Abstract
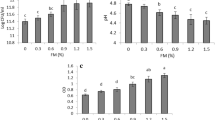
This study was aimed to evaluate the effect of galactomannan mucilage (0–2%) extracted from wild sage seed on the growth of Lactobacillus casei and the potential of this mucilage in combination with alginate, as an encapsulating agent, for protecting L. casei under gastrointestinal conditions. For this purpose, wild sage seed mucilage (WSSM) was extracted and its extraction yield and monosaccharide components were determined. Then, the effect of WSSM (0–2%) on the growth of L. casei in MRS broth and MRS agar was investigated. Finally, L. casei was encapsulated by a mixture of sodium alginate (2%) and WSSM (0, 0.5, 1, and 1.5%) using the emulsion technique and their thermal properties, zeta potential as well as their stability in simulated gastrointestinal conditions were studied. According to the results, WSSM had a relatively high extraction yield (12.31 ± 0.98%) and the most important monosaccharides in its structure were galactose and mannose. This mucilage had a stimulatory effect on the growth of L. casei. The mean diameter of the microspheres and their negative charge significantly increased by adding WSSM (P < 0.05). According to the differential scanning calorimetry results, the addition of WSSM significantly increased the melting enthalpy, melting temperature, and glass transition temperature of microcapsules (P < 0.05). Moreover, it was observed that the addition of WSSM significantly improved the resistance of L. casei against gastrointestinal conditions (P < 0.05). However, no significant difference was observed between the microcapsules containing 1 and 1.5% WSSM (P > 0.05).




Similar content being viewed by others
References
P. Darjani, M.H. Nezhad, R. Kadkhodaee, E. Milani, LWT 73, 162–167 (2016)
A. Shafizadeh, L. Golestan, M. Ahmadi, P. Darjani, A. Ghorbani-HasanSaraei, J. Food Meas. Charact. 14(4), 1901–1908 (2020)
M. Chávarri, I. Marañón, R. Ares, F.C. Ibáñez, F. Marzo, M. del Carmen Villarán, Int. J. Food Microbiol. 142(1–2), 185–189 (2010)
S. Misra, P. Pandey, H.N. Mishra, Trends Food Sci. Technol. 109, 340–351 (2021)
X.-S. Qin, Q.-Y. Gao, Z.-G. Luo, Food Hydrocolloids 116, 106658 (2021)
K. Kailasapathy, Curr. Issues Intest. Microbiol. 3(2), 39–48 (2002)
J. Burgain, C. Gaiani, M. Linder, J. Scher, J. Food Eng. 104(4), 467–483 (2011)
G.R. Gibson, H.M. Probert, J. Van Loo, R.A. Rastall, M.B. Roberfroid, Nutr. Res. Rev. 17(2), 259–275 (2004)
F. Nazzaro, F. Fratianni, R. Coppola, A. Sada, P. Orlando, J. Funct. Foods 1(3), 319–323 (2009)
S. Sathyabama, R. Vijayabharathi, LWT 57(1), 419–425 (2014)
R. Li, N. Tang, X. Jia et al., Int. J. Biol. Macromol. 162, 1217–1226 (2020)
M. Majeed, S. Majeed, K. Nagabhushanam et al., Food Sci. Nutr. 6(3), 666–673 (2018)
R. Badia, R. Lizardo, P. Martínez, J. Brufau, Gut Microbes 4(1), 72–75 (2013)
S. Rimoldi, S. Torrecillas, D. Montero et al., PLoS ONE 15(4), e0231494 (2020)
S.M.A. Razavi, S.W. Cui, Q. Guo, H. Ding, Food Hydrocolloids 35, 453–462 (2014)
A. Bostan, S.M. Razavi, R. Farhoosh, Int. J. Food Prop. 13(6), 1380–1392 (2010)
P. Kaushik, K. Dowling, R. Adhikari, C.J. Barrow, B. Adhikari, Food Chem. 215, 333–340 (2017)
M. Bustamante, M. Villarroel, M. Rubilar, C. Shene, LWT 62(2), 1162–1168 (2015)
D. Mudgil, S. Barak, A. Patel, N. Shah, Int. J. Biol. Macromol. 112, 207–210 (2018)
I.M. Fareez, S.M. Lim, R.K. Mishra, K. Ramasamy, Int. J. Biol. Macromol. 72, 1419–1428 (2015)
P. de Vos, B.J. de Haan, J.A. Kamps, M.M. Faas, T. Kitano, J. Biomed. Mater. Res. A 80(4), 813–819 (2007)
K.A. Tiani, T.W. Yeung, D.J. McClements, D.A. Sela, Int J. Food Sci. Nutr. 69(2), 155–164 (2018)
S. Mokhtari, S.M. Jafari, M. Khomeiri, Y. Maghsoudlou, M. Ghorbani, Food Res. Int. 96, 19–26 (2017)
M. Hesami, M. Tohidfar, M. Alizadeh, M.H. Daneshvar, J. For. Res. 31(3), 789–796 (2020)
S. Magazù, F. Migliardo, M. Telling, Food Chem. 106(4), 1460–1466 (2008)
W. Cui, G. Mazza, B. Oomah, C. Biliaderis, LWT 27(4), 363–369 (1994)
A.M. Amin, A.S. Ahmad, Y.Y. Yin, N. Yahya, N. Ibrahim, Food Hydrocolloids 21(2), 273–279 (2007)
L.-E. Shi, Z.-H. Li, Z.-L. Zhang et al., LWT 54(1), 147–151 (2013)
K. Damodharan, S.A. Palaniyandi, S.H. Yang, J.W. Suh, Biotechnol. Bioprocess Eng. 22(3), 265–271 (2017)
B. Haghshenas, N. Abdullah, Y. Nami, D. Radiah, R. Rosli, A. Yari Khosroushahi, J. Appl. Microbiol. 118(4), 1048–1057 (2015)
S. Beikzadeh, A. Khezerlou, S.M. Jafari, Z. Pilevar, A.M. Mortazavian, Adv. Colloid Interface Sci. 280, 102164 (2020)
M. HadiNezhad, C. Duc, N.F. Han, F. Hosseinian, J. Food Res. 2(5), 152 (2013)
M.U. Ude, I. Oluka, P.C. Eze, J. Bioresour. Bioprod. 5(4), 283–290 (2020)
Y. Wu, G. Zhang, LWT 93, 135–141 (2018)
A. Shafizadeh, L. Golestan, M. Ahmadi, P. Darjani, A. Ghorbani-Hasan Saraei, J. Food Meas. Charact. 14, 1901–1908 (2020)
S. Talebzadeh, A. Sharifan, J. Food Process Pres. 41(1), e13026 (2017)
P. Allan-Wojtas, L.T. Hansen, A. Paulson, LWT 41(1), 101–108 (2008)
M. Fahimdanesh, N. Mohammadi, H. Ahari, M.K. Zanjani, F.Z. Hargalani, K. Behrouznasab, Afr. J. Microbiol. Res. 6(40), 6853–6858 (2012)
A. Peredo, C. Beristain, L. Pascual, E. Azuara, M. Jimenez, LWT 73, 191–196 (2016)
M.A.K. Zanjani, B.G. Tarzi, A. Sharifan, N. Mohammadi, Iran. J. Pharm. Sci. 13(3), 843 (2014)
W. Krasaekoopt, B. Bhandari, H. Deeth, Int. dairy J. 14(8), 737–743 (2004)
N. Annan, A. Borza, L.T. Hansen, Food Res. Int. 41(2), 184–193 (2008)
M. Martin, F. Lara-Villoslada, M. Ruiz, M. Morales, LWT 53(2), 480–486 (2013)
S.M.A. Razavi, M. HasanAbadi, G.R. Ghadiri, E. Salehi, Int. Food Res. J. 20(6), 3111 (2013)
S.P. Hlaing, J. Kim, J. Lee, D. Kwak, H. Kim, J.-W. Yoo, Pharmaceutics 12(7), 662 (2020)
A.P. Domínguez Rubio, J.H. Martínez, D.C. Martínez Casillas, F. Coluccio Leskow, M. Piuri, O.E. Pérez, Front. Microbiol. 8, 1783 (2017)
K. Nakamura, Y. Nishimura, T. Hatakeyama, H. Hatakeyama, Thermochim. Acta 267, 343–353 (1995)
M. Del Piano, S. Carmagnola, M. Ballarè et al., Gut Microbes 2(2), 120–123 (2011)
M. de Araújo Etchepare, G.C. Raddatz, É.M. de Moraes Flores et al., LWT 65, 511–517 (2016)
F.J. Rodrigues, M.H. Omura, M.F. Cedran, R.F. Dekker, A.M. Barbosa-Dekker, S. Garcia, J. Microencapsul. 34(5), 431–439 (2017)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nasiri, H., Golestan, L., Shahidi, SA. et al. Encapsulation of Lactobacillus casei in sodium alginate microcapsules: improvement of the bacterial viability under simulated gastrointestinal conditions using wild sage seed mucilage. Food Measure 15, 4726–4734 (2021). https://doi.org/10.1007/s11694-021-01022-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-021-01022-5