Abstract
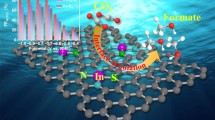
Clearly understanding the structure-function relationship and rational design of efficient CO2 electrocatalysts are still the challenges. This article describes the molecular origin of high selectivity of formic acid on N-doped SnO2 nanoparticles, which obtained via thermal treatment of g-C3N4 and SnCl2·2H2O precursor. Combined with density functional theory (DFT) calculations, we discover that N-doping effectively introduces oxygen vacancies and increases the charge density of Sn sites, which plays a positive role in CO2 activation. In addition, N-doping further regulates the adsorption energy of *OCHO, *COOH, *H and promotes HCOOH generation. Benefited from above modulation, the obtained N-doped SnO2 catalysts with oxygen vacancies (Ov-N-SnO2) exhibit faradaic efficiency of 93% for C1 formation, 88% for HCOOH production and well-suppression of H2 evolution over a wide range of potentials.
Similar content being viewed by others
References
Schreier M, Héroguel F, Steier L, Ahmad S, Luterbacher JS, Mayer MT, Luo J, Grätzel M. Nat Energy, 2017, 2: 17087
Jiang B, Zhang XG, Jiang K, Wu DY, Cai WB. J Am Chem Soc, 2018, 140: 2880–2889
Dai L, Qin Q, Wang P, Zhao X, Hu C, Liu P, Qin R, Chen M, Ou D, Xu C, Mo S, Wu B, Fu G, Zhang P, Zheng N. Sci Adv, 2017, 3: e1701069
Lu L, Sun X, Ma J, Zhu Q, Wu C, Yang D, Han B. Sci China Chem, 2018, 61: 228–235
Li X, He X, Liu X, He LN. Sci China Chem, 2017, 60: 841–852
Qiao J, Liu Y, Hong F, Zhang J. Chem Soc Rev, 2014, 43: 631–675
Abanades JC, Rubin ES, Mazzotti M, Herzog HJ. Energy Environ Sci, 2017, 10: 2491–2499
Sun Z, Ma T, Tao H, Fan Q, Han B. Chem, 2017, 3: 560–587
Koppenol WH, Rush JD. J Phys Chem, 1987, 91: 4429–4430
Wen G, Lee DU, Ren B, Hassan FM, Jiang G, Cano ZP, Gostick J, Croiset E, Bai Z, Yang L, Chen Z. Adv Energy Mater, 2018, 8: 1802427
Zhang S, Kang P, Meyer TJ. J Am Chem Soc, 2014, 136: 1734–1737
Li F, Chen L, Knowles GP, MacFarlane DR, Zhang J. Angew Chem Int Ed, 2017, 56: 505–509
Bai X, Chen W, Zhao C, Li S, Song Y, Ge R, Wei W, Sun Y. Angew Chem, 2017, 129: 12387–12391
Lei F, Liu W, Sun Y, Xu J, Liu K, Liang L, Yao T, Pan B, Wei S, Xie Y. Nat Commun, 2016, 7: 12697
Geng Z, Kong X, Chen W, Su H, Liu Y, Cai F, Wang G, Zeng J. Angew Chem Int Ed, 2018, 57: 6054–6059
Martin O, Martín AJ, Mondelli C, Mitchell S, Segawa TF, Hauert R, Drouilly C, Curulla-Ferré D, Pérez-Ramírez J. Angew Chem Int Ed, 2016, 55: 6261–6265
Zheng Y, Qiao SZ. Nat Chem, 2018, 10: 899–902
Chikhale LP, Patil JY, Shaikh FI, Rajgure AV, Pawar RC, Mulla IS, Suryavanshi SS. Mater Sci Semicon Proc, 2014, 27: 121–129
Li Q, Wang Z, Zhang M, Hou P, Kang P. J Energy Chem, 2017, 26: 825–829
Kim DW, Kim DH, Kim HJ, So HW, Hong MP. Curr Appl Phys, 2011, 11: S67–S72
Liang Y, Li Y, Wang H, Zhou J, Wang J, Regier T, Dai H. Nat Mater, 2011, 10: 780–786
Niu P, Zhang L, Liu G, Cheng HM. Adv Funct Mater, 2012, 22: 4763–4770
Liu X, Zhou K, Wang L, Wang B, Li Y. J Am Chem Soc, 2009, 131: 3140–3141
Fan JCC, Goodenough JB. J Appl Phys, 1977, 48: 3524–3531
Kar A, Kundu S, Patra A. J Phys Chem C, 2011, 115: 118–124
Choi WK, Jung HJ, Koh SK. J Vacuum Sci Tech A, 1996, 14: 359–366
Lin AWC, Armstrong NR, Kuwana T. Anal Chem, 1977, 49: 1228–1235
Wang J, Tafen DN, Lewis JP, Hong Z, Manivannan A, Zhi M, Li M, Wu N. J Am Chem Soc, 2009, 131: 12290–12297
Rogers C, Perkins WS, Veber G, Williams TE, Cloke RR, Fischer FR. J Am Chem Soc, 2017, 139: 4052–4061
Lu Q, Rosen J, Zhou Y, Hutchings GS, Kimmel YC, Chen JG, Jiao F. Nat Commun, 2014, 5: 3242
Gattrell M, Gupta N, Co A. J Electroanal Chem, 2006, 594: 1–19
Garand E, Wende T, Goebbert DJ, Bergmann R, Meijer G, Neumark DM, Asmis KR. J Am Chem Soc, 2010, 132: 849–856
Firet NJ, Smith WA. ACS Catal, 2016, 7: 606–612
Baruch MF, Pander Iii JE, White JL, Bocarsly AB. ACS Catal, 2015, 5: 3148–3156
Gao D, Zhou H, Cai F, Wang D, Hu Y, Jiang B, Cai WB, Chen X, Si R, Yang F, Miao S, Wang J, Wang G, Bao X. Nano Res, 2017, 10: 2181–2191
Acknowledgements
This work was supported by the National Key R&D Program of China (2016YFB0600901), the National Natural Science Foundation of China (21525626, 21606169, 21722608), and the Program of Introducing Talents of Discipline to Universities (B06006).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Hu, C., Zhang, L., Li, L. et al. Theory assisted design of N-doped tin oxides for enhanced electrochemical CO2 activation and reduction. Sci. China Chem. 62, 1030–1036 (2019). https://doi.org/10.1007/s11426-019-9474-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-019-9474-0