Abstract
Purpose
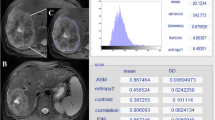
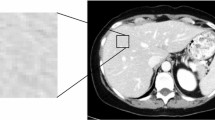
Bevacizumab added to chemotherapy can improve survival in patients with metastatic colorectal cancer, but no predictive factors of efficacy are available in clinical practice. The aim of this study is to assess the predictive and prognostic value of texture analysis on pretreatment contrast-enhanced CT in patients affected by colorectal liver metastases.
Materials and methods
Forty-three patients with colorectal liver metastases were retrospectively included in the study: 23 treated with bevacizumab-containing chemotherapy (group A), and 20 with standard chemotherapy (group B). Target liver lesions were analyzed by texture analysis of pretreatment contrast-enhanced CT. Texture analysis produced the parameter uniformity, describing lesion heterogeneity. Radiological response was classified after 3 months according to RECIST-1.1. Overall survival (OS) and progression-free survival (PFS) were considered to be outcome indicators. Multivariable logistic regression and survival analysis were performed.
Results
Uniformity was lower in responders than in nonresponders (p < 0.001) in group A but not in group B. Lesion CT density was lower in nonresponders in both groups (p = 0.03 and 0.02, respectively). In group A, uniformity was independently correlated with radiological response (odds ratio = 20, p = 0.01), OS and PFS (relative risks 6.94 and 5.05, respectively; p = 0.005 and p = 0.004, respectively). In group B, no variables were correlated with radiological response, OS or PFS.
Conclusion
Texture analysis on contrast-enhanced CT stratified response probability and prognosis in patients with colorectal liver metastases treated with bevacizumab-containing therapy. This result was specific for the bevacizumab group.



Similar content being viewed by others
Abbreviations
- CT:
-
Computed tomography
- CECT:
-
Contrast-enhanced computed tomography
- DCE-MRI:
-
Dynamic contrast-enhanced magnetic resonance imaging
- OS:
-
Overall survival
- PFS:
-
Progression-free survival
- RECIST:
-
Response evaluation criteria in solid tumors
- U :
-
Uniformity
References
Stewart BW, World Health Organization, International Agency for Research on Cancer et al (2014) World cancer report. IARC Press, Lyon
Manfredi S, Lepage C, Hatem C et al (2006) Epidemiology and management of liver metastases from colorectal cancer. Ann Surg 244:254–259. https://doi.org/10.1097/01.sla.0000217629.94941.cf
Leporrier J, Maurel J, Chiche L et al (2006) A population-based study of the incidence, management and prognosis of hepatic metastases from colorectal cancer. Br J Surg 93:465–474. https://doi.org/10.1002/bjs.5278
Landreau P, Drouillard A, Launoy G et al (2015) Incidence and survival in late liver metastases of colorectal cancer: liver colorectal cancer metastases survival. J Gastroenterol Hepatol 30:82–85. https://doi.org/10.1111/jgh.12685
Lam VWT, Spiro C, Laurence JM et al (2012) A systematic review of clinical response and survival outcomes of downsizing systemic chemotherapy and rescue liver surgery in patients with initially unresectable colorectal liver metastases. Ann Surg Oncol 19:1292–1301. https://doi.org/10.1245/s10434-011-2061-0
[No authors listed] (2000) Palliative chemotherapy for advanced or metastatic colorectal cancer. Colorectal Meta-analysis Collaboration. Cochrane Database Syst Rev CD001545. https://doi.org/10.1002/14651858.CD001545
Qu C-Y (2015) Value of bevacizumab in treatment of colorectal cancer: a meta-analysis. World J Gastroenterol 21:5072. https://doi.org/10.3748/wjg.v21.i16.5072
Schwarz RE, Berlin JD, Lenz HJ et al (2013) Systemic cytotoxic and biological therapies of colorectal liver metastases: expert consensus statement. HPB 15:106–115. https://doi.org/10.1111/j.1477-2574.2012.00558.x
Selcukbiricik F, Erdamar S, Ozkurt CU et al (2013) The role of K-RAS and B-RAF mutations as biomarkers in metastatic colorectal cancer. J BUON Off J Balk Union Oncol 18:116–123
Hirashima Y, Yamada Y, Tateishi U et al (2012) Pharmacokinetic parameters from 3-Tesla DCE-MRI as surrogate biomarkers of antitumor effects of bevacizumab plus FOLFIRI in colorectal cancer with liver metastasis. Int J Cancer 130:2359–2365. https://doi.org/10.1002/ijc.26282
O’Connor JPB, Rose CJ, Jackson A et al (2011) DCE-MRI biomarkers of tumour heterogeneity predict CRC liver metastasis shrinkage following bevacizumab and FOLFOX-6. Br J Cancer 105:139–145. https://doi.org/10.1038/bjc.2011.191
Schirin-Sokhan R, Winograd R, Roderburg C et al (2012) Response evaluation of chemotherapy in metastatic colorectal cancer by contrast enhanced ultrasound. World J Gastroenterol 18:541–545. https://doi.org/10.3748/wjg.v18.i6.541
Ganeshan B, Miles KA (2013) Quantifying tumour heterogeneity with CT. Cancer Imaging 13:140–149. https://doi.org/10.1102/1470-7330.2013.0015
Chong Y, Kim J-H, Lee HY et al (2014) Quantitative CT variables enabling response prediction in neoadjuvant therapy with EGFR-TKIs: are they different from those in neoadjuvant concurrent chemoradiotherapy? PLoS ONE 9:e88598. https://doi.org/10.1371/journal.pone.0088598
Ravanelli M, Farina D, Morassi M et al (2013) Texture analysis of advanced non-small cell lung cancer (NSCLC) on contrast-enhanced computed tomography: prediction of the response to the first-line chemotherapy. Eur Radiol 23:3450–3455. https://doi.org/10.1007/s00330-013-2965-0
Yip C, Landau D, Kozarski R et al (2014) Primary esophageal cancer: heterogeneity as potential prognostic biomarker in patients treated with definitive chemotherapy and radiation therapy. Radiology 270:141–148. https://doi.org/10.1148/radiol.13122869
Yip C, Davnall F, Kozarski R et al (2015) Assessment of changes in tumor heterogeneity following neoadjuvant chemotherapy in primary esophageal cancer. Dis Esophagus Off J Int Soc Dis Esophagus 28:172–179. https://doi.org/10.1111/dote.12170
Zhang H, Graham CM, Elci O et al (2013) Locally advanced squamous cell carcinoma of the head and neck: CT texture and histogram analysis allow independent prediction of overall survival in patients treated with induction chemotherapy. Radiology 269:801–809. https://doi.org/10.1148/radiol.13130110
Korn R, Osarogiagbon R, Newbold R et al (2014) 186 The use of Quantitative Textural Analysis imaging biomarkers to predict response to temsirolimus treatment in advanced HCC subjects. Eur J Cancer 50:61. https://doi.org/10.1016/S0959-8049(14)70312-0
Ahn SY, Park CM, Park SJ et al (2015) Prognostic value of computed tomography texture features in non-small cell lung cancers treated with definitive concomitant chemoradiotherapy. Invest Radiol 50:719–725. https://doi.org/10.1097/RLI.0000000000000174
Qiu Y, Tan M, McMeekin S et al (2016) Early prediction of clinical benefit of treating ovarian cancer using quantitative CT image feature analysis. Acta Radiol Stockh Swed 57:1149–1155. https://doi.org/10.1177/0284185115620947
Rao S-X, Lambregts DM, Schnerr RS et al (2016) CT texture analysis in colorectal liver metastases: a better way than size and volume measurements to assess response to chemotherapy? United Eur Gastroenterol J 4:257–263. https://doi.org/10.1177/2050640615601603
Goh V, Ganeshan B, Nathan P et al (2011) Assessment of response to tyrosine kinase inhibitors in metastatic renal cell cancer: CT texture as a predictive biomarker. Radiology 261:165–171. https://doi.org/10.1148/radiol.11110264
Hayano K, Tian F, Kambadakone AR et al (2015) Texture analysis of non-contrast-enhanced computed tomography for assessing angiogenesis and survival of soft tissue Sarcoma. J Comput Assist Tomogr 39:607–612. https://doi.org/10.1097/RCT.0000000000000239
Smith AD, Gray MR, del Campo SM et al (2015) Predicting overall survival in patients with metastatic melanoma on antiangiogenic therapy and RECIST stable disease on initial posttherapy images using CT texture analysis. Am J Roentgenol 205:W283–W293. https://doi.org/10.2214/AJR.15.14315
Hayano K, Kulkarni NM, Duda DG et al (2016) Exploration of imaging biomarkers for predicting survival of patients with advanced non-small cell lung cancer treated with antiangiogenic chemotherapy. AJR Am J Roentgenol 206:987–993. https://doi.org/10.2214/AJR.15.15528
Edge SB, American Joint Committee on Cancer (2010) AJCC cancer staging manual, 7th edn. Springer, New York
Ganeshan B, Abaleke S, Young RCD et al (2010) Texture analysis of non-small cell lung cancer on unenhanced computed tomography: initial evidence for a relationship with tumour glucose metabolism and stage. Cancer Imaging 10:137–143. https://doi.org/10.1102/1470-7330.2010.0021
Choi H, Charnsangavej C, Faria SC et al (2007) Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated at a single institution with Imatinib Mesylate: proposal of new computed tomography response criteria. J Clin Oncol 25:1753–1759. https://doi.org/10.1200/JCO.2006.07.3049
Development Core Team R (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Chun YS, Vauthey J-N, Boonsirikamchai P et al (2009) Association of computed tomography morphologic criteria with pathologic response and survival in patients treated with bevacizumab for colorectal liver metastases. JAMA 302:2338–2344. https://doi.org/10.1001/jama.2009.1755
Chung W-S, Park M-S, Shin SJ et al (2012) Response evaluation in patients with colorectal liver metastases: RECIST version 1.1 versus modified CT criteria. AJR Am J Roentgenol 199:809–815. https://doi.org/10.2214/AJR.11.7910
De Bruyne S, Van Damme N, Smeets P et al (2012) Value of DCE-MRI and FDG-PET/CT in the prediction of response to preoperative chemotherapy with bevacizumab for colorectal liver metastases. Br J Cancer 106:1926–1933. https://doi.org/10.1038/bjc.2012.184
Ranpura V, Hapani S, Wu S (2011) Treatment-related mortality with bevacizumab in cancer patients: a meta-analysis. JAMA 305:487–494. https://doi.org/10.1001/jama.2011.51
Lee SJ, Zea R, Kim DH et al (2018) CT texture features of liver parenchyma for predicting development of metastatic disease and overall survival in patients with colorectal cancer. Eur Radiol 28:1520–1528. https://doi.org/10.1007/s00330-017-5111-6
Simpson AL, Doussot A, Creasy JM et al (2017) Computed tomography image texture: a noninvasive prognostic marker of hepatic recurrence after hepatectomy for metastatic colorectal cancer. Ann Surg Oncol 24:2482–2490. https://doi.org/10.1245/s10434-017-5896-1
Ganeshan B, Miles KA, Young RCD, Chatwin CR (2009) Texture analysis in non-contrast enhanced CT: impact of malignancy on texture in apparently disease-free areas of the liver. Eur J Radiol 70:101–110. https://doi.org/10.1016/j.ejrad.2007.12.005
Beckers RCJ, Lambregts DMJ, Schnerr RS et al (2017) Whole liver CT texture analysis to predict the development of colorectal liver metastases—a multicentre study. Eur J Radiol 92:64–71. https://doi.org/10.1016/j.ejrad.2017.04.019
Rao S-X, Lambregts DM, Schnerr RS et al (2014) Whole-liver CT texture analysis in colorectal cancer: does the presence of liver metastases affect the texture of the remaining liver? United Eur Gastroenterol J 2:530–538. https://doi.org/10.1177/2050640614552463
Lubner MG, Stabo N, Lubner SJ et al (2015) CT textural analysis of hepatic metastatic colorectal cancer: pre-treatment tumor heterogeneity correlates with pathology and clinical outcomes. Abdom Imaging 40:2331–2337. https://doi.org/10.1007/s00261-015-0438-4
Beckers RCJ, Lambregts DMJ, Lahaye MJ et al (2018) Advanced imaging to predict response to chemotherapy in colorectal liver metastases—a systematic review. HPB 20:120–127. https://doi.org/10.1016/j.hpb.2017.10.013
Betge J, Barat A, Murphy V et al (2016) Outcome of colorectal cancer patients treated with combination bevacizumab therapy: a pooled retrospective analysis of three European Cohorts from the angiopredict initiative. Digestion 94:129–137. https://doi.org/10.1159/000449412
Ng F, Kozarski R, Ganeshan B, Goh V (2013) Assessment of tumor heterogeneity by CT texture analysis: can the largest cross-sectional area be used as an alternative to whole tumor analysis? Eur J Radiol 82:342–348. https://doi.org/10.1016/j.ejrad.2012.10.023
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ravanelli, M., Agazzi, G.M., Tononcelli, E. et al. Texture features of colorectal liver metastases on pretreatment contrast-enhanced CT may predict response and prognosis in patients treated with bevacizumab-containing chemotherapy: a pilot study including comparison with standard chemotherapy. Radiol med 124, 877–886 (2019). https://doi.org/10.1007/s11547-019-01046-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-019-01046-4