Abstract
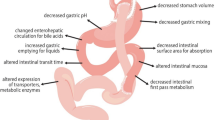
An important issue in the follow-up of patients with bariatric surgery remains to determine whether their therapeutic management should be different after surgery. In this article, we first reviewed all pharmacokinetic studies involving at least four subjects who underwent the Roux-en-Y gastric bypass (RYGB) bariatric surgery. Twenty-five publications were selected and, overall, 25 drugs were studied. Drug solubility and permeability parameters for each drug were defined using different parameters or classifications. Increased rates of oral drug absorption were predominantly observed. Conversely, drug exposure differed from one drug to another. Considering the galenic formulation and the Biopharmaceutics Classification System (BCS) class may help the prediction of oral drug exposure outcome after RYGB. We propose a strategy aiming to guide prescription and drug monitoring in patients with RYGB. But further research is clearly needed due to the unique characteristics of the bariatric population. Priority should be given to drugs that do not have clinical or biological surrogates for dose adaptation.


Similar content being viewed by others
References
Sjöström L. Review of the key results from the Swedish obese subjects (SOS) trial—a prospective controlled intervention study of bariatric surgery. J Intern Med. 2013;273(3):219–34.
Sjöström CD, Lissner L, Wedel H, et al. Reduction in incidence of diabetes, hypertension and lipid disturbances after intentional weight loss induced by bariatric surgery: the SOS intervention study. Obes Res. 1999;7(5):477–84.
Chang SH, Stoll CR, Song J, et al. The effectiveness and risks of bariatric surgery: an updated systematic review and meta-analysis, 2003-2012. JAMA Surg. 2014;149(3):275–87.
Buchwald H. The evolution of metabolic/bariatric surgery. Obes Surg. 2014;24(8):1126–35.
Nguyen NT, Nguyen B, Gebhart A, et al. Changes in the makeup of bariatric surgery: a national increase in use of laparoscopic sleeve gastrectomy. J Am Coll Surg. 2013;216(2):252–7.
Padwal R, Brocks D, Sharma AM. A systematic review of drug absorption following bariatric surgery and its theoretical implications. Obes Rev. 2010;11(1):41–50.
Brocks DR, Ben-Eltriki M, Gabr RQ, et al. The effects of gastric bypass surgery on drug absorption and pharmacokinetics. Expert Opin Drug Metab Toxicol. 2012;8(12):1505–19.
Darwich AS, Pade D, Ammori BJ, et al. A mechanistic pharmacokinetic model to assess modified oral drug bioavailability post bariatric surgery in morbidly obese patients: interplay between CYP3A gut wall metabolism, permeability and dissolution. J Pharm Pharmacol. 2012;64(7):1008–24.
De Smet J, Van Bocxlaer J, Boussery K. The influence of bypass procedures and other anatomical changes in the gastrointestinal tract on the oral bioavailability of drugs. J Clin Pharmacol. 2013;53(4):361–76.
Stein J, Stier C, Raab H, et al. Review article: the nutritional and pharmacological consequences of obesity surgery. Aliment Pharmacol Ther. 2014;40(6):582–609.
Greenblatt HK, Greenblatt DJ. Altered drug disposition following bariatric surgery: a research challenge. Clin Pharmacokinet. 2015;54(6):573–9.
Skottheim IB, Stormark K, Christensen H, et al. Significantly altered systemic exposure to atorvastatin acid following gastric bypass surgery in morbidly obese patients. Clin Pharmacol Ther. 2009;86(3):311–8.
Jakobsen GS, Skottheim IB, Sandbu R, et al. Long-term effects of gastric bypass and duodenal switch on systemic exposure of atorvastatin. Surg Endosc. 2013;27(6):2094–101.
Irwin AN, McCool KH, Delate T, et al. Assessment of warfarin dosing requirements after bariatric surgery in patients requiring long-term warfarin therapy. Pharmacotherapy. 2013;33(11):1175–83.
Steffen KJ, Wonderlich JA, Erickson AL, et al. Comparison of warfarin dosages and international normalized ratios before and after Roux-en-Y gastric bypass surgery. Pharmacotherapy. 2015;35(9):876–80.
Hamad GG, Helsel JC, Perel JM, et al. The effect of gastric bypass on the pharmacokinetics of serotonin reuptake inhibitors. Am J Psychiatry. 2012;169(3):256–63.
Dahan A, Miller JM, Amidon GL. Prediction of solubility and permeability class membership: provisional BCS classification of the world’s top oral drugs. AAPS J. 2009;11(4):740–6.
: Shah VP, Amidon GL. G.L. Amidon, H. Lennernas, V.P. Shah, and J.R. Crison. A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability, Pharm Res 12, 413-420, 1995--backstory of BCS. AAPS J 2014;16(5):894–898.
De Smet J, Colin P, De Paepe P, et al. Oral bioavailability of moxifloxacin after Roux-en-Y gastric bypass surgery. J Antimicrob Chemother. 2012;67(1):226–9.
Colin P, Eleveld DJ, Struys MM, et al. Moxifloxacin dosing in post-bariatric surgery patients. Br J Clin Pharmacol. 2014;78(1):84–93.
Padwal RS, Ben-Eltriki M, Wang X, et al. Effect of gastric bypass surgery on azithromycin oral bioavailability. J Antimicrob Chemother. 2012;67(9):2203–6.
Hamilton R, Thai XC, Ameri D, et al. Oral bioavailability of linezolid before and after Roux-en-Y gastric bypass surgery: is dose modification necessary in obese subjects? J Antimicrob Chemother. 2013;68(3):666–73.
Rogers CC, Alloway RR, Alexander JW, et al. Pharmacokinetics of mycophenolic acid, tacrolimus and sirolimus after gastric bypass surgery in end-stage renal disease and transplant patients: a pilot study. Clin Transpl. 2008;22(3):281–91.
Roerig JL, Steffen K, Zimmerman C, et al. Preliminary comparison of sertraline levels in postbariatric surgery patients versus matched nonsurgical cohort. Surg Obes Relat Dis. 2012;8(1):62–6.
Roerig JL, Steffen KJ, Zimmerman C, et al. A comparison of duloxetine plasma levels in postbariatric surgery patients versus matched nonsurgical control subjects. J Clin Psychopharmacol. 2013;33(4):479–84.
Marzinke MA, Petrides AK, Steele K, et al. Decreased escitalopram concentrations post-Roux-en-Y gastric bypass surgery. Ther Drug Monit. 2015;37(3):408–12.
Lloret-Linares C, Hirt D, Bardin C, et al. Effect of a Roux-en-Y gastric bypass on the pharmacokinetics of oral morphine using a population approach. Clin Pharmacokinet. 2014;53(10):919–30.
Mitrov-Winkelmolen L, van Buul-Gast MC, Swank DJ, et al. The effect of Roux-en-Y gastric bypass surgery in morbidly obese patients on pharmacokinetics of (acetyl)salicylic acid and omeprazole: the ERY-PAO study. Obes Surg. 2016;26(9):2051–8.
Tandra S, Chalasani N, Jones DR, et al. Pharmacokinetic and pharmacodynamic alterations in the Roux-en-Y gastric bypass recipients. Ann Surg. 2013;258(2):262–9.
Gesquiere I, Darwich AS, Van der Schueren B, et al. Drug disposition and modelling before and after gastric bypass: immediate and controlled-release metoprolol formulations. Br J Clin Pharmacol. 2015;80(5):1021–30.
Chan LN, Lin YS, Tay-Sontheimer JC, et al. Proximal Roux-en-Y gastric bypass alters drug absorption pattern but not systemic exposure of CYP3A4 and P-glycoprotein substrates. Pharmacotherapy. 2015;35(4):361–9.
Padwal RS, Gabr RQ, Sharma AM, et al. Effect of gastric bypass surgery on the absorption and bioavailability of metformin. Diabetes Care. 2011;34(6):1295–300.
Sakhaee K, Pak C. Superior calcium bioavailability of effervescent potassium calcium citrate over tablet formulation of calcium citrate after Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2013;9(5):743–8.
Tondapu P, Provost D, Adams-Huet B, et al. Comparison of the absorption of calcium carbonate and calcium citrate after Roux-en-Y gastric bypass. Obes Surg. 2009;19(9):1256–61.
Klockhoff H, Näslund I, Jones AW. Faster absorption of ethanol and higher peak concentration in women after gastric bypass surgery. Br J Clin Pharmacol. 2002;54(6):587–91.
Steffen KJ, Engel SG, Pollert GA, et al. Blood alcohol concentrations rise rapidly and dramatically after Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2013;9(3):470–3.
Rubio IG, Galrão AL, Santo MA, et al. Levothyroxine absorption in morbidly obese patients before and after Roux-en-Y gastric bypass (RYGB) surgery. Obes Surg. 2012;22(2):253–8.
Gkotsina M, Michalaki M, Mamali I, et al. Improved levothyroxine pharmacokinetics after bariatric surgery. Thyroid. 2013;23(4):414–9.
Pirola I, Formenti AM, Gandossi E, et al. Oral liquid L-thyroxine (L-t4) may be better absorbed compared to L-T4 tablets following bariatric surgery. Obes Surg. 2013;23(9):1493–6.
Brill MJ, van Rongen A, van Dongen EP, et al. The pharmacokinetics of the CYP3A substrate midazolam in morbidly obese patients before and one year after bariatric surgery. Pharm Res. 2015;32(12):3927–36.
Tsume Y, Mudie DM, Langguth P, et al. The Biopharmaceutics Classification System: subclasses for in vivo predictive dissolution (IPD) methodology and IVIVC. Eur J Pharm Sci. 2014;57:152–63.
Allain F, Minogianis EA, Roberts DC, et al. How fast and how often: the pharmacokinetics of drug use are decisive in addiction. Neurosci Biobehav Rev. 2015;56:166–79.
Acknowledgments
We would like to thank Julie Lecas for English review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Authors 1, 2, 3, and 4 declare that they have no conflict of interest. The last author (5) received 7500 euros from the APICIL study group for conducting a clinical study on morphine absorption following RYGB, OBEMO 2, that was conducted during the year 2016.
Ethical Approval
For this review, informed consent was not necessary. But in each article included in the review, informed consent was obtained from all individual participants for whom identifying information was included.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Grant Information
None
Additional information
Special characters used in the paper:
↗: northeast arrow, unicode 2197
↘: southeast arrow, unicode 2198
=: equal sign, unicode 003D
Rights and permissions
About this article
Cite this article
Hachon, L., Declèves, X., Faucher, P. et al. RYGB and Drug Disposition: How to Do Better? Analysis of Pharmacokinetic Studies and Recommendations for Clinical Practice. OBES SURG 27, 1076–1090 (2017). https://doi.org/10.1007/s11695-016-2535-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-016-2535-z