Abstract
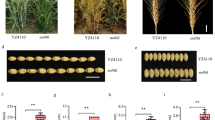
The exploration of new genes controlling rice leaf shape is an important foundation for rice functional genomics and plant architecture improvement. In the present study, we identified a rolling leaf mutant from indica variety Yuefeng B, named rl 11(t) , which exhibited reduced plant height, rolling and narrow leaves. Leaves in rl 11(t) mutant showed abnormal number and morphology of veins compared with those in wild type plants. In addition, rl 11(t) mutant was less sensitive to the inhibitory effect of auxin than the wild type. Genetic analysis suggested that the mutant was controlled by a single recessive gene. Gene rl 11(t) was initially mapped between SSR markers RM6089 and RM124 on chromosome 4. Thirty-two new STS markers around the rl 11(t) region were developed for fine mapping. A physical map encompassing the rl 11(t) locus was constructed and the target gene was finally delimited to a 31.6 kb window between STS4-25 and STS4-26 on BAC AL606645. This provides useful information for cloning of rl 11(t) gene.
Similar content being viewed by others
References
Zhu D F, Lin X Q, Cao W X. Comparison of leaf photosynthetic characteristics among rice hybrids with different leaf rolling index (in Chinese). Acta Agron Sin, 2001, 27:329–333
Lang Y Z, Zhang, Z J, Gu, X Y, et al. Physiological and ecological effects of crimpy leaf character in rice (Oryza sativa L.) I. Leaf orientation, canopy structure and light distribution (in Chinese). Acta Agron Sin, 2004, 30:739–744
Lang Y Z, Zhang, Z J, Gu, X Y, et al. Physiological and ecological effects of crimpy leaf character in rice (Oryza sativa L.) II. Photosynthetic character, dry mass production and yield forming (in Chinese). Acta Agron Sin, 2004, 30:883–887
Yuan L P. Super-high yield hybrid rice breeding (in Chinese). Hybrid Rice, 1997, 12:1–6
Chen Z X, Pan X B, Hu J, et al. Relationship between rolling leaf and ideal plant type of rice (Oryza sativa L.) (in Chinese). J Jiangsu Agric Res, 2001, 22:88–91
Li S G, Ma Y Q, He P, et al. Genetic analysis and mapping the flag leaf rolling rice (Oryza sativa L.) (in Chinese). J Sichuan Agric Univ, 1998, 16:391–393
Shao, Y J, Chen Z X, Zhang Y F, et al. One major QTL mapping and physical map construction for rolling leaf in rice (in Chinese). Acta Genet Sin, 2005, 32:501–506
Gu X Y, Gu M H. Genetic analysis of a rolling leaf character in rice (in Chinese). Hereditas, 1995, 17:20–23
Shao Y J, Pan C H, Chen Z X, et al. Fine mapping of an incomplete recessive gene for leaf rolling in rice (Oryza sativa L.). Chinese Sci Bull, 2005, 50:2466–2472
Yan C J, Yan S, Zhang Z Q, et al. Genetic analysis and gene fine mapping for a rice novel mutant (rl 9(t)) with rolling leaf character. Chinese Sci Bull, 2006, 50:63–69
Luo Z, Yang Z, Zhong B, et al. Genetic analysis and fine mapping of a dynamic rolled leaf gene, RL10(t), in rice (Oryza sativa L.). Genome, 2007, 50:811–817
Yu D, Wu H B, Yang W T, et al. Genetic analysis and mapping of the unilateral rolled leaf trait of rice mutant B157 (in Chinese). Mol Plant Breeding, 2008, 6:220–226
Wang D K, Liu H Q, Li K L, et al. Genetic analysis and gene mapping of a narrow leaf mutant in rice (Oryza sativa L.). Chinese Sci Bull, 2009, 54:752–758
Kong Y, Wang Z, Gu Y J, et al. Observations on the structure of Chlorophytum comosum roots under soil and water culture (in Chinese). Acta Horticult Sin, 2009, 36:533–538
Nelson T, Dengler N. Leaf vascular pattern formation. Plant Cell, 1997, 9:1121–1135
Hobbie L, McGovern M, Hurwitz L R, et al. The axr6 mutants of Arabidopsis thaliana define a gene involved in auxin response and early development. Development, 2000, 127:23–32
Shi Z, Wang J, Wan X, et al. Over-expression of rice OsAGO7 gene induces upward curling of the leaf blade that enhanced erect-leaf habit. Planta, 2007, 226:99–108
Yan S, Yan C J, Zeng X H, et al. ROLLED LEAF 9, encoding a GARP protein, regulates the leaf abaxial cell fate in rice. Plant Mol Biol, 2008, 68:239–250
Zhang G H, Xu Q, Zhu X D, et al. SHALLOT-LIKE1 is a KANADI transcription factor that modulates rice leaf rolling by regulating leaf abaxial cell development. Plant Cell, 2009, 21:719–735
Woo Y M, Park H J, Su’udi M, et al. Constitutively wilted 1, a member of the rice YUCCA gene family, is required for maintaining water homeostasis and an appropriate root to shoot ratio. Plant Mol Biol, 2007, 65:125–136
Fujino K, Matsuda Y, Ozawa K, et al. NARROW LEAF 7 controls leaf shape mediated by auxin in rice. Mol Genet Genomics, 2008, 279:499–507
Qi J, Qian Q, Bu Q, et al. Mutation of the rice Narrow leaf1 gene, which encodes a novel protein, affects vein patterning and polar auxin transport. Plant Physiol, 2008, 147:1947–1959
Cheng Y, Dai X, Zhao Y. Auxin biosynthesis by the YUCCA flavin monooxygenases controls the formation of floral organs and vascular tissues in Arabidopsis. Genes Dev, 2006, 20:1790–1799
Zhao Y, Christensen S K, Fankhauser C, et al. A role for flavin monooxygenase-like enzymes in auxin biosynthesis. Science, 2001, 291:306–309
Cheng Y, Dai X, Zhao Y. Auxin synthesized by the YUCCA flavin monooxygenases is essential for embryogenesis and leaf formation in Arabidopsis. Plant Cell, 2007, 19:2430–2439
Qin G, Gu H, Zhao Y, et al. An indole-3-acetic acid carboxyl methyltransferase regulates Arabidopsis leaf development. Plant Cell, 2005, 17:2693–2704
Blakeslee J J, Peer W A, Murphy A S. Auxin transport. Curr Opin Plant Biol, 2005, 8:494–500
Fukuda H. Signals that control plant vascular cell differentiation. Nat Rev Mol Cell Biol, 2004, 5:379–391
Sauer M, Balla J, Luschnig C, et al. Canalization of auxin flow by Aux/IAA-ARF-dependent feedback regulation of PIN polarity. Genes Dev, 2006, 20:2902–2911
Teale W D, Paponov I A, Palme K. Auxin in action: Signalling, transport and the control of plant growth and development. Nat Rev Mol Cell Biol, 2006, 7:847–859
Galweiler L, Guan C, Muller A, et al. Regulation of polar auxin transport by AtPIN1 in Arabidopsis vascular tissue. Science, 1998, 282:2226–2230
Mattsson J, Sung Z R, Berleth T. Responses of plant vascular systems to auxin transport inhibition. Development, 1999, 126:2979–2991
Okada K, Ueda J, Komaki M K, et al. Requirement of the auxin polar transport system in early stages of Arabidopsis floral bud formation. Plant Cell, 1991, 3:677–684
Geisler M, Blakeslee J J, Bouchard R, et al. Cellular efflux of auxin catalyzed by the Arabidopsis MDR/PGP transporter AtPGP1. Plant J, 2005, 44:179–194
Noh B, Bandyopadhyay A, Peer W A, et al. Enhanced gravi- and phototropism in plant mdr mutants mislocalizing the auxin efflux protein PIN1. Nature, 2003, 423:999–1002
Noh B, Murphy A S, Spalding E P. Multidrug resistance-like genes of Arabidopsis required for auxin transport and auxin-mediated development. Plant Cell, 2001, 13:2441–2454
Friml J, Yang X, Michniewicz M, et al. A PINOID-dependent binary switch in apical-basal PIN polar targeting directs auxin efflux. Science, 2004, 306:862–865
Dai Y, Wang H, Li B, et al. Increased expression of MAP KINASE KINASE7 causes deficiency in polar auxin transport and leads to plant architectural abnormality in Arabidopsis. Plant Cell, 2006, 18:308–320
Hamann T, Benkova E, Baurle I, et al. The Arabidopsis BODENLOS gene encodes an auxin response protein inhibiting MONOPTEROSmediated embryo patterning. Genes Dev, 2002, 16:1610–1615
Hardtke C S, Berleth T. The Arabidopsis gene MONOPTEROS encodes a transcription factor mediating embryo axis formation and vascular development. EMBO J, 1998, 17:1405–1411
Harper R M, Stowe-Evans E L, Luesse D R, et al. The NPH4 locus encodes the auxin response factor ARF7, a conditional regulator of differential growth in aerial Arabidopsis tissue. Plant Cell, 2000, 12:757–770
Leyser H M, Pickett F B, Dharmasiri S, et al. Mutations in the AXR3 gene of Arabidopsis result in altered auxin response including ectopic expression from the SAUR-AC1 promoter. Plant J, 1996, 10:403–413
Tian Q, Reed J W. Control of auxin-regulated root development by the Arabidopsis thaliana SHY2/IAA3 gene. Development, 1999, 126:711–721
Zgurski J M, Sharma R, Bolokoski D A, et al. Asymmetric auxin response precedes asymmetric growth and differentiation of asymmetric leaf1 and asymmetric leaf2 Arabidopsis leaves. Plant Cell, 2005, 17:77–91
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work
About this article
Cite this article
Zhou, Y., Fang, Y., Zhu, J. et al. Genetic analysis and gene fine mapping of a rolling leaf mutant (rl 11(t) ) in rice (Oryza sativa L.). Chin. Sci. Bull. 55, 1763–1769 (2010). https://doi.org/10.1007/s11434-010-3137-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-010-3137-0