Abstract
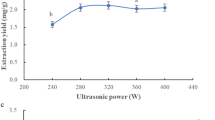
Medicinal and edible homologous plant is receiving widespread attention due to its particularity. As an edible wild vegetable, Cirsium setosum (Willd.) MB is used to treat various bleeding diseases. This work aims to investigate the technological conditions for ultrasound assisted extraction of flavonoids in C. setosum (Willd.) MB. Using a Box–Behnken design as an experimental model, the optimum extraction process of flavonoids was studied by response surface methodology. The conditions for optimal parameters were: the ratio of liquid–solid 39.21 mL/g, the volume fraction of ethanol 71.55%, the ultrasonic time 37.76 min and the temperature 65.28 °C. This study demonstrated that flavonoids yield was greatly affected by the solid/liquid ratio as well as ultrasonic temperature, while, ethanol volume fraction and ultrasonic time had less influence during extraction. In contrast to Vc and BHT, the C. setosum (Willd.) MB flavonoids extract had a more potent scavenging ability towards DPPH radicals, hydroxyl radicals and superoxide anions. Furthermore, the reduction activity was much stronger than BHT but slightly weaker than Vc. Four flavonoids, the main contributors to the antioxidant activity of C. setosum (Willd.) MB including rutin, nicotiflorin, diosmin and linarin were identified through UPLC–MS.




Similar content being viewed by others
References
Q.G. Ma, R.R. Wei, W.M. Liu, Z.P. Sang, S. Zhang, Q.Q. Wang, Z.W. Feng, L.J. Li, Y.P. Li, China J. Chin. Mater. Med. 41, 5 (2016)
Q. Sun, L. Chang, Y.P. Ren, L. Cao, Y.G. Sun, Y.F. Du, X.W. Shi, X. Wang, L.T. Zhang, J. Sep. Sci. 35, 21 (2012)
Z.M. Feng, Y.N. Yang, J.S. Jiang, P.C. Zhang, Chin. J. Exp. Tradit. Med. Formulae 18, 6 (2012)
H. Jiang, Y.H. Meng, L. Yang, Q.H. Wang, B.Y. Yang, C. Liu, H.X. Kuang, Chin. J. Nat. Med. 11, 5 (2013)
Q.G. Ma, Y.M. Guo, B.M. Luo, W.M. Liu, R.R. Wei, C.X. Yang, C.H. Ding, X.F. Xu, M.H. He, China J. Chin. Mater. Med. 41, 5 (2016)
N. Luan, W.D. Wei, A.L. Wang, X.L. Wu, Y. Qi, J.J. Li, J.Q. Zheng, X.Y. Shang, J. Asian Nat. Prod. Res. 18, 11 (2016)
P. Hodek, P. Trefil, M. Stiborová, Chem.-Biol. Interact. 139, 1 (2002)
H. Zakaryan, E. Arabyan, A. Oo, K. Zandi, Arch. Virol. 162, 9 (2017)
B.C. Han, L.I. Ning, L.I. Xian, J. Shenyang Pharm. Univ. 25, 10 (2008)
Q.Q. Liang, L.Q. Ding, Y. Jiao, C.X. Li, J. Hexi Univ. 24, 5 (2008)
X.G. Liu, F.Y. Jiang, P.Y. Gao, M. Jin, D. Yang, Z.F. Nian, Z.X. Zhang, J. Mex. Chem. Soc. 59, 1 (2015)
H.Y. Xu, Y.H. Bao, Food Sci. Technol. Res. 20, 3 (2014)
L.B.D.S. Nascimento, P.F.D. Aguiar, M.V. Leal-Costa, M.A.S. Coutinho, M.P.G. Borsodi, B. Rossi-Bergmann, E.S. Tavares, S.S. Costa, Phytochem. Anal. 29, 3 (2018)
N. Ćujić, K. Šavikin, T. Janković, D. Pljevljakušic, G. Zdunic, Food Chem. 194, 135 (2016)
J.L. Xu, W.C. Wang, H. Liang, Q. Zhang, Q.Y. Li, Ind. Crops Prod. 76, 487 (2015)
A. Raza, F. Li, X. Xu, J. Tang, Int. J. Biol. Macromol. 94, 335 (2017)
F.L. Chen, Q. Zhang, S.M. Fei, H.Y. Gu, L. Yang, Ultrason. Sonochem. 35, 161 (2017)
D. Baş, İ.H. Boyacı, J. Food Eng. 78, 3 (2007)
K. Zhong, Q. Wang, Carbohydr. Polym. 80, 1 (2010)
Y. Wu, L. Yi, E.T. Li, Y.Y. Li, Y. Lu, P.J. Wang, H.L. Zhou, J.G. Liu, Y.L. Hu, D.Y. Wang, Int. J. Biol. Macromol. 102, 68 (2017)
H.B. Zhu, Y.Z. Wang, Y.X. Liu, Y.L. Xia, T. Tang, Food Anal. Methods 3, 2 (2010)
X. Wang, J.G. Cao, Y.H. Wu, Q.X. Wang, J.B. Xiao, Molecules 21, 3 (2016)
Y. Liu, H. Wang, X. Cai, J. Food Sci. Technol. 52, 4 (2015)
A.F. Li, A.L. Sun, R.M. Liu, Y.Q. Zhang, J.C. Cui, J. Chromatogr. 965, 150 (2014)
N. Chen, H. Yang, Y. Sun, J. Niu, S.Y. Liu, Peptides 38, 2 (2012)
X. Jiang, Y. Chen, L. Shi, Food Sci. Biotechnol. 22, 6 (2013)
P.P. Wu, G.Z. Ma, N.H. Li, Q. Deng, Y.Y. Yin, R.Q. Huang, Food Chem. 173, 194 (2015)
J.R. Liu, Y.C. Yang, L.S. Shi, C.C. Peng, J. Agric. Food Chem. 56, 23 (2008)
W.X. Zhou, Z. Tian, J. Beijing Med. Univ. 26, 4 (1994)
M.A. Rostagno, M. Palma, C.G. Barroso, J. Chromatogr. A. 1012, 2 (2003)
M. Igual, E. García-Martínez, M.M. Camacho, N. Martínez-Navarrete, Innov. Food Sci. Emerg. Technol. 12, 2 (2011)
F.M. Ursache, I.O. Ghinea, M. Turturică, I. Aprodu, G. Râpeanu, N. Stănciuc, Food Chem. 233, 442 (2017)
Y.Q. Wang, Y.J. Gao, H. Ding, S.J. Liu, X. Han, J.Z. Gui, D. Liu, Food Chem. 218, 152 (2017)
D.C. Bassani, D.S. Nunes, D. Granato, An. Acad. Bras. Cienc. 86, 2 (2014)
F. Dahmoune, B. Nayak, K. Moussi, H. Remini, K. Madani, Food Chem. 166, 585 (2015)
J. Lee, N. Koo, D.B. Min, Compr. Rev. Food Sci. Food Saf. 3, 1 (2004)
N.S.S. Kumar, R.A. Nazeer, R. Jaiganesh, Amino Acids. 42, 5 (2012)
M. Affes, J. Fakhfakh, I. Douad, V. Brieudes, M. Halabalaki, A. El Feki, N. Allouche, Chem. Biodivers. 14, 9 (2017)
J.X. Yang, J. Guo, J.F. Yuan, LWT-Food Sci. Technol. 41, 6 (2008)
Y.F. Zhang, S. Xiao, L.J. Sun, Z.W. Ge, F.K. Fang, W. Zhang, Y. Wang, Y.Y. Cheng, Anal. Chim. Acta 777, 5 (2013)
A. Taamalli, D. Arráez-Román, L. Abaza, I. Iswaldi, A. Fernández-Gutiérrez, M. Zarrouk, C. Segura, Phytochem. Anal. 26, 5 (2015)
M.H. Sun, Z.Q. Luo, Y. Liu, R.R. Yang, L.N. Lu, G.H. Yu, X.Y. Ma, A.X. Liu, Y.F. Guo, H.Y. Zhao, J. Food Sci. 81, 10 (2016)
Funding
This work was financially supported by the National Natural Science Foundation of China (Grant No. 31400589).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, B., Lv, D., Huang, P. et al. Optimization, evaluation and identification of flavonoids in Cirsium setosum (Willd.) MB by using response surface methodology. Food Measure 13, 1175–1184 (2019). https://doi.org/10.1007/s11694-019-00033-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-019-00033-7