et al. DNA-functionalized hollow mesoporous silica nanoparticles with dual cargo loading for nearinfrared-responsive synergistic chemo-photothermal treatment of cancer cells. ACS Applied Nano Materials, 2018, 1(7): 3486–3497
Li Y, Li N, Pan W, et al. Hollow mesoporous silica nanoparticles with tunable structures for controlled drug delivery. ACS Applied Materials & Interfaces, 2017, 9(3): 2123–2129
Shen J, Song G, An M, et al. The use of hollow mesoporous silica nanospheres to encapsulate bortezomib and improve efficacy for non-small cell lung cancer therapy. Biomaterials, 2014, 35(1): 316–326
Zhang K, Chen H, Zheng Y, et al. A facile in situ hydrophobic layer protected selective etching strategy for the synchronous synthesis/modification of hollow or rattle-type silica nanoconstructs. Journal of Materials Chemistry, 2012, 22(25): 12553–12561
Wang X, Zhou L, Liu Y, et al. Transformation from single-mesoporous to dual-mesoporous organosilica nanoparticles. Nanoscale, 2017, 9(19): 6362–6369
Prajapati R, Gontsarik M, Yaghmur A, et al. pH-responsive nano-self-assemblies of the anticancer drug 2-hydroxyoleic acid. Langmuir, 2019, 35(24): 7954–7961
Zohreh N, Alipour S, Hosseini S H, et al. Natural salep/PEGylated chitosan double layer toward a more sustainable pH-responsive magnetite nanocarrier for targeted delivery of DOX and hyperthermia application. ACS Applied Nano Materials, 2019, 2(2): 853–866
He D, He X, Wang K, et al. Reversible stimuli-responsive controlled release using mesoporous silica nanoparticles functionalized with a smart DNA molecule-gated switch. Journal of Materials Chemistry, 2012, 22(29): 14715–14721
Du M, Chen Y, Tu J, et al. Ultrasound responsive magnetic mesoporous silica nanoparticle-loaded microbubbles for efficient gene delivery. ACS Biomaterials Science & Engineering, 2020, 6(5): 2904–2912
Lai J, Mu X, Xu Y, et al. Light-responsive nanogated ensemble based on polymer grafted mesoporous silica hybrid nanoparticles. Chemical Communications, 2010, 46(39): 7370–7372
Huang P, Zeng B, Mai Z, et al. Novel drug delivery nanosystems based on out-inside bifunctionalized mesoporous silica yolk-shell magnetic nanostars used as nanocarriers for curcumin. Journal of Materials Chemistry B: Materials for Biology and Medicine, 2016, 4(1): 46–56
Kim H, Kang Y J, Kang S, et al. Monosaccharide-responsive release of insulin from polymersomes of polyboroxole block copolymers at neutral pH. Journal of the American Chemical Society, 2012, 134(9): 4030–4033
Oroval M, Díez P, Aznar E, et al. Self-regulated glucose-sensitive neoglycoenzyme-capped mesoporous silica nanoparticles for insulin delivery. Chemistry, 2017, 23(6): 1353–1360
Wu Q, Wang L, Yu H, et al. Organization of glucose-responsive systems and their properties. Chemical Reviews, 2011, 111(12): 7855–7875
Yan J, Fang H, Wang B. Boronolectins and fluorescent boronolectins: An examination of the detailed chemistry issues important for the design. Medicinal Research Reviews, 2005, 25(5): 490–520
Zhang G, Zhang X, Shen H, et al. Smarter glucose-sensitivity of polymeric micelles formed from phenylborate ester-co-pyrenylboronic ester for insulin delivery at physiological pH. RSC Advances, 2014, 4(91): 49964–49973
Mo R, Jiang T, Di J, et al. Emerging micro- and nanotechnology based synthetic approaches for insulin delivery. Chemical Society Reviews, 2014, 43(10): 3595–3629
Yu W, Jiang G, Liu D, et al. Transdermal delivery of insulin with bioceramic composite microneedles fabricated by gelatin and hydroxyapatite. Materials Science and Engineering C, 2017, 73: 425–428
Hu Z, Meduri C S, Ingrole R S J, et al. Solid and hollow metallic glass microneedles for transdermal drug-delivery. Applied Physics Letters, 2020, 116(20): 203703
Jayaneththi V R, Aw K, Sharma M, et al. Controlled transdermal drug delivery using a wireless magnetic microneedle patch: Preclinical device development. Sensors and Actuators B: Chemical, 2019, 297: 126708
Kim D, Kim H, Lee P C W, et al. Universally applicable RNA membrane-based microneedle system for transdermal drug delivery. Materials Horizons, 2020, 7(5): 1317–1326
Chen J, Cheng P, Sun C, et al. A minimally invasive hollow microneedle with a cladding structure: Ultra-thin but strong, batch manufacturable. IEEE Transactions on Biomedical Engineering, 2019, 66(12): 3480–3485
Waghule T, Singhvi G, Dubey S K, et al. Microneedles: A smart approach and increasing potential for transdermal drug delivery system. Biomedicine and Pharmacotherapy, 2019, 109: 1249–1258
Tong Z, Zhou J, Zhong J, et al. Glucose- and H2O2-responsive polymeric vesicles integrated with microneedle patches for glucose-sensitive transcutaneous delivery of insulin in diabetic rats. ACS Applied Materials & Interfaces, 2018, 10(23): 20014–20024
Ingrole R S J, Gill H S. Microneedle coating methods: A review with a perspective. The Journal of Pharmacology and Experimental Therapeutics, 2019, 370(3): 555–569
Sun W, Araci Z, Inayathullah M, et al. Polyvinylpyrrolidone microneedles enable delivery of intact proteins for diagnostic and therapeutic applications. Acta Biomaterialia, 2013, 9(8): 7767–7774
Yang S, Wu F, Liu J, et al. Phase-transition microneedle patches for efficient and accurate transdermal delivery of insulin. Advanced Functional Materials, 2015, 25(29): 4633–4641
Yu W, Jiang G, Liu D, et al. Fabrication of biodegradable composite microneedles based on calcium sulfate and gelatin for transdermal delivery of insulin. Materials Science and Engineering C, 2017, 71: 725–734
Yu W, Jiang G, Zhang Y, et al. Polymer microneedles fabricated from alginate and hyaluronate for transdermal delivery of insulin. Materials Science and Engineering C, 2017, 80: 187–196
Wang J, Ye Y, Yu J, et al. Core-shell microneedle gel for self-regulated insulin delivery. ACS Nano, 2018, 12(3): 2466–2473
Zhang Y, Wang J, Yu J, et al. Bioresponsive microneedles with a sheath structure for H2O2 and pH cascade-triggered insulin delivery. Small, 2018, 14(14): 1704181
Wang J, Ye Y, Yu J, et al. Core-shell microneedle gel for self-regulated insulin delivery. ACS Nano, 2018, 12(3): 2466–2473
Zhang J, Lu S F, Zhu H J, et al. Amino-functionalized mesoporous silica based polyethersuflone-polyvinylpyrrolidone composite membrane for elevated temperature fuel cells. ECS Transactions, 2016, 75(14): 581–588
Jin Y, Song Y, Zhu X, et al. Goblet cell-targeting nanoparticles for oral insulin delivery and the influence of mucus on insulin transport. Biomaterials, 2012, 33(5): 1573–1582
Reed M J, Meszaros K, Entes L J, et al. A new rat model of type 2 diabetes: The fat-fed, streptozotocin-treated rat. Metabolism: Clinical and Experimental, 2000, 49(11): 1390–1394
Xu B, Jiang G, Yu W, et al. H2O2-responsive mesoporous silica nanoparticles integrated with microneedle patches for the glucosemonitored transdermal delivery of insulin. Journal of Materials Chemistry B: Materials for Biology and Medicine, 2017, 5(41): 8200–8208
Khanna P, Flam B R, Osborn B, et al. Skin penetration and fracture strength testing of silicon dioxide microneedles. Sensors and Actuators A: Physical, 2011, 170(1–2): 180–186
McGrath M G, Vucen S, Vrdoljak A, et al. Production of dissolvable microneedles using an atomised spray process: Effect of microneedle composition on skin penetration. European Journal of Pharmaceutics and Biopharmaceutics, 2014, 86(2): 200–211
References
Lai J T, Filla D, Shea R. Functional polymers from novel carboxylterminated trithiocarbonates as highly efficient RAFT agents. Macromolecules, 2002, 35(18): 6754–6756
Li M, Zhang C, Yang X L, et al. Controllable synthesis of hollow mesoporous silica nanoparticles templated by kinetic self-assembly using a gemini surfactant. RSC Advances, 2013, 3(37): 16304–16307
Zhang J, Lu S, Zhu H, et al. Amino-functionalized mesoporous silica based polyethersulfone-polyvinylpyrrolidone composite membranes for elevated temperature proton exchange membrane fuel cells. RSC Advances, 2016, 6(89): 86575–86585
Tong Z, Zhou J, Zhong J, et al. Glucose- and H2O2-responsive polymeric vesicles integrated with microneedle patches for glucose-sensitive transcutaneous delivery of insulin in diabetic rats. ACS Applied Materials & Interfaces, 2018, 10(23): 20014–20024
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Grant No. 21604073), the Natural Science Foundation of Zhejiang Province (LY20E030005), the Fundamental Research Funds of Zhejiang Sci-Tech University (2019Q003), and the Opening Project of Jiangxi Province Key Laboratory of Polymer Micro/Nano Manufacturing and Devices (PMND201905).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Y., Cheng, S., Hu, W. et al. Polymer-grafted hollow mesoporous silica nanoparticles integrated with microneedle patches for glucose-responsive drug delivery. Front. Mater. Sci. 15, 98–112 (2021). https://doi.org/10.1007/s11706-021-0532-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11706-021-0532-1