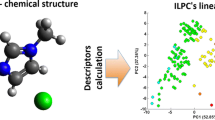
Abstract
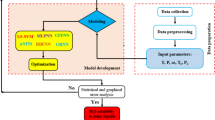
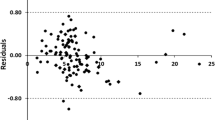
In this study, a total of 160 experimental data points of Henry's law constant of CO2 in 32 imidazole ionic liquids (ILs) were collected, with the temperatures range from 283 to 350 K. Herein intuitive and explanatory descriptors related to Henry's law constant (HLC) were suggested from the 2D structural features of the ILs according to experimental experience and laws. Temperature was used as another variable due to its significant effect on Henry's law constant. Three machine learning methods were used to construct models to fast predict the HLC based on suggested descriptors. Multi-layer Perceptrowas mainly used to build the model and compared with the results of Random forest and Multiple Linear Regression after investigating the outliers and variable selection. In addition, if only one data point was left at a similar temperature and the reduced dataset was also used to build models in the same procedure, the results were not as good as those of the full dataset but still satisfactory.


Similar content being viewed by others
References
Abraham MH, Acree WE, Hoekman D (2019) A new method for the determination of Henry’s law constants (air-water-partition coefficients). Fluid Phase Equilib 502:112300. https://doi.org/10.1016/j.fluid.2019.112300
Aghaie M, Rezaei N, Zendehboudi S (2019) Assessment of carbon dioxide solubility in ionic liquid/toluene/water systems by extended PR and PC-SAFT EOSs: carbon capture implication. J Mol Liq 275:323–337. https://doi.org/10.1016/j.molliq.2018.11.038
Aki SNVK, Mellein BR, Saurer EM (2004) High-pressure phase behavior of carbon dioxide with imidazolium-based ionic liquids. J Phys Chem B 108:20355–20365. https://doi.org/10.1021/jp046895
Anderson JL, Dixon JK, Brennecke JF (2007) Solubility of CO2, CH4, C2H6, C2H4, O2, and N2 in 1-Hexyl-3-methylpyridinium Bis(trifluoromethylsulfonyl)imide: comparison to other ionic liquids. Acc Chem Res 40:1208–1216. https://doi.org/10.1021/ar7001649
Andrea TA, Kalayeh H (1991) Applications of neural networks in quantitative structure-activity relationships of dihydrofolate reductase inhibitors. J Med Chem 34:2824–2836. https://doi.org/10.1021/jm00113a022
Baghban A, Ahmadi MA, Shahraki BH (2015) Prediction carbon dioxide solubility in presence of various ionic liquids using computational intelligence approaches. J Supercrit Fluids 98:50–64. https://doi.org/10.1016/j.supflu.2015.01.002
Bahadur I, Osman K, Coquelet C (2015) Solubilities of carbon dioxide and oxygen in the ionic liquids methyl trioctyl ammonium Bis(trifluoromethylsulfonyl)imide, 1-butyl-3-methyl imidazolium Bis(trifluoromethylsulfonyl)imide, and 1-butyl-3-methyl imidazolium methyl sulfate. J Phys Chem B 119:1503–1514. https://doi.org/10.1021/jp5061057
Barycki M, Sosnowska A, Jagiello K, Puzyn T (2018) Multi-objective genetic algorithm (MOGA) as a feature selecting strategy in ionic liquids’ quantitative toxicity-toxicity relationship models’ development. J Chem Inf Model. https://doi.org/10.1021/acs.jcim.8b00378
Bhangare RC, Ajmal PY, Rathod TD (2018) Experimental and theoretical determination of henry’s law constant for polychlorinated biphenyls: its dependence on solubility and degree of chlorination. Arch Environ Contam Toxicol 76:142–152. https://doi.org/10.1007/s00244-018-0577-z
Blanchard LA, Hancu D, Beckman EJ (1999) Green processing using ionic liquids and CO2. Nature 399:28–29. https://doi.org/10.1038/19887
Breiman L (2001) Random forests. Mach Learn 45:5–32. https://doi.org/10.1023/A:1010933404324
Cadena C, Anthony JL, Shah JK (2004) Why is CO2 so soluble in imidazolium-based ionic liquids? J Am Chem Soc 126:5300–5308. https://doi.org/10.1021/ja039615x
Cerecedo Cordoba JA, González Barbosa JJ, Frausto Solís J (2019) Melting temperature estimation of imidazole ionic liquids with clustering methods. J Chem Inf Model 59:7. https://doi.org/10.1021/acs.jcim.9b00203
Chen MY, Wu T, Zhao TF, Zhou YM, Zhang QY, Aires-de-Sousa J (2019) Machine learning to predict the specific optical rotations of chiral fluorinated molecules. Spectrochim Acta Part A 223:117289. https://doi.org/10.1016/j.saa.2019.117289
Deng T, Liu F, Jia G (2019) Prediction carbon dioxide solubility in ionic liquids based on deep learning. Mol Phys 11:1–8. https://doi.org/10.1080/00268976.2019.1652367
Dutcher B, Fan M, Russell AG (2015) Amine-based CO2 capture technology development from the beginning of 2013-a review. ACS Appl Mater Interfaces 7:2137–2148. https://doi.org/10.1021/am507465f
Ghaslani D, Gorji ZE, Gorji AE (2017) Descriptive and predictive models for Henry’s law constant of CO2, in ionic liquids: a QSPR study. Chem Eng Res Des 120:15–25. https://doi.org/10.1016/j.cherd.2016.12.020
Gorji AE, Gorji ZE, Riahi S (2017) Quantitative structure-property relationship (QSPR) for prediction of CO2 Henry’s law constant in some physical solvents with consideration of temperature effects. Korean J Chem Eng 34:405–1415. https://doi.org/10.1007/s11814-017-0018-0
Hall M, Frank E, Holmes G (2009) The WEKA data mining software: an update. SIGKDD Explor 11:10–18. https://doi.org/10.1145/1656274.1656278
Hallett JP, Welton T (2011) Room-temperature ionic liquids: solvents for synthesis and catalysis. 2. Chem Rev 111:3508–3576. https://doi.org/10.1021/cr1003248
Ihaka R, Gentleman R (1996) R: a language for data analysis and graphics. J Comput Graph Stat 5:299–314. https://doi.org/10.2307/1390807
Kapelner A, Krieger A, Blanford WJ (2016) Optimal experimental designs for estimating Henry’s law constants via the method of phase ratio variation. J Chromatogr 1468:183–191. https://doi.org/10.1016/j.chroma.2016.08.049
Kelley SP, Flores LA, Shannon MS (2017) Understanding carbon dioxide solubility in ionic liquids by exploring the link with liquid clathrate formation. Chem Eur J 23:14332–14337. https://doi.org/10.1002/chem.201703117
Kim JE, Kim HJ, Lim LS (2014) Solubility of CO2 in ionic liquids containing cyanide anions: [c2mim][SCN], [c2mim][N(CN)2], [c2mim][C(CN)3. Fluid Phase Equilib 367:151–158. https://doi.org/10.1016/j.fluid.2014.01.042
Koi ZK, Yahya WZN, Talip RAA (2019) Prediction of the viscosity of imidazolium-based ionic liquids at different temperatures using the quantitative structure property relationship approach. New J Chem 43:16207. https://doi.org/10.1039/C9NJ03436F
Liu XY, He MJ, Lv N (2016) Selective absorption of CO2 from H2, O2 and N2 by 1-hexyl-3-methylimidazolium tris(pentafluoroethyl)trifluorophosphate. J Chem Thermodyn 97:48–54. https://doi.org/10.1016/j.jct.2016.01.013
Marcus Y (2018) The solubility parameter of carbon dioxide and its solubility in ionic liquids. J Solut Chem 48:1025–1034. https://doi.org/10.1007/s10953-018-0816-y
Odabas M, Adali M (2016) Determination of temperature dependent Henry’s law constants of polychlorinated naphthalenes: application to air-sea exchange in Izmir Bay, Turkey. Atmos Environ 147:200–208. https://doi.org/10.1016/j.atmosenv.2016.10.009
Orhan OY, Ume CS, Alper E (2016) The absorption kinetics of CO2 into ionic liquid—CO2 binding organic liquid and hybrid solvents. Energy Eff Solvents CO2 Capture Gas-Liq Absorpt. https://doi.org/10.1007/978-3-319-47262-1_11
Pereira F, Xiao KX, Latino DARS, Wu CC, Zhang QY, Aires-de-Sousa J (2017) Machine learning methods to predict density functional theory B3LYP energies of HOMO and LUMO orbitals. J Chem Inf Model 57:11–21. https://doi.org/10.1021/acs.jcim.6b00340
Rogers RD, Seddon KR (2003) Ionic liquids-solvents of the future? Science 302:792–793. https://doi.org/10.1126/science.1090313
Sander R (2015) Compilation of Henry’s law constants (version 4.0) for water as solvent. Atmos Chem Phys 15:4399–4981. https://doi.org/10.5194/acp-15-4399-2015
Wang C, Yuan T, Wood SA (2017) Uncertain Henry’s law constants compromise equilibrium partitioning calculations of atmospheric oxidation products. Atmos Chem Phys. https://doi.org/10.5194/acp-2017-92
Welton T (1999) Room-temperature ionic liquids. Solvents for synthesis and catalysis. Chem Rev 99(8):2071. https://doi.org/10.1021/cr980032t
Wu T, Li W, Chen M, Zhou Y, Zhang Q (2020) Estimation of ionic liquids toxicity against leukemia rat cell line IPC-81 based on the empirical-like models using intuitive and explainable fingerprint descriptors. Mol Inf 39:2000102. https://doi.org/10.1002/minf.202000102
Yim JH, Lim JS (2013) CO2 solubility measurement in 1-hexyl-3-methylimidazolium ([HMIM]) cation based ionic liquids. Fluid Phase Equilib 352:67–74. https://doi.org/10.1016/j.fluid.2013.05.005
Zhang L, Chen J, Lv JX (2013) Progress and Development of Capture for CO2 by Ionic Liquids. Asian J Chem 25:2355–2358. https://doi.org/10.14233/ajchem.2013.13552
Zhao YS, Rafiqul I (2017) Ionic liquids for absorption and separation of gases: an eSxtensive database and a systematic screening method. AIChE J 63:1353–1367. https://doi.org/10.1002/aic.15618
Acknowledgements
This work was supported by the National Natural Science Foundation of China [No. 21576071; 21776061]; the Foundation of International Science and Technology Cooperation of Henan Province [No. 162102410012]; the Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry [No. 20091001] and the program for Science & Technology Innovation Team in Universities of Henan Province [No.19IRTSTHN029]
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, T., Li, WL., Chen, MY. et al. Prediction of Henry's law constants of CO2 in imidazole ionic liquids using machine learning methods based on empirical descriptors. Chem. Pap. 75, 1619–1628 (2021). https://doi.org/10.1007/s11696-020-01415-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01415-8