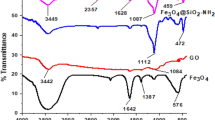
Abstract
Simple and efficient removal of Pb(II) ion from aqueous solution through adsorption has accelerated the development of many new composites to improve this popular method. In this study, the composites of graphene oxide (GO), zeolitic imidazolate framework-8 (ZIF-8), and magnetic materials were synthesized via coprecipitation method utilizing a different molar ratio between FeCl2 and FeCl3 of 1:0.5, 2:1, 3:1.5, and 4:2. The ZIF-8/GO was prepared via room temperature synthesis method prior to its further modification with magnetic materials for ease of separation. It was observed that the MZIF-8/GO2 of molar ratio 2:1 showed the best performance in adsorbing Pb(II) ion. As confirmed by FESEM image, it appeared to be ZIF-8 particles that have grown all over the GO platform and overlayed with Fe3O4 granular-shaped particles. The MZIF-8/GO2 successfully achieved 99% removal of Pb(II) within 10 min. The optimum values obtained for the initial concentration of Pb (II) were 100 mg/L, pH of 4 to 6, and adsorbent dosage used was 10 mg. The Langmuir isotherm and the pseudo-second-order kinetic model were deemed suitable to evaluate the adsorption of Pb(II) using MZIF-8/GO2. Results showed that MZIF-8GO2 achieved a maximum adsorption capacity of 625 mg/g of Pb(II) adsorption. All parent materials demonstrated a good synergistic effects, while exhibiting a significant contribution in providing active sites for Pb(II) adsorption. Therefore, this ternary composite of MZIF-8/GO2 is expected to be a promising adsorbent for Pb(II) adsorption from aqueous solution with an added value of ease of post phase separation using external magnetic field.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available, upon reasonable request.
References
Abdi J, Vossoughi M, Mohammad N (2017) Synthesis of metal-organic framework hybrid nanocomposites based on GO and CNT with high adsorption capacity for dye removal. Chem Eng J 326:1145–1158. https://doi.org/10.1016/j.cej.2017.06.054
Agnihotri A, Seth CS (2019) Chapter 11 - transgenic brassicaceae: a promising approach for phytoremediation of heavy metals. In: Prasad MNV (ed) Transgenic Plant Technology for Remediation of Toxic Metals and Metalloids. Elsevier Inc., Amsterdam, pp 239–255. https://doi.org/10.1016/B978-0-12-814389-6.00011-0
Agnihotri A, Seth CS (2020) Does jasmonic acid regulate photosynthesis, clastogenecity, and phytochelatins in Brassica juncea L. in response to Pb-subcellular distribution? Chemosphere 243:125361. https://doi.org/10.1016/j.chemosphere.2019.125361
Ahmad SZN, Salleh WNW, Ismail NH, Razali NAM, Hamdan R, Ismail AF (2021a) Effects of operating parameters on cadmium removal for wastewater treatment using zeolitic imidazolate framework-L/graphene oxide composite. J Environ Chem Eng 9:106139. https://doi.org/10.1016/j.jece.2021.106139
Ahmad SZN, Salleh WNW, Ismail NH, Rosman N, Razali NAM, Hamdan R, Ismail AF (2021b) Zeolitic imidazolate framework-L incorporated graphene oxide hybrid for cadmium removal. Mater Today Proc 42:8–14. https://doi.org/10.1016/j.matpr.2020.08.339
Ahmad SZN, Salleh WNW, Yusof N, Yusop MZM, Hamdan R, Ismail AF (2022) Synthesis of zeolitic imidazolate framework-8 (ZIF-8) using different solvents for lead and cadmium adsorption. Appl Nanosci 13:4005–4019. https://doi.org/10.1007/s13204-022-02680-7
Ahmad SZN, Salleh WNW, Yusop MZM, Hamdan R, Aziz F, Awang NA, Ismail AF (2023) Synthesis of zeolitic imidazolate framework-8 modified graphene oxide composite and its application for lead removal. J Chem Technol Biotechnol 98:2668–2676. https://doi.org/10.1002/jctb.7337
Ahmed W, Nunez-Delgado A, Mehmood S, Ali S, Qaswar M, Shakoor A, Chen D (2021) Highly efficient uranium (VI) capture from aqueous solution by means of a hydroxyapatite-biochar nanocomposite: adsorption behavior and mechanism. Environ Res 201:111518. https://doi.org/10.1016/j.envres.2021.111518
Beatrice A, Varco JJ, Dygert A, Atsar FS, Solomon S, Thirumalai RVKG, Pittman CU Jr, Mlsna T (2022) Lead immobilization in simulated polluted soil by Douglas fir biochar-supported phosphate. Chemosphere 292:133355. https://doi.org/10.1016/j.chemosphere.2021.133355
Chen K, Ni L, Zhang H, Li L, Guo X, Qi J, Zhou Y, Zhu Z, Sun X, Li J (2023) Phenolic resin regulated interface of ZIF-8 based mixed matrix membrane for enhanced gas separation. J Memb Sci 666:121117. https://doi.org/10.1016/j.memsci.2022.121117
Chirra S, Wang L, Aggarwal H, Tsai M, Soorian SS, Siliveri S, Goskula S, Gujjula SR, Narayanan V (2021) Rapid synthesis of a novel nano-crystalline mesoporous faujasite type metal-organic framework, ZIF-8 catalyst, its detailed characterization, and NaBH 4 assisted, enhanced catalytic Rhodamine B degradation. Mater Today Commun 26:101993. https://doi.org/10.1016/j.mtcomm.2020.101993
Cui L, Wang Y, Hu L, Gao L, Du B, Wei Q (2015) Mechanism of Pb(ii) and methylene blue adsorption onto magnetic carbonate hydroxyapatite/graphene oxide. RSC Adv 5:9759–9770. https://doi.org/10.1039/C4RA13009J
Dave PN, Macwan PM (2024) 6 Functionalized conductive polymenr-based thin film coating as corrosion inhibitor. In: Kumar A, Verma C, Thakur A (eds) Corrosion Mitigation Coatings. De Gruyter, Berlin, pp 115–158. https://doi.org/10.1515/9783111016160-006
Dave PN, Macwan PM, Kamaliya BP (2023c) Synthesis and characterization of biodegradable gum ghatti-cl-poly(AA-co-NIPAm)/GO based hydrogel for metformin and sodium diclofenac combined drug delivery system. Colloids Surfaces A Physicochem Eng Asp 673:131815. https://doi.org/10.1016/j.colsurfa.2023.131815
Dave PN, Macwan PM, Kamaliya BP (2023d) Preparation and enhancing properties of pH-sensitive hydrogel in light of gum ghatti-cl-poly(acrylic acid)/ – o-MWCNT for sodium diclofenac drug release. Macromol Chem Phys 224:2300038. https://doi.org/10.1002/macp.202300038
Dave PN, Macwan PM, Kamaliya B (2023a) Biodegradable Gg-cl-poly(NIPAm-co-AA)/-o-MWCNT based hydrogel for combined drug delivery system of metformin and sodium diclofenac: in vitro studies. RSC Adv 33. https://doi.org/10.1039/D3RA04728H
Dave PN, Macwan PM, Kamaliya BP (2023b) Synthesis and rheological investigations of gum-ghatti-cl-poly(NIPA-co-AA)-graphene oxide based hydrogels. Mater Adv 14. https://doi.org/10.1039/D3MA00092C
Dave PN, Macwan PM, Kamaliya BP (2024) The effect of adding cobalt ferrite (CoFe3O4) nanoparticles as fillers on rheological and structural behaviour of gum ghatti-cl-poly(NIPAm) hydrogels. Mech Time-Dependent Mater https://doi.org/10.1007/s11043-024-09676-6
Ekinci S, Ilter Z, Ercan S, Cinar E, Cakmak R (2021) Magnetite nanoparticles grafted with murexide-terminated polyamidoamine dendrimers for removal of lead (II) from aqueous solution: synthesis, characterization, adsorption and antimicrobial activity studies. Heliyon 7:e06600. https://doi.org/10.1016/j.heliyon.2021.e06600
Gnanaprakash G, Mahadevan S, Jayakumar T, Kalyanasundaram P, Philip J, Raj B (2007) Effect of initial pH and temperature of iron salt solutions on formation of magnetite nanoparticles. Mater Chem Phys 103:168–175. https://doi.org/10.1016/j.matchemphys.2007.02.011
Gupta P, Seth CS (2019) Nitrate supplementation attenuates As(V) toxicity in Solanum lycopersicum L. cv Pusa Rohini: Insights into As(V) sub-cellular distribution, photosynthesis, nitrogen assimilation, and DNA damage. Plant Physiol Biochem 139:44–55. https://doi.org/10.1016/j.plaphy.2019.03.007
Hu L, Yang Z, Cui L, Li Y, Hao H, Wang Y, Wei Q, Ma H, Yan L, Du B (2016) Fabrication of hyperbranched polyamine functionalized graphene for high-efficiency removal of Pb (II) and methylene blue. Chem Eng J 287:545–556. https://doi.org/10.1016/j.cej.2015.11.059
Imran M, Haq Khan ZU, Iqbal J, Shah NS, Muzammil S, Ali S, Muhammad N, Aziz A, Murtaza B, Naeem MA, Amjad M, Shahid M, Zakir A, Rizwan M (2020) Potential of siltstone and its composites with biochar and magnetite nanoparticles for the removal of cadmium from contaminated aqueous solutions: Batch and column scale studies. Environ Pollut 259:113938. https://doi.org/10.1016/j.envpol.2020.113938
Jia Y, Zou Y, Zou X, Jiang Y, Song S, Qin J, Wang Y, Zhu L (2023) Study on the adsorption performance of multi-base composite magnesia cementitious material microfiltration membrane for different heavy metal ions. Mater Lett 335:133488. https://doi.org/10.1016/j.matlet.2022.133488
Kalam S, Abu-Khamsin SA, Kamal MS, Patil S (2021) Surfactant Adsorption Isotherms: a Review. ACS Omega 6:32342–32348. https://doi.org/10.1021/acsomega.1c04661
Kamaliya BP, Dave PN, Chopda LV (2023) Synthesis of GG- g-P(NIPAM- co-AA)/GO and evaluation of adsorption activity for the diclofenac and metformin. J Environ Heal Sci Eng 21:403–416. https://doi.org/10.1007/s40201-023-00867-w
Kim DW, Eum K, Kim H, Kim D, Mello M, Park K, Tsapatsis M (2019) Continuous ZIF-8/reduced graphene oxide nanocoating for ultrafast oil/water separation. Chem Eng J 372:509–515. https://doi.org/10.1016/j.cej.2019.04.179
Kumar D, Singh R, Upadhyay SK, Verma KK, Tripathi RM, Liu H, Dhankher OP, Tripathi RD, Sahi SV, Seth CS (2024) Review on interactions between nanomaterials and phytohormones: Novel perspectives and opportunities for mitigating environmental challenges. Plant Sci 340:111964. https://doi.org/10.1016/j.plantsci.2023.111964
Lange A, Sawosz E, Wierzbicki M, Kutwin M, Daniluk K, Strojny B, Ostrowska A, Wojcik B, Lojkowski M, Golebiewski M, Chwalibog A, Jaworski S (2022) Nanocomposites of graphene oxide — silver nanoparticles for enhanced antibacterial activity: mechanism of action and medical textiles coating. Mat 15:3122. https://doi.org/10.3390/ma15093122
Lee S, Jeong D, Kim C, Lee C, Kang H, Woo HY, Kim BJ (2020) Eco-friendly polymer solar cells: advances in green-solvent processing and material design. ACS Nano 14:14493–14527. https://doi.org/10.1021/acsnano.0c07488
Li K, Miwornunyuie N, Chen L, Jingyu H, Amaniampong PS, Koomson DA, Ewusi-Mensah D, Xue W, Li G, Lu H (2021) Sustainable application of ZIF-8 for heavy-metal removal in aqueous solutions. Sustain 13:1–11. https://doi.org/10.3390/su13020984
Li H, Jiang Q, Zhang J, Wang Y, Zhang Y (2022) Synchronization adsorption of Pb(II) and Ce(III) by biochar supported phosphate-doped ferrihydrite in aqueous solution: adsorption efficiency and mechanisms. Colloids Surfaces A Physicochem Eng Asp 648:129230. https://doi.org/10.1016/j.colsurfa.2022.129230
Lu Z, Yu J, Zeng H, Liu Q (2017) Polyamine-modified magnetic graphene oxide nanocomposite for enhanced selenium removal. Sep Purif Technol 183:249–257. https://doi.org/10.1016/j.seppur.2017.04.010
Lu M, Chen Y, Chen S, Liu J, Mei M, Wang T, Li J (2023) Potentiality of the porous geopolymer sphere in adsorption of Pb (II) from aqueous solutions: behaviors and mechanisms. Ceram Int 49:698–706. https://doi.org/10.1016/j.ceramint.2022.09.040
Mahmoodi NM, Oveisi M, Bakhtiari M, Hayati B, Akbar A, Bagheri A, Rahimi S (2019) Environmentally friendly ultrasound-assisted synthesis of magnetic zeolitic imidazolate framework - graphene oxide nanocomposites and pollutant removal from water. J Mol Liq 282:115–130. https://doi.org/10.1016/j.molliq.2019.02.139
Mahmoud ME, Khalifa MA, El-Mallah NM, Hassouba HM, Nabil GM (2021) Performance of MnO2 nanoparticles-coated cationic CTAB for detoxification and decolorization of sulfonated remazol red and reactive black 5 dyes from water. Int J Environ Sci Technol 19:141–158. https://doi.org/10.1007/s13762-021-03153-0
Mariyam S, Upadhyay SK, Chakraborty K, Verma KK, Duhan JS, Muneer S, Meena M, Sharma RK, Ghodake G, Seth CS (2024) Nanotechnology, a frontier in agricultural science, a novel approach in abiotic stress management and convergence with new age medicine-a review. Sci Total Environ 912:169097. https://doi.org/10.1016/j.scitotenv.2023.169097
Masibi EG, Makhetha TA, Moutloali RM (2022) Effect of the incorporation of ZIF-8@GO into the thin-film membrane on salt rejection and BSA fouling. Membranes (basel) 12:436. https://doi.org/10.3390/membranes12040436
Mojoudi F, Hamidian AH, Goodarzian N, Eagderi S (2018) Effective removal of heavy metals from aqueous solution by porous activated carbon/thiol functionalized graphene oxide composite. Desalin Water Treat 124:106–116. https://doi.org/10.5004/dwt.2018.22695
Nyairo WN, Eker YR, Tor A, Ongeri DM (2017) Efficient removal of lead(II) ions from aqueous solutions using methyl-β-cyclodextrin modified graphene oxide. Water Air Soil Pollut 228:406. https://doi.org/10.1007/s11270-017-3589-9
Othman FEC, Yusof N, Hasbullah H, Jaafar J, Ismail AF, Abdullah N, Nordin NAHM, Aziz F, Salleh WNW (2017) Polyacrylonitrile/magnesium oxide-based activated carbon nanofibers with well-developed microporous structure and their adsorption performance for methane. J Ind Eng Chem 51:281–287. https://doi.org/10.1016/j.jiec.2017.03.014
Pan Y, Liu Y, Zeng G, Zhao L, Lai Z (2011) Rapid synthesis of zeolitic imidazolate framework-8 (ZIF-8) nanocrystals in an aqueous system. Chem Commun 47:2071–2073. https://doi.org/10.1039/C0CC05002D
Pap S, Šolević Knudsen T, Radonić J, Maletić S, Igić SM, Turk Sekulić M (2017) Utilization of fruit processing industry waste as green activated carbon for the treatment of heavy metals and chlorophenols contaminated water. J Clean Prod 162:958–972. https://doi.org/10.1016/j.jclepro.2017.06.083
Phouthavong V, Yan R, Nijpanich S, Hagio T, Ichino R, Kong L, Li L (2022) Magnetic adsorbents for wastewater treatment: advancements in their synthesis methods. Materials (basel) 15:1053. https://doi.org/10.3390/ma15031053
Pirouz AA, Selam J, Iqbal SZ, Mirhoss H, Karjiban RA, Bakar FA (2017) The use of innovative and efficient nanocomposite (magnetic graphene oxide) for the reduction on of Fusarium mycotoxins in palm kernel cake. Sci Rep 7:12453. https://doi.org/10.1038/s41598-017-12341-3
Pirveysian M, Ghiaci M (2018) Synthesis and characterization of sulfur functionalized graphene oxide nanosheets as efficient sorbent for removal of Pb2+, Cd2+, Ni2+ and Zn2+ ions from aqueous solution: A combined thermodynamic and kinetic studies. Appl Surf Sci 428:98–109. https://doi.org/10.1016/j.apsusc.2017.09.105
Pokhrel J, Bhoria N, Anastasiou S, Tsoufis T (2018) CO2 adsorption behavior of amine-functionalized ZIF-8, graphene oxide, and ZIF-8/ graphene oxide composites under dry and wet conditions. Microporous Mesoporous Mater 267:53–67. https://doi.org/10.1016/j.micromeso.2018.03.012
Ranjbari S, Ayati A, Shahrak MN, Tanhaei B, Tabrizi SH (2023) Design of [BmIm]3PW12O40 ionic liquid encapsulated-ZIF-8 nanocomposite for cationic dye adsorptive removal: modeling by response surface methodology. Ind Eng Chem Res 62:4636–4645. https://doi.org/10.1021/acs.iecr.2c02943
Sahraei R, Hemmati K, Ghaemy M (2016) Adsorptive removal of toxic metals and cationic dyes by magnetic adsorbent based on functionalized graphene oxide from water. RSC Adv 6:72487–72499. https://doi.org/10.1039/C6RA12934J
Tejada-Tovar C, Bonilla-Mancilla H, Pino-Moreyra JD, Villabona-Ortiz A, Ortega-Toro R (2020) Effect of the adsorbent dose in Pb(II) removal using sugar cane bagasse: kinetics and isotherms. Rev Mex Ingeniería Química 19:1413–1423. https://doi.org/10.24275/rmiq%2Fia1101
Thommes M, Kaneko K, Neimark AV, Olivier JP, Rodriguez-reinoso F, Rouquerol J, Sing KSW (2015) Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl Chem 87:1051–1069. https://doi.org/10.1515/pac-2014-1117
Wang X, Jiang C, Hou B, Wang Y, Hao C, Wu J (2018) Carbon composite lignin-based adsorbents for the adsorption of dyes. Chemosphere 206:587–596. https://doi.org/10.1016/j.chemosphere.2018.04.183
Wang J, Li Y, Lv Z, Xie Y, Shu J, Alsaedi A, Hayat T, Chen C (2019) Exploration of the adsorption performance and mechanism of zeolitic imidazolate framework-8 @ graphene oxide for Pb(II) and 1-naphthylamine from aqueous solution. J Colloid Interface Sci 542:410–420. https://doi.org/10.1016/j.jcis.2019.02.039
Wang J, Zhan J, Zhou H, Yi X, Liu Y (2023) Removal of lead from aqueous solution by synergistic interaction between LiCoO2 and tourmaline: synthesis, characterization and mechanism investigation. Appl Surf Sci 631:157485. https://doi.org/10.1016/j.apsusc.2023.157485
Xia Y, Yang T, Zhu N, Li D, Chen Z, Lang Q, Liu Z, Jiao W (2019) Enhanced adsorption of Pb(II) onto modified hydrochar: modeling and mechanism analysis. Biores 288:121593. https://doi.org/10.1016/j.biortech.2019.121593
Xue ZF, Cheng WC, Xie YX, Wang L, Hu W, Zhang B (2023) Investigating immobilization efficiency of Pb in solution and loess soil using bio-inspired carbonate precipitation. Environ Pollut 322:121218. https://doi.org/10.1016/j.envpol.2023.121218
Yang W, Cao M (2023) Synthesis of ZIF-8@GO-COOH and its adsorption for Cu(II) and Pb(II) from water: capability and mechanism. Sep Purif Technol 309:122957. https://doi.org/10.1016/j.seppur.2022.122957
Yang G, Tang L, Lei X, Zeng G, Cai Y, Wei X, Zhou Y, Li S, Fang Y, Zhang Y (2014) Cd(II) removal from aqueous solution by adsorption on α-ketoglutaric acid-modified magnetic chitosan. Appl Surf Sci 292:710–716. https://doi.org/10.1016/j.apsusc.2013.12.038
Yang X, Chen L, Ren D, Wang S, Ren Z (2022) Adsorption of Pb(II) from water by treatment with an O-hydroxyphenyl thiourea-modified chitosan. Int J Biol Macromol 220:280–290. https://doi.org/10.1016/j.ijbiomac.2022.08.090
Yang W, Kong Y, Yin H, Cao M (2023) Study on the adsorption performance of ZIF-8 on heavy metal ions in water and the recycling of waste ZIF-8 in cement. J Solid State Chem 326:124217. https://doi.org/10.1016/j.jssc.2023.124217
Zhou K, Chaemchuen S, Wu Z, Verpoort F (2017) Rapid room temperature synthesis forming pillared metal-organic frameworks with Kagomé net topology. Microporous Mesoporous Mater 239:28–33. https://doi.org/10.1016/j.micromeso.2016.09.038
Zhou J, Liu Y, Li B, Li H, Chen G, Qiu R (2023) Coagulation of trace arsenic and cadmium from drinking water using titanium potassium oxalate. npj Clean Water 6:9. https://doi.org/10.1038/s41545-023-00227-z
Zhu J, Zeng G, Nie F, Xu X, Chen S, Han Q, Wang X (2010) Decorating graphene oxide with CuOnanoparticles in a water–isopropanol system. Nanoscale 2:988–994. https://doi.org/10.1039/B9NR00414A
Funding
This work was supported by the Universiti Teknologi Malaysia under UTM Encouragement Research (project number Q.J130000.3846.31J70).
Author information
Authors and Affiliations
Contributions
All authors contributed to the conceptualization. Material preparation, data collection, analysis, and original draft were performed by Siti Zu Nurain Ahmad. All authors commented on the previous versions of manuscript. Equipment and material resources were provided by Wan Norharyati Wan Salleh, Norhaniza Yusof, Mohd Zamri Mohd Yusop, and Rafidah Hamdan. Funding was acquired by Wan Norharyati Wan Salleh and Ahmad Fauzi Ismail. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Angeles Blanco
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zu Nurain Ahmad, S., Salleh, W.N.W., Yusof, N. et al. Efficiency of magnetic zeolitic imidazolate framework-8 (ZIF-8) modified graphene oxide for lead adsorption. Environ Sci Pollut Res (2024). https://doi.org/10.1007/s11356-024-33322-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11356-024-33322-w