Abstract
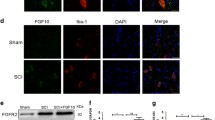
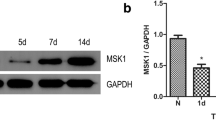
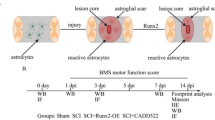
Neuronal damage, glial inflammation, and astrogliosis/astroglial scar formation are major secondary injury mechanisms that are significant contributors to functional deficits after spinal cord injury (SCI). The objectives of the study were to evaluate the distinct roles of ERK2 vs. ERK1/2 and ERK1/2-calpain 1–NF-κB signal transduction in the tissue damage and astrogliosis/astroglial scar formation following SCI in rats. RNAi approaches, pharmacological intervention (U0126), Western blot analysis, immunofluorescence analysis, and histological assessment were used to target ERK1/2-calpain 1-NF-κB signal transduction pathway for neuroprotection. Histological staining analysis demonstrated that selectively reducing pERK2 using ERK2 siRNA, but not inhibition of pERK1/2 with U0126, significantly reduced lesion volume and improved total tissue sparing, white matter sparing, and gray matter sparing in spinal cord two weeks after contusive SCI. An ERK1/2-calpain 1-NF-κB signal transduction pathway was involved in the astroglial scar formation after SCI. Blockade of ERK1/2 by U0126 decreased calpain 1 expression 4 h following SCI. Selective calpain 1 reduction by lentiviral shRNA attenuated astroglial NF-κB activity and astroglial scar formation after SCI in rats. Taken together, these results demonstrate the involvement of individual ERK2 and calpain 1 signaling pathways in tissue damage and astrogliosis/astroglial scar formation in animal models of SCI. Therefore, targeting individual ERK and its downstream signal transduction of calpain 1-NF-κB may provide greater potential as novel therapeutics for minimizing tissue damage and astroglial scar formation following SCI.
Similar content being viewed by others
References
Agrawal A, Dillon S, Denning T L, Pulendran B (2006). ERK1-/- mice exhibit Th1 cell polarization and increased susceptibility to experimental autoimmune encephalomyelitis. J Immunol, 176(10): 5788–5796
Aigner A (2006). Gene silencing through RNA interference (RNAi) in vivo: strategies based on the direct application of siRNAs. J Biotechnol, 124(1): 12–25
Borgens R B, Liu-Snyder P (2012). Understanding secondary injury. Q Rev Biol, 87(2): 89–127
Brambilla R, Hurtado A, Persaud T, Esham K, Pearse D D, Oudega M, Bethea J R (2009). Transgenic inhibition of astroglial NF-kappa B leads to increased axonal sparing and sprouting following spinal cord injury. J Neurochem, 110(2): 765–778
Bramlett H M, Dietrich W D (2007). Progressive damage after brain and spinal cord injury: pathomechanisms and treatment strategies. Prog Brain Res, 161: 125–141
Cargnello M, Roux P P (2011). Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol Mol Biol Rev, 75(1): 50–83
Colak A, Kaya M, Karaoglan A, Sagmanligil A, Akdemir O, Sahan E, Celik O (2009). Calpain inhibitor AK 295 inhibits calpain-induced apoptosis and improves neurologic function after traumatic spinal cord injury in rats. Neurocirugia (Astur), 20(3): 245–254
Doshi S, Lynch D R (2009). Calpain and the glutamatergic synapse. Front Biosci (Schol Ed), 1(1): 466–476
Drake C R, Aissaoui A, Argyros O, Serginson J M, Monnery B D, Thanou M, Steinke J H, Miller A D (2010). Bioresponsive small molecule polyamines as noncytotoxic alternative to polyethylenimine. Mol Pharm, 7(6): 2040–2055
Geddes J W, Saatman K E (2010). Targeting individual calpain isoforms for neuroprotection. Exp Neurol, 226(1): 6–7
Hetman M, Gozdz A (2004). Role of extracellular signal regulated kinases 1 and 2 in neuronal survival. Eur J Biochem, 271(11): 2050–2055
Ishikawa T, Suzuki H, Ishikawa K, Yasuda S, Matsui T, Yamamoto M, Kakeda T, Yamamoto S, Owada Y, Yaksh T L (2014). Spinal cord ischemia/injury. Curr Pharm Des, 20(36): 5738–5743
Karimi-Abdolrezaee S, Billakanti R (2012). Reactive astrogliosis after spinal cord injury-beneficial and detrimental effects. Mol Neurobiol, 46(2): 251–264
Kim J Y, Park K J, Kim G H, Jeong E A, Lee D Y, Lee S S, Kim D J, Roh G S, Song J, Ki S H, Kim W H (2013). In vivo activating transcription factor 3 silencing ameliorates the AMPK compensatory effects for ER stress-mediated ß-cell dysfunction during the progression of type-2 diabetes. Cell Signal, 25(12): 2348–2361
Kuchay S M, Chishti A H (2007). Calpain-mediated regulation of platelet signaling pathways. Curr Opin Hematol, 14(3): 249–254
Kwon B K, Sekhon L H, Fehlings M G (2010). Emerging repair, regeneration, and translational research advances for spinal cord injury. Spine, 35(21 Suppl): S263–S270
Li L, Wu Y, Wang C, Zhang W (2012). Inhibition of PAX2 gene expression by siRNA (polyethylenimine) in experimental model of obstructive nephropathy. Ren Fail, 34(10): 1288–1296
Liu J, Liu M C, Wang K K (2008). Calpain in the CNS: from synaptic function to neurotoxicity. Sci Signal, 1(14): re1
Liu L, Zhang R, Liu K, Zhou H, Tang Y, Su J, Yu X, Yang X, Tang M, Dong Q (2009). Tissue kallikrein alleviates glutamate-induced neurotoxicity by activating ERK1. J Neurosci Res, 87(16): 3576–3590
Lu P Y, Woodle MC (2008). Delivering small interfering RNA for novel therapeutics. Methods Mol Biol, 437: 93–107
Luo M C, Zhang D Q, Ma S W, Huang Y Y, Shuster S J, Porreca F, Lai J (2005). An efficient intrathecal delivery of small interfering RNA to the spinal cord and peripheral neurons. Mol Pain, 1(1): 29
Nakazawa T, Shimura M, Ryu M, Nishida K, Pagès G, Pouysségur J, Endo S (2008). ERK1 plays a critical protective role against Nmethyl- D-aspartate-induced retinal injury. J Neurosci Res, 86(1): 136–144
Rabchevsky A G, Fugaccia I, Sullivan P G, Blades D A, Scheff S W (2002). Efficacy of methylprednisolone therapy for the injured rat spinal cord. J Neurosci Res, 68(1): 7–18
Ray S K, Hogan E L, Banik N L (2003). Calpain in the pathophysiology of spinal cord injury: neuroprotection with calpain inhibitors. Brain Res Brain Res Rev, 42(2): 169–185
Saatman K E, Creed J, Raghupathi R (2010). Calpain as a therapeutic target in traumatic brain injury. Neurotherapeutics, 7(1): 31–42
Schumacher P A, Siman R G, Fehlings M G (2000). Pretreatment with calpain inhibitor CEP-4143 inhibits calpain I activation and cytoskeletal degradation, improves neurological function, and enhances axonal survival after traumatic spinal cord injury. J Neurochem, 74(4): 1646–1655
Springer J E, Azbill R D, Kennedy S E, George J, Geddes J W (1997). Rapid calpain I activation and cytoskeletal protein degradation following traumatic spinal cord injury: attenuation with riluzole pretreatment. J Neurochem, 69(4): 1592–1600
Sribnick E A, Samantaray S, Das A, Smith J, Matzelle D D, Ray S K, Banik N L (2010). Postinjury estrogen treatment of chronic spinal cord injury improves locomotor function in rats. J Neurosci Res, 88 (8): 1738–1750
Tian D S, Yu Z Y, Xie M J, Bu B T, Witte O W, Wang W (2006). Suppression of astroglial scar formation and enhanced axonal regeneration associated with functional recovery in a spinal cord injury rat model by the cell cycle inhibitor olomoucine. J Neurosci Res, 84(5): 1053–1063
Wall E A, Zavzavadjian J R, Chang M S, Randhawa B, Zhu X, Hsueh R C, Liu J, Driver A, Bao X R, Sternweis P C, Simon M I, Fraser I D (2009). Suppression of LPS-induced TNF-alpha production in macrophages by cAMP is mediated by PKA-AKAP95-p105. Sci Signal, 2(75): ra28
Wittrup A, Lieberman J (2015). Knocking down disease: a progress report on siRNA therapeutics. Nat Rev Genet, 16(9): 543–552
Wu J, Pajoohesh-Ganji A, Stoica B A, Dinizo M, Guanciale K, Faden A I (2012). Delayed expression of cell cycle proteins contributes to astroglial scar formation and chronic inflammation after rat spinal cord contusion. J Neuroinflammation, 9(1): 169
Yiu G, He Z (2006). Glial inhibition of CNS axon regeneration. Nat Rev Neurosci, 7(8): 617–627
Yu C G (2012). Distinct roles for ERK1 and ERK2 in pathophysiology of CNS. Front Biol, 7 (3): 267–276
Yu C G, Geddes J W (2007). Sustained calpain inhibition improves locomotor function and tissue sparing following contusive spinal cord injury. Neurochem Res, 32(12): 2046–2053
Yu C G, Li Y, Raza K, Yu X X, Ghoshal S, Geddes J W (2013). Calpain 1 knockdown improves tissue sparing and functional outcomes after spinal cord injury in rats. J Neurotrauma, 30(6): 427–433
Yu C G, Singh R, Crowdus C, Raza K, Kincer J, Geddes J W (2014). Fenbendazole improves pathological and functional recovery following traumatic spinal cord injury. Neuroscience, 256: 163–169
Yu C G, Yezierski R P (2005). Activation of the ERK1/2 signaling cascade by excitotoxic spinal cord injury. Brain Res Mol Brain Res, 138(2): 244–255
Yu C G, Yezierski R P, Joshi A, Raza K, Li Y, Geddes J W (2010). Involvement of ERK2 in traumatic spinal cord injury. J Neurochem, 113(1): 131–142
Yuan Y M, He C (2013). The glial scar in spinal cord injury and repair. Neurosci Bull, 29(4): 421–435
Zhang S X, Underwood M, Landfield A, Huang F F, Gison S, Geddes J W (2000). Cytoskeletal disruption following contusion injury to the rat spinal cord. J Neuropathol Exp Neurol, 59(4): 287–296
Zhao P, Waxman S G, Hains B C (2007). Extracellular signal-regulated kinase-regulated microglia-neuron signaling by prostaglandin E2 contributes to pain after spinal cord injury. J Neurosci, 27(9): 2357–2368
Zhuang S, Schnellmann R G (2006). A death-promoting role for extracellular signal-regulated kinase. J Pharmacol Exp Ther, 319(3): 991–997
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, X.X., Bondada, V., Rogers, C. et al. Targeting ERK1/2-calpain 1-NF-κB signal transduction in secondary tissue damage and astrogliosis after spinal cord injury. Front. Biol. 10, 427–438 (2015). https://doi.org/10.1007/s11515-015-1373-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11515-015-1373-z