Abstract
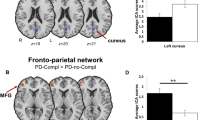
To investigate brain network centrality and connectivity alterations in different Parkinson’s disease (PD) clinical subtypes using resting-state functional magnetic resonance imaging (RS-fMRI), and to explore the correlation between baseline connectivity changes and the clinical progression. Ninety-two PD patients were enrolled at baseline, alongside 38 age- and sex-matched healthy controls. Of these, 85 PD patients underwent longitudinal assessments with a mean of 2.75 ± 0.59 years. Two-step cluster analysis integrating comprehensive motor and non-motor manifestations was performed to define PD subtypes. Degree centrality (DC) and secondary seed-based functional connectivity (FC) were applied to identify brain network centrality and connectivity changes among groups. Regression analysis was used to explore the correlation between baseline connectivity changes and clinical progression. Cluster analysis identified two main PD subtypes: mild PD and moderate PD. Two different subtypes within the mild PD were further identified: mild motor-predominant PD and mild-diffuse PD. Accordingly, the disrupted DC and seed-based FC in the left inferior frontal orbital gyrus and left superior occipital gyrus were severe in moderate PD. The DC and seed-based FC alterations in the right gyrus rectus and right postcentral gyrus were more severe in mild-diffuse PD than in mild motor-predominant PD. Moreover, disrupted DC were associated with clinical manifestations at baseline in patients with PD and predicted motor aspects progression over time. Our study suggested that brain network centrality and connectivity changes were different among PD subtypes. RS-fMRI holds promise to provide an objective assessment of subtype-related connectivity changes and predict disease progression in PD.





Similar content being viewed by others
Data availability
Origin data of the current study are available from the corresponding author on reasonable request.
References
Albanese, A., Bonuccelli, U., Brefel, C., Chaudhuri, K. R., Colosimo, C., Eichhorn, T., Melamed, E., Pollak, P., Van Laar, T., & Zappia, M. (2001). Consensus statement on the role of acute dopaminergic challenge in Parkinson’s disease. Movement Disorders, 16(2), 197–201. https://doi.org/10.1002/mds.1069
Belvisi, D., Fabbrini, A., De Bartolo, M. I., Costanzo, M., Manzo, N., Fabbrini, G., Defazio, G., Conte, A., & Berardelli, A. (2021). The pathophysiological correlates of Parkinson’s disease clinical subtypes. Movement Disorders, 36(2), 370–379. https://doi.org/10.1002/mds.28321
Bloem, B. R., Okun, M. S., & Klein, C. (2021). Parkinson’s disease. The Lancet, 397(10291), 2284–2303. https://doi.org/10.1016/s0140-6736(21)00218-x
Brendel, M., Su, C., Hou, Y., Henchcliffe, C., & Wang, F. (2021). Comprehensive subtyping of Parkinson’s disease patients with similarity fusion: A case study with BioFIND data. NPJ Parkinson’s Disease, 7(1), 83. https://doi.org/10.1038/s41531-021-00228-0
Conte, A., Khan, N., Defazio, G., Rothwell, J. C., & Berardelli, A. (2013). Pathophysiology of somatosensory abnormalities in Parkinson disease. Nature Reviews Neurology, 9(12), 687–697. https://doi.org/10.1038/nrneurol.2013.224
De Micco, R., Agosta, F., Basaia, S., Siciliano, M., Cividini, C., Tedeschi, G., Filippi, M., & Tessitore, A. (2021a). Functional connectomics and disease progression in drug-Naïve Parkinson’s disease patients. Movement Disorders, 36(7), 1603–1616. https://doi.org/10.1002/mds.28541
De Micco, R., Satolli, S., Siciliano, M., Di Nardo, F., Caiazzo, G., Russo, A., Giordano, A., Esposito, F., Tedeschi, G., & Tessitore, A. (2021b). Connectivity correlates of anxiety symptoms in drug-naive Parkinson’s disease patients. Movement Disorders, 36(1), 96–105. https://doi.org/10.1002/mds.28372
De Pablo-Fernández, E., Lees, A. J., Holton, J. L., & Warner, T. T. (2019). Prognosis and neuropathologic correlation of clinical subtypes of Parkinson disease. JAMA Neurology, 76(4), 470–479. https://doi.org/10.1001/jamaneurol.2018.4377
Elwood, R. W. (1991). The Wechsler memory scale-revised: Psychometric characteristics and clinical application. Neuropsychology Review, 2(2), 179–201. https://doi.org/10.1007/bf01109053
Erro, R., Picillo, M., Scannapieco, S., Cuoco, S., Pellecchia, M. T., & Barone, P. (2020). The role of disease duration and severity on novel clinical subtypes of Parkinson disease. Parkinsonism & Related Disorders, 73, 31–34. https://doi.org/10.1016/j.parkreldis.2020.03.013
Fereshtehnejad, S. M., & Postuma, R. B. (2017). Subtypes of Parkinson’s disease: What do they tell us about disease progression? Current Neurology and Neuroscience Reports, 17(4), 34. https://doi.org/10.1007/s11910-017-0738-x
Fereshtehnejad, S. M., Romenets, S. R., Anang, J. B., Latreille, V., Gagnon, J. F., & Postuma, R. B. (2015). New clinical subtypes of Parkinson disease and their longitudinal progression: A prospective cohort comparison with other phenotypes. JAMA Neurology, 72(8), 863–873. https://doi.org/10.1001/jamaneurol.2015.0703
Fereshtehnejad, S. M., Zeighami, Y., Dagher, A., & Postuma, R. B. (2017). Clinical criteria for subtyping Parkinson’s disease: Biomarkers and longitudinal progression. Brain: A Journal of Neurology, 140(7), 1959–1976. https://doi.org/10.1093/brain/awx118
Filippi, M., Sarasso, E., & Agosta, F. (2019). Resting-state functional MRI in parkinsonian syndromes. Movement disorders clinical practice, 6(2), 104–117. https://doi.org/10.1002/mdc3.12730
Filippi, M., Basaia, S., Sarasso, E., Stojkovic, T., Stankovic, I., Fontana, A., Tomic, A., Piramide, N., Stefanova, E., Markovic, V., Kostic, V. S., & Agosta, F. (2021). Longitudinal brain connectivity changes and clinical evolution in Parkinson’s disease. Molecular Psychiatry, 26(9), 5429–5440. https://doi.org/10.1038/s41380-020-0770-0
Friston, K. J., Williams, S., Howard, R., Frackowiak, R. S., & Turner, R. (1996). Movement-related effects in fMRI time-series. Magnetic Resonance in Medicine, 35(3), 346–355. https://doi.org/10.1002/mrm.1910350312
Goetz, C. G., Poewe, W., Rascol, O., Sampaio, C., Stebbins, G. T., Counsell, C., Giladi, N., Holloway, R. G., Moore, C. G., Wenning, G. K., Yahr, M. D., & Seidl, L. (2004). Movement disorder society task force report on the Hoehn and Yahr staging scale: Status and recommendations. Movement Disorders, 19(9), 1020–1028. https://doi.org/10.1002/mds.20213
Goetz, C. G., Stebbins, G. T., & Tilley, B. C. (2012). Calibration of unified Parkinson’s disease rating scale scores to movement disorder society-unified Parkinson’s disease rating scale scores. Movement Disorders, 27(10), 1239–1242. https://doi.org/10.1002/mds.25122
Golden, C. J. (1975). A group version of the Stroop color and word test. Journal of Personality Assessment, 39(4), 386–388. https://doi.org/10.1207/s15327752jpa3904_10
Gratwicke, J., Jahanshahi, M., & Foltynie, T. (2015). Parkinson’s disease dementia: A neural networks perspective. Brain: A Journal of Neurology, 138(Pt 6), 1454–1476. https://doi.org/10.1093/brain/awv104
Greenland, J. C., Williams-Gray, C. H., & Barker, R. A. (2019). The clinical heterogeneity of Parkinson’s disease and its therapeutic implications. European Journal of Neuroscience, 49(3), 328–338. https://doi.org/10.1111/ejn.14094
Guan, X., Guo, T., Zeng, Q., Wang, J., Zhou, C., Liu, C., Wei, H., Zhang, Y., Xuan, M., Gu, Q., Xu, X., Huang, P., Pu, J., Zhang, B., & Zhang, M. M. (2019). Oscillation-specific nodal alterations in early to middle stages Parkinson’s disease. Translational Neurodegeneration, 8, 36. https://doi.org/10.1186/s40035-019-0177-5
Guo, Q., Zhao, Q., Chen, M., Ding, D., & Hong, Z. (2009). A comparison study of mild cognitive impairment with 3 memory tests among Chinese individuals. Alzheimer Disease and Associated Disorders, 23(3), 253–259. https://doi.org/10.1097/WAD.0b013e3181999e92
Guo, M., Ren, Y., Yu, H., Yang, H., Cao, C., Li, Y., & Fan, G. (2020). Alterations in degree centrality and functional connectivity in Parkinson’s disease patients with freezing of Gait: A resting-state functional magnetic resonance imaging study. Frontiers in Neuroscience, 14, 582079. https://doi.org/10.3389/fnins.2020.582079
Hartman, D. E. (2009). Wechsler adult intelligence scale IV (WAIS IV): Return of the gold standard. Applied Neuropsychology, 16(1), 85–87. https://doi.org/10.1080/09084280802644466
Hendricks, R. M., & Khasawneh, M. T. (2021). A systematic review of Parkinson’s disease cluster analysis research. Aging Dis, 12(7), 1567–1586. https://doi.org/10.14336/AD.2021.0519
Hummel, T., Konnerth, C. G., Rosenheim, K., & Kobal, G. (2001). Screening of olfactory function with a four-minute odor identification test: Reliability, normative data, and investigations in patients with olfactory loss. The Annals of Otology Rhinology and Laryngology, 110(10), 976–981. https://doi.org/10.1177/000348940111001015
Javed, K., Reddy, V., & Lui, F. (2022). Neuroanatomy, cerebral cortex. In: StatPearls. StatPearls Publishing. Copyright © (2022). StatPearls Publishing LLC. Treasure Island (FL).
Jia, X. Z., Wang, J., Sun, H. Y., Zhang, H., Liao, W., Wang, Z., Yan, C. G., Song, X. W., & Zang, Y. F. (2019). RESTplus: An improved toolkit for resting-state functional magnetic resonance imaging data processing. Science Bulletin, 64(14), 953–954. https://doi.org/10.1016/j.scib.2019.05.008
LaBarge, E., Edwards, D., & Knesevich, J. W. (1986). Performance of normal elderly on the Boston naming test. Brain and Language, 27(2), 380–384. https://doi.org/10.1016/0093-934x(86)90026-x
Lawton, M., Ben-Shlomo, Y., May, M. T., Baig, F., Barber, T. R., Klein, J. C., Swallow, D. M. A., Malek, N., Grosset, K. A., Bajaj, N., Barker, R. A., Williams, N., Burn, D. J., Foltynie, T., Morris, H. R., Wood, N. W., Grosset, D. G., & Hu, M. T. M. (2018). Developing and validating Parkinson’s disease subtypes and their motor and cognitive progression. Journal of Neurology Neurosurgery and Psychiatry, 89(12), 1279–1287. https://doi.org/10.1136/jnnp-2018-318337
Li, S. X., Wing, Y. K., Lam, S. P., Zhang, J., Yu, M. W., Ho, C. K., Tsoh, J., & Mok, V. (2010). Validation of a new REM sleep behavior disorder questionnaire (RBDQ-HK). Sleep Medicine, 11(1), 43–48. https://doi.org/10.1016/j.sleep.2009.06.008
Lou, Y., Huang, P., Li, D., Cen, Z., Wang, B., Gao, J., Xuan, M., Yu, H., Zhang, M., & Luo, W. (2015). Altered brain network centrality in depressed Parkinson’s disease patients. Movement Disorders, 30(13), 1777–1784. https://doi.org/10.1002/mds.26321
Marin, C., Vilas, D., Langdon, C., Alobid, I., López-Chacón, M., Haehner, A., Hummel, T., & Mullol, J. (2018). Olfactory dysfunction in neurodegenerative diseases. Current Allergy and Asthma Reports, 18(8), 42. https://doi.org/10.1007/s11882-018-0796-4
Mestre, T. A., Fereshtehnejad, S. M., Berg, D., Bohnen, N. I., Dujardin, K., Erro, R., Espay, A. J., Halliday, G., van Hilten, J. J., Hu, M. T., Jeon, B., Klein, C., Leentjens, A. F. G., Marinus, J., Mollenhauer, B., Postuma, R., Rajalingam, R., Rodriguez-Violante, M., Simuni, T., Surmeier, D. J., Weintraub, D., McDermott, M. P., Lawton, M., & Marras, C. (2021). Parkinson’s disease subtypes: Critical appraisal and recommendations. J Parkinsons Dis, 11(2), 395–404. https://doi.org/10.3233/JPD-202472
Nakamura, M., Nestor, P. G., Levitt, J. J., Cohen, A. S., Kawashima, T., Shenton, M. E., & McCarley, R. W. (2008). Orbitofrontal volume deficit in schizophrenia and thought disorder. Brain: A Journal of Neurology, 131(Pt 1), 180–195. https://doi.org/10.1093/brain/awm265
Nutt, J. G. (2016). Motor subtype in Parkinson’s disease: Different disorders or different stages of disease? Movement Disorders, 31(7), 957–961. https://doi.org/10.1002/mds.26657
Olde Dubbelink, K. T., Schoonheim, M. M., Deijen, J. B., Twisk, J. W., Barkhof, F., & Berendse, H. W. (2014). Functional connectivity and cognitive decline over 3 years in Parkinson disease. Neurology, 83(22), 2046–2053. https://doi.org/10.1212/wnl.0000000000001020
Postuma, R. B., Berg, D., Stern, M., Poewe, W., Olanow, C. W., Oertel, W., Obeso, J., Marek, K., Litvan, I., Lang, A. E., Halliday, G., Goetz, C. G., Gasser, T., Dubois, B., Chan, P., Bloem, B. R., Adler, C. H., & Deuschl, G. (2015). MDS clinical diagnostic criteria for Parkinson’s disease. Movement Disorders, 30(12), 1591–1601. https://doi.org/10.1002/mds.26424
Raichle, M. E. (2015). The brain’s default mode network. Annual Review of Neuroscience, 38, 433–447. https://doi.org/10.1146/annurev-neuro-071013-014030
Rudebeck, P. H., & Rich, E. L. (2018). Orbitofrontal cortex. Current Biology: CB, 28(18), R1083–r1088. https://doi.org/10.1016/j.cub.2018.07.018
Stebbins, G. T., Goetz, C. G., Burn, D. J., Jankovic, J., Khoo, T. K., & Tilley, B. C. (2013). How to identify tremor dominant and postural instability/gait difficulty groups with the movement disorder society unified Parkinson’s disease rating scale: Comparison with the unified Parkinson’s disease rating scale. Movement Disorders, 28(5), 668–670. https://doi.org/10.1002/mds.25383
Suo, X., Lei, D., Li, N., Cheng, L., Chen, F., Wang, M., Kemp, G. J., Peng, R., & Gong, Q. (2017). Functional brain connectome and its relation to Hoehn and Yahr stage in Parkinson disease. Radiology, 285(3), 904–913. https://doi.org/10.1148/radiol.2017162929
Takahashi, T., Nakamura, M., Nishikawa, Y., Komori, Y., Nishiyama, S., Takayanagi, Y., Furuichi, A., Kido, M., Sasabayashi, D., Higuchi, Y., Noguchi, K., & Suzuki, M. (2019). Potential role of orbitofrontal surface morphology on social and cognitive functions in high-risk subjects for psychosis and schizophrenia patients. Psychiatry Research Neuroimaging, 283, 92–95. https://doi.org/10.1016/j.pscychresns.2018.12.002
Tessitore, A., Cirillo, M., & De Micco, R. (2019). Functional connectivity signatures of Parkinson’s disease. J Parkinsons Dis, 9(4), 637–652. https://doi.org/10.3233/jpd-191592
Tomlinson, C. L., Stowe, R., Patel, S., Rick, C., Gray, R., & Clarke, C. E. (2010). Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Movement Disorders, 25(15), 2649–2653. https://doi.org/10.1002/mds.23429
Trenkwalder, C., Kohnen, R., Högl, B., Metta, V., Sixel-Döring, F., Frauscher, B., Hülsmann, J., Martinez-Martin, P., & Chaudhuri, K. R. (2011). Parkinson’s disease sleep scale–validation of the revised version PDSS-2. Movement Disorders, 26(4), 644–652. https://doi.org/10.1002/mds.23476
Tuovinen, N., Seppi, K., de Pasquale, F., Müller, C., Nocker, M., Schocke, M., Gizewski, E. R., Kremser, C., Wenning, G. K., Poewe, W., Djamshidian, A., Scherfler, C., & Seki, M. (2018). The reorganization of functional architecture in the early-stages of Parkinson’s disease. Parkinsonism & Related Disorders, 50, 61–68. https://doi.org/10.1016/j.parkreldis.2018.02.013
Vaizey, C. J., Carapeti, E., Cahill, J. A., & Kamm, M. A. (1999). Prospective comparison of faecal incontinence grading systems. Gut, 44(1), 77–80. https://doi.org/10.1136/gut.44.1.77
van Rooden, S. M., Heiser, W. J., Kok, J. N., Verbaan, D., van Hilten, J. J., & Marinus, J. (2010). The identification of Parkinson’s disease subtypes using cluster analysis: A systematic review. Movement Disorders, 25(8), 969–978. https://doi.org/10.1002/mds.23116
Wang, H., Chen, H., Wu, J., Tao, L., Pang, Y., Gu, M., Lv, F., Luo, T., Cheng, O., Sheng, K., Luo, J., Hu, Y., & Fang, W. (2018). Altered resting-state voxel-level whole-brain functional connectivity in depressed Parkinson’s disease. Parkinsonism & Related Disorders, 50, 74–80. https://doi.org/10.1016/j.parkreldis.2018.02.019
Xu, P., Chen, A., Li, Y., Xing, X., & Lu, H. (2019). Medial prefrontal cortex in neurological diseases. Physiological Genomics, 51(9), 432–442. https://doi.org/10.1152/physiolgenomics.00006.2019
Yan, C. G., Wang, X. D., Zuo, X. N., & Zang, Y. F. (2016). DPABI: Data processing & analysis for (Resting-State). Brain Imaging Neuroinformatics, 14(3), 339–351. https://doi.org/10.1007/s12021-016-9299-4
Yau, Y., Zeighami, Y., Baker, T. E., Larcher, K., Vainik, U., Dadar, M., Fonov, V. S., Hagmann, P., Griffa, A., Mišić, B., Collins, D. L., & Dagher, A. (2018). Network connectivity determines cortical thinning in early Parkinson’s disease progression. Nature Communications, 9(1), 12. https://doi.org/10.1038/s41467-017-02416-0
Yoon, E. J., & Monchi, O. (2021). Probable REM sleep behavior disorder is associated with longitudinal cortical thinning in Parkinson’s disease. NPJ Parkinson’s Disease, 7(1), 19. https://doi.org/10.1038/s41531-021-00164-z
Zhang, Z., Liao, W., Chen, H., Mantini, D., Ding, J. R., Xu, Q., Wang, Z., Yuan, C., Chen, G., Jiao, Q., & Lu, G. (2011). Altered functional-structural coupling of large-scale brain networks in idiopathic generalized epilepsy. Brain: A Journal of Neurology, 134(Pt 10), 2912–2928. https://doi.org/10.1093/brain/awr223
Zhong, M., Yang, W., Huang, B., Jiang, W., Zhang, X., Liu, X., Wang, L., Wang, J., Zhao, L., Zhang, Y., Liu, Y., Lin, J., & Huang, R. (2019). Effects of levodopa therapy on voxel-based degree centrality in Parkinson’s disease. Brain Imaging and Behavior, 13(5), 1202–1219. https://doi.org/10.1007/s11682-018-9936-7
Zuo, X. N., Ehmke, R., Mennes, M., Imperati, D., Castellanos, F. X., Sporns, O., & Milham, M. P. (2012). Network centrality in the human functional connectome. Cerebral cortex (New York, NY: 1991), 22(8), 1862–1875. https://doi.org/10.1093/cercor/bhr269
Acknowledgements
We gratefully acknowledge all the participants.
Funding
This work was supported by Science and Technology Planning Project of Guangzhou (No.202201000005); National Natural Science Foundation of China (No.82071419); Key Research and Development Program of Guangzhou (No.202206010086); Guangdong Provincial Key Laboratory of Artificial Intelligence in Medical Image Analysis and Application (No.2022B1212010011); Research Project of Wuhan Health Care Commission (No.WX21A08); Research Project of Wuhan Health Care Commission (No.WZ22Q38).
Author information
Authors and Affiliations
Contributions
Conception and organization of the research project: Z.Z.C., Y.H.Z.; Collection of clinical data of participant: C.T.H., P.Z., X.H.L., W.L.H., P.Y.Z.; Statistical analysis: Z.Z.C., X.C., S.F.H.; Review and Critique of the statistical analysis: Y.H.Z.; Writing of the first draft: Z.Z.C.; Review and critique on the manuscript: P.Y.Z., M.F.C., L.J.W.; Final approval for submission: Y.H.Z.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Approval was obtained from the ethics committee of Guangdong Provincial People’s Hospital. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Informed consent
Written informed consent was obtained from all participants after they had received a full explanation of the study procedures.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, Z., He, C., Zhang, P. et al. Brain network centrality and connectivity are associated with clinical subtypes and disease progression in Parkinson’s disease. Brain Imaging and Behavior (2024). https://doi.org/10.1007/s11682-024-00862-1
Accepted:
Published:
DOI: https://doi.org/10.1007/s11682-024-00862-1