Abstract
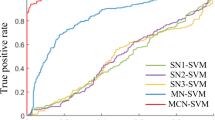
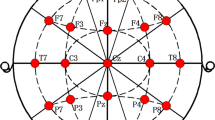
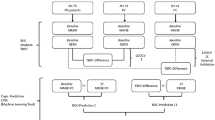
How mild cognitive impairment (MCI) is instantiated in dynamically interacting and spatially distributed functional brain networks remains an unexplored mystery in early Parkinson’s disease (PD). We applied a machine-learning technology based on personalized sliding-window algorithm to track continuously time-varying and overlapping subnetworks under the functional brain networks calculated form resting state electroencephalogram data within a sample of 33 early PD patients (13 early PD patients with MCI and 20 early PD patients without MCI). We decoded a set of subnetworks that captured surprisingly dynamically varying and integrated interactions among certain brain lobes. We observed that the master expressed subnetworks were particularly transient, and flexibly switching between high and low expression during integration into a dynamic brain network. This transience was particularly salient in a subnetwork predominantly linking temporal-parietal-occipital lobes, which decreases in both expression and flexibility in early PD patients with MCI and expresses their degree of cognitive impairment. Moreover, MCI induced a regularly interrupted, slow evolution of subnetworks in functional brain network dynamics in early PD at the individual level, and the dynamic expression characteristics of subnetworks also reflected the degree of cognitive impairment in patients with early PD. Collectively, these results provide novel and deeper insights regarding MCI-induced abnormal dynamical interaction and large-scale changes in functional brain network of early PD.





Similar content being viewed by others
Data available statement
The datasets generated for this study are available on request to the corresponding author.
References
Aarsland D, Bronnick K, Williams-Gray C et al (2010) Mild cognitive impairment in Parkinson disease A multicenter pooled analysis. Neurology 75:1062–1069
Babiloni C, Ferri R, Noce G et al (2021) Abnormalities of cortical sources of resting state alpha electroencephalographic rhythms are related to education attainment in cognitively unimpaired seniors and patients with Alzheimer’s disease and amnesic mild cognitive impairment. Cereb Cortex 31:2220–2237
Bassett DS, Wymbs NF, Porter MA, Mucha PJ, Carlson JM, Grafton ST (2011) Dynamic reconfiguration of human brain networks during learning. Proc Natl Acad Sci USA 108:7641–7646
Bassett DS, Yang MZ, Wymbs NF, Grafton ST (2015) Learning-induced autonomy of sensorimotor systems. Nat Neurosci 18:744–751
Bassett DS, Wymbs NF, Rombach MP, Porter MA, Mucha PJ, Grafton ST (2013) Task-based core-periphery organization of human brain dynamics. Plos Computat Biol 9:e1003171
Blackwell KA, Munakata Y (2014) Costs and benefits linked to developments in cognitive control. Dev Sci 17:203–211
Boutsidis C, Gallopoulos E (2008) SVD based initialization: a head start for nonnegative matrix factorization. Pattern Recogn 41:1350–1362
Bullmore ET, Sporns O (2012) The economy of brain network organization. Nat Rev Neurosci 13:336–349
Chai LR, Khambhati AN, Ciric R, Moore TM, Gur RC, Gur RE, Satterthwaite TD, Bassett DS (2017) Evolution of brain network dynamics in neurodevelopment. Netw Neurosci 1:14–30
Chen WT, Wang ZZ, Xie HB, Yu WX (2007) Characterization of surface EMG signal based on fuzzy entropy. IEEE Trans Neural Syst Rehabil Eng 15:266–272
Chen X, Wu Z, Huang NE (2010) The time-dependent intrinsic correlation based on the empirical mode decomposition. Adv Adapt Data Anal 2:233–265
Chrysikou EG, Weber MJ, Thompson-Schill SL (2014) A matched filter hypothesis for cognitive control. Neuropsychologia 62:341–355
Cordes D, Zhuang XW, Kaleem M et al (2018) Advances in functional magnetic resonance imaging data analysis methods using Empirical Mode Decomposition to investigate temporal changes in early Parkinson’s disease. Alzheimers Dement 4:372–386
Cozac, VV, Gschwandtner U, Hatz F et al (2016) Quantitative EEG and cognitive decline in Parkinson's disease, Parkinsons Disease
Fang F, Potter T, Nguyen T, Zhang YC (2020) Dynamic Reorganization of the Cortical Functional Brain Network in Affective Processing and Cognitive Reappraisal. Int J Neural Syst 30:2050051
Filippi M, Canu E, Donzuso G et al (2020) Tracking cortical changes throughout cognitive decline in Parkinson’s disease. Mov Disord 35:1987–1998
Fox MD, Snyder AZ, Vincent JL et al (2005) The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci USA 102:9673–9678
Garcia-Diaz AI, Segura B, Baggio HC et al (2018) Cortical thinning correlates of changes in visuospatial and visuoperceptual performance in Parkinson’s disease: a 4-year follow-up. Parkinsonism Relat Disord 46:62–68
Gill DJ, Freshman A, Blender JA et al (2008) The montreal cognitive assessment as a screening tool for cognitive impairment in Parkinson’s disease. Mov Disord 23:1043–1046
Goetz CG, Poewe W, Rascol O et al (2004) Movement disorder society task force report on the Hoehn and Yahr staging scale: status and recommendations. Mov Disord 19:1020–1028
Goldman JG, Litvan I (2011) Mild cognitive impairment in Parkinson’s disease. Minerva Med 102:441–459
Gu LY, Yu ZH, Ma T et al (2020) EEG-based classification of lower limb motor imagery with brain network analysis. Neuroscience 436:93–109
Hansen ECA, Battaglia D, Spiegler A, Deco G, Jirsa VK (2015) Functional connectivity dynamics: modeling the switching behavior of the resting state. Neuroimage 105:525–535
Hassan M, Wendling F (2018) Electroencephalography source connectivity: aiming for high resolution of brain networks in time and space. IEEE Signal Process Mag 35:81–96
Hinault T, Mijalkov M, Pereira JB, Volpe G, Bakke A, Courtney SM (2021) Age-related differences in network structure and dynamic synchrony of cognitive control. Neuroimage 236:118070
Hobson P, Meara J (2015) Mild cognitive impairment in Parkinson’s disease and its progression onto dementia: a 16-year outcome evaluation of the Denbighshire cohort. Int J Geriatr Psychiatry 30:1048–1055
Huang NE, Shen Z, Long SR et al (1998) The empirical mode decomposition and the Hubert spectrum for nonlinear and non-stationary time series analysis. Proc R Soc Math Phys Eng Sci 454:903–995
Huang NE, Wu ZH (2008) A review on Hilbert-Huang transform: method and its applications to geophysical studies. Rev Geophys 46: RG2006
Huang NE (2005) Introduction to the Hilbert-Huang transform and its related mathematical problems. In: Joint meeting of the society-for-industrial-and-applied-mathematics/Canadian-applied-and-indu strial-mathematics-Society 1–26 Montreal, Canada.
Hutchison RM, Womelsdorf T, Allen EA et al (2013) Dynamic functional connectivity: promise, issues, and interpretations. Neuroimage 80:360–378
Hyvarinen A (1999) Fast and robust fixed-point algorithms for independent component analysis. IEEE Trans Neural Netw 10:626–634
Kabbara A, Paban V, Weill A, Modolo J, Hassan M (2020) Brain network dynamics correlate with personality traits. Brain Connect 10:108–120
Kawada T (2015) Predictors of dementia in Parkinson disease: a prospective cohort study. Neurology 84:1285–1285
Kehagia AA, Barker RA, Robbins TW (2010) Neuropsychological and clinical heterogeneity of cognitive impairment and dementia in patients with Parkinson’s disease. Lancet Neurol 9:1200–1213
Khambhati AN, Davis KA, Lucas TH, Litt B, Bassett DS (2016) Virtual cortical resection reveals push-pull network control preceding seizure evolution. Neuron 91:1170–1182
Khambhati AN, Mattar MG, Wymbs NF, Grafton ST, Bassett DS (2018) Beyond modularity: fine-scale mechanisms and rules for brain network reconfiguration. Neuroimage 166:385–399
Khambhati AN, Bassett DS, Oommen BS, Chen SH, Lucas TH, Davis KA, Litt B (2017) Recurring functional interactions predict network architecture of interictal and ictal states in neocortical Epilepsy. Eneuro 4:pmid:28303256
Kopell NJ, Gritton HJ, Whittington MA, Kramer MA (2014) Beyond the connectome: the dynome. Neuron 83:1319–1328
Kucyi A, Daitch A, Raccah O et al (2020) Electrophysiological dynamics of antagonistic brain networks reflect attentional fluctuations. Nat Commun 11:325
Lee DD, Seung HS (1999) Learning the parts of objects by non-negative matrix factorization. Nature 401:788–791
Li FL, Yi CL, Song LM et al (2019) Brain network reconfiguration during motor imagery revealed by a large-scale network analysis of scalp EEG. Brain Topogr 32:304–314
Litvan I, Aarsland D, Adler CH et al (2011) MDS task force on mild cognitive impairment in Parkinson’s disease: critical review of PD-MCI. Mov Disord 26:1814–1824
Litvan I, Goldman JG, Troster AI et al (2012) Diagnostic criteria for mild cognitive impairment in Parkinson’s disease: movement disorder society task force guidelines. Mov Disord 27:349–356
Liu CY, Li Y, Song ST, Zhang JC (2020) Decoding disparity categories in 3-dimensional images from fMRI data using functional connectivity patterns. Cogn Neurodyn 14:169–179
Mak E, Su L, Williams GB et al (2015) Baseline and longitudinal grey matter changes in newly diagnosed Parkinson’s disease: ICICLE-PD study. Brain 138:2974–2986
Mattar MG, Cole MW, Thompson-Schill SL, Bassett DS (2015) A functional cartography of cognitive systems. Plos Comput Biol 11:e1004533
Meunier D, Lambiotte R, Bullmore ET (2010) Modular and hierarchically modular organization of brain networks. Front Neurosci 4:200
Michels L, Riese F, Meyer R et al (2021) EEG-fMRI signal coupling is modulated in subjects with mild cognitive impairment and amyloid deposition. Front Aging Neurosci 13:631172
Naim-Feil J, Rubinson M, Freche D, Grinshpoon A, Peled A, Moses E, Levit-Binnun N (2018) Altered brain network dynamics in schizophrenia: a cognitive electroencephalography study. Biol Psychiat-Cogne Neurosci Neuroimag 3:88–98
Patel R, Steele CJ, Chen AGX et al (2020) Investigating microstructural variation in the human hippocampus using non-negative matrix factorization. Neuroimage 207:116348
Patrick F, Goncalves P (2011) Empirical mode decompositions as data-driven wavelet-like expansions. Int J Wavelets Multiresolut Inf Process 2:1–20
Pedersen KF, Larsen JP, Tysnes OB, Alves G (2013) Prognosis of mild cognitive impairment in early Parkinson disease. JAMA Neurol 70:580–586
Preti MG, Bolton TAW, Van De Ville D (2017) The dynamic functional connectome: State-of-the-art and perspectives. Neuroimage 160:41–54
Protopapa F, Siettos CI, Myatchin I, Lagae L (2016) Children with well controlled epilepsy possess different spatio-temporal patterns of causal network connectivity during a visual working memory task. Cogn Neurodyn 10:99–111
Sargent K, Chavez-Baldini U, Master SL et al (2021) Resting-state brain oscillations predict cognitive function in psychiatric disorders: a transdiagnostic machine learning approach. Neuroimage-Clin 30:102617
Sen Bhattacharya B, Coyle D, Maguire LP (2011) A thalamo-cortico-thalamic neural mass model to study alpha rhythms in Alzheimer’s disease. Neural Netw 24:631–645
Sotiras A, Resnick SM, Davatzikos C (2015) Finding imaging patterns of structural covariance via Non-Negative Matrix Factorization. Neuroimage 108:1–16
Stam CJ, Nolte G, Daffertshofer A (2007) Phase lag index: Assessment of functional connectivity from multi channel EEG and MEG with diminished bias from common sources. Hum Brain Mapp 28:1178–1193
Williams-Gray CH, Foltynie T, Brayne CEG, Robbins TW, Barker RA (2007) Evolution of cognitive dysfunction in an incident Parkinson’s disease cohort. Brain 130:1787–1798
Yi GS, Wang LF, Chu CG et al (2022) Analysis of complexity and dynamic functional connectivity based on resting-state EEG in early Parkinson’s disease patients with mild cognitive impairment. Cogn Neurodyn 16:309–323
Yuan Y, Pan XC, Wang RB (2021) Biophysical mechanism of the interaction between default mode network and working memory network. Cogn Neurodyn 15:1101–1124
Zhang Y, Pan XC, Wang RB, Sakagami M (2016) Functional connectivity between prefrontal cortex and striatum estimated by phase locng value. Cogn Neurodyn 10:245–254
Zhang W, Han XX, Qiu SJ et al (2022) Analysis of brain functional network based on EEG signals for early-stage Parkinson’s disease detection. IEEE Access 10:21347–21358
Zhuang XW, Yang ZS, Mishra V, Sreenivasan K, Bernick C, Cordes D (2020) Single-scale time-dependent window-sizes in sliding-window dynamic functional connectivity analysis: a validation study. Neuroimage 220:117111
Acknowledgements
This work was supported in part by the National Natural Science Foundation of China under Grant No. 62173241, in part by the Natural Science Foundation of Tianjin, China (Grant No. 20JCQNJC01160), in part by Ministry of Science and Technology of China (Grant Nos. 2016YFC1306500, 2016YFC1306504), in part by National Key R&D Program of China (Grant No. 2016YFC1306501). The authors also gratefully acknowledge the financial support provided by Opening Fundation of Key Laboratory of Opto-technology and Intelligent Control (Lanzhou Jiaotong University), Ministry of Education (KFKT2020-01).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical statement
The studies involving human participants were reviewed and approved by Medical Ethics Committee of Tianjin Medical University General Hospital. All subjects understood the purpose of collecting the data and the significance of the study, and signed the written informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chu, C., Zhang, Z., Wang, J. et al. Evolution of brain network dynamics in early Parkinson’s disease with mild cognitive impairment. Cogn Neurodyn 17, 681–694 (2023). https://doi.org/10.1007/s11571-022-09868-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11571-022-09868-1