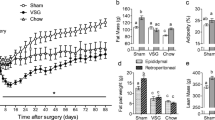
Abstract
Purpose
Ileal interposition recapitulates many of the metabolic improvements similar to Roux-en-Y gastric bypass. We aimed to determine whether the metabolic improvements seen following ileal interposition were conferred solely by the interposed segment by examining changes in neighboring intestinal segments as well as the composition of the bile acid pool.
Materials and Methods
Adult male rats were treated with either sham or ileal interposition surgeries. Glucose tolerance tests, body composition analysis, polymer chain reaction, enzyme-linked immunosorbent assay, and mass spectrometry were done after the surgeries.
Results
This study showed that ileal interposition improved glucose tolerance and enhanced both fasting and glucose-stimulated GLP-1 secretion in diabetic rats. Total bile acid pool was similar between groups but the composition favored glycine-conjugation in rats with ileal interposition. Insulin secretion was highly correlated with the 12-alpha-hydroxylase index of activity. The interposed ileum exhibited an increase in mRNA for preproglucagon and peptide YY; however, the bile acid transporter, apical sodium bile acid transporter, was dramatically reduced compared to sham rats. The interposed segment becomes jejunized in its new location as indicated by an increase in Glut2 and Pepck mRNA, genes predominantly synthesized within the jejunum.
Conclusion
Ileal relocation alone can significantly alter the bile acid pool to favor a more insulin-sensitive metabolism in association with intestinal wide alterations in mRNA for a variety of genes. Ileal interposition may confer metabolic improvement via both the interposed segment and the associated intestinal changes in all segments of the intestine, including the colon.







Similar content being viewed by others
References
Pories WJ, Swanson MS, MacDonald KG, et al. Who would have thought it? An operation proves to be the most effective therapy for adult-onset diabetes mellitus. Ann Surg. 1995;222(3):339–50. discussion 50-2
Strader AD, Clausen TR, Goodin SZ, et al. Ileal interposition improves glucose tolerance in low dose streptozotocin-treated diabetic and euglycemic rats. Obes Surg. 2009;19(1):96–104.
Strader AD, Vahl TP, Jandacek RJ, et al. Weight loss through ileal transposition is accompanied by increased ileal hormone secretion and synthesis in rats. Am J Physiol Endocrinol Metab. 2005;288(2):E447–53.
Pories WJ. Bariatric surgery: risks and rewards. J Clin Endocrinol Metabol. 2008;93(11_supplement_1):s89–96.
Tsuchiya T, Ishizuka J, Sato K, et al. Effect of ileo-jejunal transposition on the growth of the GI tract and pancreas in young and aged rats. J Gerontol A Biol Sci Med Sci. 1995;50(3):M155–61.
Junod A, Lambert AE, Stauffacher W, et al. Diabetogenic action of streptozotocin: relationship of dose to metabolic response. J Clin Invest. 1969;48(11):2129–39.
Kohli R, Kirby M, Setchell KD, et al. Intestinal adaptation after ileal interposition surgery increases bile acid recycling and protects against obesity-related comorbidities. Am J Physiol Gastrointest Liver Physiol. 2010;299(3):G652–60.
Korner J, Bessler M, Cirilo LJ, et al. Effects of Roux-en-Y gastric bypass surgery on fasting and postprandial concentrations of plasma ghrelin, peptide YY, and insulin. J Clin Endocrinol Metab. 2005;90(1):359–65.
Ishida H, Yamashita C, Kuruta Y, et al. Insulin is a dominant suppressor of sterol 12 alpha-hydroxylase P450 (CYP8B) expression in rat liver: possible role of insulin in circadian rhythm of CYP8B. J Biochem. 2000;127(1):57–64.
Kimura A, Ushijima K, Yamakawa R, et al. Large amounts of 1 beta-hydroxylated bile acids in urine during taurine therapy. Kurume Med J. 1992;39(2):105–11.
Uchida K, Satoh T, Takase H, et al. Altered bile acid metabolism related to atherosclerosis in alloxan diabetic rats. J Atheroscler Thromb. 1996;3(1):52–8.
Haeusler RA, Pratt-Hyatt M, Welch CL, et al. Impaired generation of 12-hydroxylated bile acids links hepatic insulin signaling with dyslipidemia. Cell Metab. 2012;15(1):65–74.
Kaur A, Patankar JV, de Haan W, et al. Loss of Cyp8b1 improves glucose homeostasis by increasing GLP-1. Diabetes. 2015;64(4):1168–79.
Li T, Francl JM, Boehme S, et al. Glucose and insulin induction of bile acid synthesis: mechanisms and implication in diabetes and obesity. J Biol Chem. 2012;287(3):1861–73.
Johnson LR, Barrett K, Gishan FK, et al. Physiology of the gastrointestinal tract. Cambridge: Elsevier Inc.; 2006. p. 1441–53.
Troy S, Soty M, Ribeiro L, et al. Intestinal gluconeogenesis is a key factor for early metabolic changes after gastric bypass but not after gastric lap-band in mice. Cell Metab. 2008;8(3):201–11.
Wolff BS, Meirelles K, Meng Q, et al. Roux-en-Y gastric bypass alters small intestine glutamine transport in the obese Zucker rat. Am J Physiol Gastrointest Liver Physiol. 2009;297(3):G594–601.
Matthews D, Hosker J, Rudenski A, et al. Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412–9.
Acknowledgements
The present study was supported by funds to Dr. Strader from NIDDK–Challenge Grant 1RC1DK086999-01. A grant (No. 42903) for Ping Zhao from the University of North Alabama College of Arts and Sciences also contributes to the project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The welfare of animals used for research has been respected. All applicable institutional and/or national guidelines for the care and use of animals were followed. All animal procedures were approved by the Institutional Animal Care and Use Committee of Southern Illinois University-Carbondale.
Informed Consent
Does not apply.
Electronic supplementary material
Supplementary figure 1
Homeostasis model assessment for insulin resistance (HOMA-IR) was calculated using the HOMA formula: HOMA-IR = Fasting insulin (mU/L) × plasma glucose (mg/dL) ÷ 405. The HOMA-IR result correlated with Hyperinsulinemic-euglycemic clamp (r = 0.88) [18]. The insulin resistance in diabetic Long-Evans rats after IT surgery was significantly reduced as compared to sham-operated rats (Student’s t-test; *p < 0.05). (GIF 11 kb)
Supplementary figure 2
Liver bile-related gene expression in non-diabetic rats following ileal interposition or sham surgery. A number of genes related to bile acid synthesis and signaling were measured within the livers of non-diabetic rats three months following surgery. Although none of the genes were significant at the level of p < 0.05, a few genes approached significance. Interestingly, Fxr exhibited a decrease following ileal interposition (p = 0.05). Sample size was n = 7–9 rats for gene expression analysis. (GIF 20 kb)
Rights and permissions
About this article
Cite this article
Zhao, P., Wendt, D., Goodin, S.Z. et al. Adaptation of Intestinal and Bile Acid Physiology Accompany the Metabolic Benefits Following Ileal Interposition in the Rat. OBES SURG 28, 725–734 (2018). https://doi.org/10.1007/s11695-017-2886-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-017-2886-0