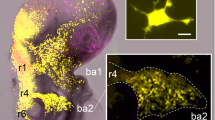
Abstract
Cell invasion and cell plasticity are critical to human development but are also striking features of cancer metastasis. By distributing a multipotent cell type from a place of birth to distal locations, the vertebrate embryo builds organs. In comparison, metastatic tumor cells often acquire a de-differentiated phenotype and migrate away from a primary site to inhabit new microenvironments, disrupting normal organ function. Countless observations of both embryonic cell migration and tumor metastasis have demonstrated complex cell signaling and interactive behaviors that have long confounded scientist and clinician alike. James D. Murray realized the important role of mathematics in biology and developed a unique strategy to address complex biological questions such as these. His work offers a practical template for constructing clear, logical, direct and verifiable models that help to explain complex cell behaviors and direct new experiments. His pioneering work at the interface of development and cancer made significant contributions to glioblastoma cancer and embryonic pattern formation using often simple models with tremendous predictive potential. Here, we provide a brief overview of advances in cell invasion and cell plasticity using the embryonic neural crest and its ancestral relationship to aggressive cancers that put into current context the timeless aspects of his work.

Similar content being viewed by others
References
Albrecht M, Lucarelli P, Kulms D, Sauter T (2020) Computational models of melanoma. Theor Biol Med Model 17(1):8. https://doi.org/10.1186/s12976-020-00126-7
Bailey CM, Kulesa PM (2014) Dynamic interactions between cancer cells and the embryonic microenvironment regulate cell invasion and reveal EphB6 as a metastasis suppressor. Mol Cancer Res 2(9):1303–1313
Barriga Eh, Franze K, Charras G, Mayor R (2018) Tissue stiffening coordinates morphogenesis by triggering collective cell migration in vivo. Nature 554(7693):523–527
Boiko AD et al (2010) Human melanoma-initiating cells express neural crest nerve growth factor receptor CD271. Nature 466(7302):133–137
Bruno et al (2020) Progress and opportunities to advance clinical cancer therapeutics using tumor dynamic models. Clin Cancer Res 26(8):1787–1795
Burgess PK, Kulesa PM, Murray JD, Alvord EC (1997) The interactions of growth rates and diffusion coefficients in a three-dimensional mathematical model of gliomas. J Neuropathol Exp Neurol 56(6):704–713
Colombi A, Scianna M, Painter KJ, Preziosi L (2020) Modelling chase-and-run migration in heterogeneous populations. J Math Biol 80(1–2):423–456
Creuzet SE, Martinez S, LeDouarin NM (2006) The cephalic neural crest exerts a critical effect on forebrain and midbrain development. PNAS 103(38):14033–14038
Darmanis S et al (2017) Single-Cell RNA-seq analysis of infiltrating neoplastic cells at the migrating front of human glioblastoma. Cell Rep 21:1399–1410
De Ieso ML, Yool AJ (2018) Mechanisms of aquaporin-facilitated cancer invasion and metastasis. Front Chem 6:135
De Ieso ML et al (2019) Combined pharmacological administration of AQP1 ion channel blocker AqB011 and water channel blocker Bacopaside II amplifies inhibition of colon cancer cell migration. Sci Rep 9:12635
Delloye-Bourgeois C, Castellani V (2019) Hijacking of embryonic programs by neural crest-derived neuroblastoma: from physiological migration to metastatic dissemination. Front MolNeurosci 12:52
Delloye-Bourgeois C et al (2017) Microenvironment-driven shift of cohesion/detachment balance within tumors induces a switch toward metastasis in neuroblastoma. Cancer Cell 32(4):427–443
Demuth T, Berens ME (2004) Molecular mechanisms of glioma cell migration and invasion. J Neurooncol 70(2):217–228
Dueck HR et al (2016) Assessing characteristics of RNA amplification methods for single cell RNA sequencing. BMC Genom 17(1):966
Dunkel H, Chaverra M, Bradley R, Lefcort F (2020) FGF signaling is required for chemokinesis and ventral migration of trunk neural crest cells. Dev Dyn 249(9):1077–1097
Eberhart JK et al (2008) MicroRNA Mirn140 modulates Pdgf signaling during palatogenesis. Nat Genet 40(3):290–298
Gadea G, Sanz-Moreno V, Self A, Godi A, Marshall CJ (2008) DOCK10-mediated Cdc42 activation is necessary for amoeboid invasion of melanoma cells. Curr Biol 18(19):1456–1465
Genuth MA, Allen C, Mikawa T, Weiner OD (2018) Chick cranial neural crest cells use progressive polarity refinement, not contact inhibition of locomotion, to guide their migration. Dev Biol 444:S252–S261
Giniunaite R, Baker RE, Kulesa PM, Maini PK (2019) Modelling collective cell migration: neural crest as a model paradigm. J Math Biol 80(1–2):481–504
Gupta PB et al (2005) The melanocyte differentiation program predisposes to metastasis after neoplastic transformation. Nat Genet 37(10):1047–1054
He F, Soriano P (2013) A critical role for PDGFR-alpha signaling in medial nasal process development. PLoS Genet 9(9):e1003851. https://doi.org/10.1371/journal.pgen.1003851
He Y, Kodali A, Wallace DI (2018) Predictive modeling of neuroblastoma growth dynamics in xenograft model after bevacizumab anti-VEGF treatment. Bull Math Biol 80(8):2026–2048
Hinshaw DC, Shevde LA (2019) The tumor microenvironment innately modulates cancer progression. Cancer Res 79(18):4557–4566
Jackson PR, Juliano J, Hawkins-Daarud A, Rockne RC, Swanson KR (2015) Patient-specific mathematical neuro-oncology: using a simple proliferation and invasion tumor model to inform clinical practice. Bull Math Biol 77(5):846–856
Johnsen JI, Dyberg C, Wickstrom M (2019) Neuroblastoma—a neural crest derived embryonal malignancy. Front MolNeurosci Rev 12:9. https://doi.org/10.3389/fnmol.2019.00009.eCollection
Karnofsky DA, Ridgway LP, Patterson PA (1952) Tumor transplantation to the chick embryo. Ann NY Acad Sci 55(2):313–329
Kasemeier-Kulesa JC, Kulesa PM (2018) The convergent roles of CD271/p75 in neural crest-derived melanoma plasticity. Dev Biol 444:S352-355
Kasemeier-Kulesa JC, McLennan R, Romine MH, Kulesa PM, Lefcort F (2010) CXCR4 controls ventral migration of sympathetic precursor cells. J Neurosci 30(39):13078–13088
Kasemeier-Kulesa JC, Morrison JA, Lefcort F, Kulesa PM (2015) TrkB/BDNF signalling patterns the sympathetic nervous system. Nat Commun 6:8281
Kasemeier-Kulesa JC, Romine MH, Morrison JA, Bailey CM, Welch DR, Kulesa PM (2018a) NGF reprograms metastatic melanoma to a bipotent glial-melanocyte neural crest-like precursor. Biol Open 7(1):bio030817. https://doi.org/10.1242/bio.030817
Kasemeier-Kulesa JC, Schnell S, Woolley T, Spengler JA, Morrison JA, McKinney MC, Pushel I, Wolfe LA, Kulesa PM (2018b) Predicting neuroblastoma using developmental signals and a logic-based model. Biophys Chem 238:30–38
Kubota Y, Ito K (2000) Chemotactic migration of mesencephalic neural crest cells in the mouse. Dev Dyn 217(2):170–179
Kulesa PM, McLennan R (2015) Neural crest migration: trailblazing ahead. F1000 Prime Rep 7:02
Kulesa PM, Ellies DL, Trainor PA (2004) Comparative analysis of neural crest cell death, migration, and function during vertebrate embryogenesis. Dev Dyn 229:14–29
Kulesa PM, Kasemeier-Kulesa JC, Teddy JM, Margaryan NV, Seftor EA, Seftor RE, Hendrix MJ (2006) Reprogramming metastatic melanoma cells to assume a neural crest cell-like phenotype in an embryonic microenvironment. PNAS 103(10):3752–3757
Kulesa PM, Teddy JM, Stark DA, Smith SE, McLennan R (2008) Neural crest invasion is a spatially-ordered progression into the head with higher cell proliferation at the migratory front as revealed by the photoactivatable protein. KikGR Dev Biol 316(2):275–287
Lake JI, Heuckeroth RO (2013) Enteric nervous system development: migration, differentiation, and disease. J PhysiolGastrointest Liver Physiol 305(1):G1–G24
LeDouarin NM, Kalcheim C (1999) The neural crest, 2nd edn. Cambridge University Press, Cambridge
Lee LMJ, Seftor EA, Bonde G, Cornell RA, Hendrix MJC (2005) The fate of human malignant melanoma cells transplanted into zebrafish embryos: assessment of migration and cell division in the absence of tumor formation. Dev Dyn 233(4):1560–1570
Lumb R, Buckberry S, Secker G, Lawrence D, Schwarz Q (2017) Transcriptomic profiling reveals expression signatures of cranial neural crest cells arising from different axial levels. BMC Dev Biol 17(1):5
MacFarland SP, Naraparaju K, Iyer R, Guan P, Kolla V, Hu Y, Tan K, Brodeur GM (2020) Mechanisms of entrectinib resistance in a neuroblastoma xenograft model. Cancer Ther 19(3):920–926
McKinney MC, McLennan R, Giniunaite R, Baker RE, Maini PK, Othmer HG, Kulesa PM (2020) Visualizing mesoderm and neural crest cell dynamics during chick head morphogenesis. Dev Biol 461(2):184–196
McLennan R, Teddy JM, Kasemeier-Kulesa JC, Romine MH, Kulesa PM (2010) Vascular endothelial growth factor (VEGF) regulates cranial neural crest migration in vivo. Dev Biol 339(1):114–125
McLennan R, Dyson L, Prather KW, Morrison JA, Baker RE, Maini PK, Kulesa PM (2012) Multiscale mechanisms of cell migration during development: theory and experiment. Development 139(16):2935–2944
McLennan R, Schumacher LJ, Morrison JA, Teddy JM, Ridenour DA, Box AC, Semerad CL, Li H, McDowell W, Kay D, Maini PK, Baker RE, Kulesa PM (2015) Neural crest migration is driven by a few trailblazer cells with a unique molecular signature narrowly confined to the invasive front. Development 142(11):2014–2025
McLennan R, McKinney MC, Teddy JM, Morrison JA, Kasemeier-Kulesa JC, Ridenour DA, Manthe CA, Giniunaite R, Robinson M, Baker RE, Maini PK (2020) Neural crest cells bulldoze through the microenvironment using Aquaporin 1 to stabilize filopodia. Development 147(1):dev185231. https://doi.org/10.1242/dev.185231
Meier F, Satyamoorthy K, Nesbit M, Jsu MY, Schittek B, Garbe C, Herlyn M (1998) Molecular events in melanoma development and progression. Front Biosci 3:D1005–D10010. https://doi.org/10.2741/a341
Merchant B, Feng JJ (2020) A Rho-GTPase based model explains group advantage in collective chemotaxis of neural crest cells. PhysBiol 17(3):036002. https://doi.org/10.1088/1478-3975/ab71f1
Merchant B, Edelstein-Keshet L, Feng JJ (2018) A Rho-GTPase based model explains spontaneous collective migration of neural crest cell clusters. Dev Biol 444:S262-273
Morais MCC et al (2017) Stochastic model of contact inhibition and the proliferation of melanoma in situ. Sci Rep 7(1):8026
Morrison JA, McLennan R, Wolfe JA, Gogol MM, Meier S, McKinney MC, Teddy JM, Holmes L, Semerad CL, Box AC, Li H, Hall KE, Perera AG, Kulesa PM (2017) Single-cell transcriptome analysis of avian neural crest migration reveals signatures of invasion and molecular transitions. Elife. 6:e28415. https://doi.org/10.7554/eLife.28415
Murray JDM (2018) My gift of polio-an unexpected life from Scotland’s rustic hills to Oxford’s hallowed halls and beyond. Chauntecleer Press, Toronto
Murray JD, Maini PK (1986) A new approach to the generation of pattern and form in embryology. Sci Prog 70(280):539–553
Murray JD, Oster GF, Harris AK (1983) A mechanical model for mesenchymal morphogenesis. J Math Biol 17(1):125–129
Olesnicky-Killian EC, Birkholz DA, Artinger KB (2009) A role for chemokine signaling in neural crest cell migration and craniofacial development. Dev Biol 333(1):161–172
Oster GF, Shubin N, Murray JD, Alberch P (1988) Evolution and morphogenetic rules: the shape of the vertebrate limb in ontogeny and phylogeny. Evolution 42(5):862–884
Panetta JC, Schaiquevich SVM, Stewart CF (2008) Using pharmacokinetic and pharmacodynamic modeling and simulation to evaluate importance of schedule in topotecan therapy for pediatric neuroblastoma. Clin Cancer Res 14(1):318–325
Plouhinec JL et al (2017) A molecular atlas of the developing ectoderm defines neural, neural crest, placode, and nonneural progenitor identity in vertebrates. PLoS Biol. 15(10):e2004045. https://doi.org/10.1371/journal.pbio.2004045
Radke J, Rossner F, Redmer T (2017) CD271 determines migratory properties of melanoma cells. Sci Rep 7:9834
Restivo G, Diener J, Cheng PF, Kiowski G, Bonalli M, Biedermann T et al (2017) Low neurotrophin receptor CD271 regulates phenotype switching in melanoma. Nat Commun 8:1988
Richardson J et al (2016) Leader cells define directionality of trunk, but not cranial, neural crest cell migration. Cell Rep 15:2076–2088
Ridenour DA, McLennan R, Teddy JM, Semerad CL, Haug JS, Kulesa PM et al (2015) The neural crest cell cycle is related to phases of migration in the head. Development 141:1095–1103
Saito D, Takase Y, Murai H, Takahashi Y (2012) The dorsal aorta initiates a molecular cascade that instructs sympatho-adrenal specification. Science 336:1578–1581
Sato A et al (2011) FGF8 signaling is chemotactic for cardiac neural crest cells. Dev Biol 354(1):18–30
Schumacher L, Kulesa PM, McLennan R, Baker RE, Maini PK (2016) Multidisciplinary approaches to understanding collective cell migration in developmental biology. Open Biol 6:160056. https://doi.org/10.1098/rsob.160056
Simoes-Costa M, Bronner ME (2016) Reprogramming of avian neural crest axial identity and cell fate. Science 352(6293):1570–1573
Soldatov R et al. (2019) Spatiotemporal structure of cell fate decisions in murine neural crest. Science 364(6444):eaas9536. https://doi.org/10.1126/science.aas9536
Song LB et al (2020) A twelve-gene signature for survival prediction in malignant melanoma patients. Ann Transl Med 8(6):312
Strizzi L, Hardy KM, Kirsammer GT, Gerami P, Hendrix MJC (2011) Embryonic signaling in melanoma: potential for diagnosis and therapy. Lab Invest 91(6):819–824
Swanson KR, Alvord EC, Murray JD (2000) A quantitative model for differential motility of gliomas in grey and white matter. Cell Prolif 33:317–329
Szabo A et al (2016) In vivo confinement promotes collective migration of neural crest cells. J Cell Biol 213(5):543–555
Szabo A et al (2019) Neural crest streaming as an emergent property of tissue interactions during morphogenesis. PLoS Comp Biol 15(4):e1007002. https://doi.org/10.1371/journal.pcbi.1007002
Taylor AM, Zon LI (2009) Zebrafish tumor assays: the state of transplantation. Zebrafish 6:339–346
Teddy JM, Kulesa PM (2004) In vivo evidence for short- and long-range cell communication in cranial neural crest cells. Development 131:6141–6151
Tetri LH et al (2020) RET receptor expression and interaction with TRK receptors in neuroblastomas. Oncol Rep 44(1):263–272
Theveneau E, Mayor R (2012) Neural crest delamination and migration: from epithelium-to-mesenchyme transition to collective cell migration. Dev Biol 366(1):34–54
Theveneau E, Steventon B, Scarpa E, Garcia S, Trepat X, Streit A, Mayor R (2013) Chase-and-run between adjacent cell populations promotes directional collective migration. Nat Cell Biol 15(7):763–772
Tirosh et al (2016) Dissecting the multicellular ecosystem of metastatic melanoma by single-cell RNA-seq. Science 352(6282):189–196
Tomita Y et al (2017) Role of aquaporin 1 signalling in cancer development and progression. Int J MolSci 18(2):299
Tracqui P, Cruywagen GC, Woodward DE, Bartoo GT, Murray JD, Alvord EC (1995) A mathematical model of glioma growth: the effect of chemotherapy on spatio-temporal growth. Cell Prolif 28(1):17–31
Trainor PA, Sobieszczuk D, Wilkinson D, Krumlauf R (2002) Signalling between the hindbrain and paraxial tissues dictates neural crest migration pathways. Development (Cambridge, England) 129:433–442
Tweedy L, Insall RH (2020) Self-generated gradients yield exceptionally robust steering cues. Front Cell Dev Biol 8:133.https://doi.org/10.3389/fcell.2020.00133
Tweedy L et al (2016) Self-generated chemoattractant gradients: attractant depletion extends the range and robustness of chemotaxis. PLoS Biol 42:46–51. https://doi.org/10.1016/j.ceb.2016.04.003
Vega-Lopez GA, Cerrizuela S, Tribulo C, Aybar MJ (2018) Neurocristopathies: new insights 150 years after the neural crest discovery. Dev Biol 444:S110-143
Veinotte CJ, Dellaire G, Berman JN (2014) Hooking the big one: the potential of zebrafish xenotransplantation to reform cancer drug screening in the genomic era. Dis Model Mech 7(7):745–754
Westcott JM et al (2015) An epigenetically distinct breast cancer cell subpopulation promotes collective invasion. J Clin Invest 125(5):1927–1943
Williams RM, Candido-Ferreira I, Repapi E, Gavriouchkina D, Senanayake U, Ling ITC, Telenius J, Taylor S, Hughes J, Sauka-Spengler T (2019) Reconstruction of the global neural crest gene regulatory network in vivo. Dev Cell 51(2):255–276
Woodward DE, Cook J, Tracqui P, Cruywagen GC, Murray JD, Alvord EC (1996) A mathematical model of glioma growth: the effect of extent of surgical resection. Cell Prolif 29(6):269–288
Wynn ML, Consul N, Merajver SD, Schnell S (2014) Inferring the effects of honokiol on the notch signaling pathway in SW480 colon cancer cells. Cancer Inform 13:1–12
Xu J, Liu Y, Adam M, Clouthier DE, Potter S, Jiang R (2019) Hedgehog signaling patterns the oral-aboral axis of the mandibular arch. Elife 14:8
Yool AJ, Ramesh S (2020) Molecular targets for combined therapeutic strategies to limit glioblastoma cell migration and invasion. FrontiPharmacol 11:358. https://doi.org/10.3389/fphar.2020.00358
Zeisel A et al (2016) Cell types in the mouse cortex and hippocampus revealed by single-cell RNA-seq. Science 347(6226):1138–1142
Acknowledgements
PMK and JCK would like to thank the generous funding from the St. Baldrick’s Foundation, and the Stowers Institute for Medical Research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kulesa, P.M., Kasemeier-Kulesa, J.C., Morrison, J.A. et al. Modelling Cell Invasion: A Review of What JD Murray and the Embryo Can Teach Us. Bull Math Biol 83, 26 (2021). https://doi.org/10.1007/s11538-021-00859-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-021-00859-7