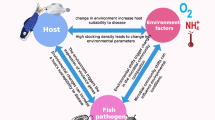
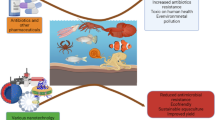
Abstract
Pathogenic bacteria, the major causative agents of aquaculture diseases, are a serious impediment to the aquaculture industry. However, the bioinformatics of pathogenic bacteria and virulence factors (VFs) in sediments, an important component of freshwater aquaculture ecosystems, are not well characterized. In this study, 20 sediment samples were collected from fish pond sediments (FPS), shrimp field sediments (SFS), fish pond sediment control (FPSC), and shrimp field sediment control (SFSC). Molecular biological information was obtained on a total of 173 pathogenic bacteria, 1093 virulence factors (VFs), and 8475 mobile genetic elements (MGEs) from these samples. The results indicated that (1) aquaculture patterns and sediment characteristics can affect the distribution of pathogenic bacteria. According to the results of the Kruskal–Wallis H test, except for Mycobacterium gilvum, there were significant differences (P < 0.05) among the four sediment types in the average abundance of major pathogenic bacteria (top 30 in abundance), and the average abundance of major pathogenic bacteria in the four sediment types followed the following pattern: FPS > SFS > FPSC > SFSC. (2) Pathogenic bacteria are able to implement a variety of complex pathogenic mechanisms such as adhesion, invasion, immune evasion, and metabolic regulation in the host because they carry a variety of VFs such as type IV pili, HSI-I, Alginate, Colibactin, and Capsule. According to the primary classification of the Virulence Factor Database (VFDB), the abundance of VFs in all four types of sediments showed the following pattern: offensive VFs > non-specific VFs > defensive VFs > regulation of virulence-related genes. (3) Total organic carbon (TOC), total phosphorus (TP), available phosphorus (AP), nitrite, and nitrate were mostly only weakly positively correlated with the major pathogenic bacteria and could promote the growth of pathogenic bacteria to some extent, whereas ammonia was significantly positively correlated with most of the major pathogenic bacteria and could play an important role in promoting the growth and reproduction of pathogenic bacteria. (4) Meanwhile, there was also a significant positive correlation between CAZyme genes and major pathogenic bacteria (0.62 ≤ R ≤ 0.89, P < 0.05). This suggests that these pathogenic bacteria could be the main carriers of CAZyme genes and, to some extent, gained a higher level of metabolic activity by degrading organic matter in the sediments to maintain their competitive advantage. (5) Worryingly, the results of correlation analyses indicated that MGEs in aquaculture sediments could play an important role in the spread of VFs (R = 0.82, P < 0.01), and in particular, plasmids (R = 0.75, P < 0.01) and integrative and conjugative elements (ICEs, R = 0.65, P < 0.05) could be these major vectors of VFs. The results of this study contribute to a comprehensive understanding of the health of freshwater aquaculture sediments and provide a scientific basis for aquaculture management and conservation.








Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Albertson LK, Daniels MD (2016) Effects of invasive crayfish on fine sediment accumulation, gravel movement, and macroinvertebrate communities. Freshw Sci 35(2):644–653. https://doi.org/10.1086/685860
Alvarez-Martinez CE, Christie PJ (2009) Biological diversity of prokaryotic type IV secretion systems. Microbiol Mol Biol Rev 73(4):775–808. https://doi.org/10.1128/MMBR.00023-09
Aly SM, Albutti A (2014) Antimicrobials use in aquaculture and their public health impact. J Aquac Res Dev 5(4):1. https://doi.org/10.4172/2155-9546.1000247
Anderson DS, Adhikari P, Nowalk AJ, Chen CY, Mietzner TA (2004) The hFbpABC transporter from Haemophilus influenzae functions as a binding-protein-dependent ABC transporter with high specificity and affinity for ferric iron. J Bacteriol 186(18):6220–6229. https://doi.org/10.1128/JB.186.18.6220-6229.2004
Andrade AC, Fróes A, Lopes FÁC, Thompson FL, Krüger RH, Dinsdale E, Bruce T (2017) Diversity of microbial carbohydrate-active enZYmes (CAZYmes) associated with freshwater and soil samples from Caatinga biome. Microb Ecol 74:89–105. https://doi.org/10.1007/s00248-016-0911-9
Armendáriz-Ruiz M, Rodríguez-González JA, Camacho-Ruíz RM, Mateos-Díaz JC (2018) Carbohydrate esterases: an overview. Lipases and phospholipases: methods and protocols 39–68. https://doi.org/10.1007/978-1-4939-8672-9_2
Austin B, Austin DA, Munn C (2007) Bacterial fish pathogens: disease of farmed and wild fish. Springer, The Netherlands, p 552
Avnimelech Y, Ritvo G (2003) Shrimp and fish pond soils: processes and management. Aquaculture 220(1–4):549–567. https://doi.org/10.1016/S0044-8486(02)00641-5
Bachman MA, Oyler JE, Burns SH, Caza M, Lépine F, Dozois CM, Weiser JN (2011) Klebsiella pneumoniae yersiniabactin promotes respiratory tract infection through evasion of lipocalin 2. Infect Immun 79(8):3309–3316. https://doi.org/10.1128/IAI.05114-11
Bleves S, Viarre V, Salacha R, Michel GPF, Filloux A, Voulhoux R (2010) Protein secretion systems in Pseudomonas aeruginosa: a wealth of pathogenic weapons. Int J Med Microbiol 300(8):534–543. https://doi.org/10.1016/j.ijmm.2010.08.005
Brige A, Cole JA, Hagen WR, Guisez Y, Van Beeumen JJ (2001) Overproduction, purification and novel redox properties of the dihaem cytochrome c, NapB, from Haemophilus influenzae. Biochem J 356(3):851–858. https://doi.org/10.1042/bj3560851
Broman T, Thelaus J, Andersson AC, Bäckman S, Wikström P, Larsson E, Granberg M, Karlsson L, Bäck E, Eliasson H, Mattsson R, Sjöstedt A, Forsman M (2011) Molecular detection of persistent Francisella tularensis subspecies holarctica in natural waters. Int J Microbiol 2011:851946. https://doi.org/10.1155/2011/851946
Buchfink B, Reuter K, Drost H-G (2021) Sensitive protein alignments at tree-of-life scale using DIAMOND. Nat Methods 18(4):366–368. https://doi.org/10.1038/s41592-021-01101-x
Cavalieri SJ, Bohach GA, Snyder I (1984) Escherichia coli alpha-hemolysin: characteristics and probable role in pathogenicity. Microbiol Rev 48(4):326–343. https://doi.org/10.1128/mr.48.4.326-343.1984
Chadha J, Harjai K, Chhibber S (2022) Revisiting the virulence hallmarks of Pseudomonas aeruginosa: a chronicle through the perspective of quorum sensing. Environ Microbiol 24(6):2630–2656. https://doi.org/10.1111/1462-2920.15784
Chen L, Yang J, Yu J, Yao Z, Sun L, Shen Y, Jin Q (2005) VFDB: a reference database for bacterial virulence factors. Nucleic Acids Res 33(suppl_1):D325–D328. https://doi.org/10.1093/nar/gki008
Christiansen C, Abou Hachem M, Janeček Š, Viksø-Nielsen A, Blennow A, Svensson B (2009) The carbohydrate-binding module family 20–diversity, structure, and function. FEBS J 276(18):5006–5029. https://doi.org/10.1111/j.1742-4658.2009.07221.x
Cianciotto NP, White RC (2017) Expanding role of type II secretion in bacterial pathogenesis and beyond. Infect Immun 85(5):10–1128. https://doi.org/10.1128/iai.00014-17
Coburn B, Sekirov I, Finlay BB (2007) Type III secretion systems and disease. Clin Microbiol Rev 20(4):535–549. https://doi.org/10.1128/cmr.00013-07
Craig L, Pique ME, Tainer JA (2004) Type IV pilus structure and bacterial pathogenicity. Nat Rev Microbiol 2(5):363–378. https://doi.org/10.1038/nrmicro885
Cress BF, Englaender JA, He W, Kasper D, Linhardt RJ, Koffas MA (2014) Masquerading microbial pathogens: capsular polysaccharides mimic host-tissue molecules. FEMS Microbiol Rev 38(4):660–697. https://doi.org/10.1111/1574-6976.12056
Damashek J, Francis CA (2018) Microbial nitrogen cycling in estuaries: from genes to ecosystem processes. Estuaries Coasts 41(3):626–660. https://doi.org/10.1007/s12237-017-0306-2
Darshanee Ruwandeepika HA, Sanjeewa Prasad Jayaweera T, Paban Bhowmick P, Karunasagar I, Bossier P, Defoirdt T (2012) Pathogenesis, virulence factors and virulence regulation of vibrios belonging to the Harveyi clade. Rev Aquac 4(2):59–74. https://doi.org/10.1111/j.1753-5131.2012.01061.x
De la Cruz F, Davies J (2000) Horizontal gene transfer and the origin of species: lessons from bacteria. Trends Microbiol 8(3):128–133. https://doi.org/10.1016/S0966-842X(00)01703-0
Defoirdt T (2014) Virulence mechanisms of bacterial aquaculture pathogens and antivirulence therapy for aquaculture. Rev Aquac 6(2):100–114. https://doi.org/10.1111/raq.12030
Donnenberg MS (2000) Pathogenic strategies of enteric bacteria. Nature 406(6797):768–774. https://doi.org/10.1038/35021212
Du F, Hua L, Zhai L, Zhang F, Fan X, Wang S, Liu Y, Liu H (2022) Rice-crayfish pattern in irrigation-drainage unit increased N runoff losses and facilitated N enrichment in ditches. Sci Total Environ 848:157721. https://doi.org/10.1016/j.scitotenv.2022.157721
Egidius E (1987) Vibriosis: pathogenicity and pathology. A review. Aquaculture 67(1–2):15–28. https://doi.org/10.1016/0044-8486(87)90004-4
Emenike EC, Iwuozor KO, Anidiobi SU (2021) Heavy metal pollution in aquaculture: sources, impacts and mitigation techniques. Biol Trace Elem Res 1–17. https://doi.org/10.1007/s12011-021-03037-x
Farver CF, Zander DS (2009) Chapter 18 - molecular basis of pulmonary disease. In: Coleman WB, Tsongalis GJ (eds) Molecular pathology. Academic Press, San Diego, pp 305–364
Feng J, Pan R, Hu H-W, Huang Q, Zheng J, Tan W, Liu Y-R, Delgado-Baquerizo M (2023) Effects of integrated rice-crayfish farming on soil biodiversity and functions. Sci Bull 68(20):2311–2315. https://doi.org/10.1016/j.scib.2023.08.037
Fuentes DE, Acuña LG, Calderón IL (2022) Stress response and virulence factors in bacterial pathogens relevant for Chilean aquaculture: current status and outlook of our knowledge. Biol Res 55(1):1–9. https://doi.org/10.1186/s40659-022-00391-5
Gamito L, Karakassis L, Pederson L, Smaal L (2001) Use of hydrodynamic and benthic models for managing environmental impacts of marine aquaculture. J Appl Ichthyol 17(4):163–172. https://doi.org/10.1046/j.1439-0426.2001.00313.x
Ganesh EA, Das S, Chandrasekar K, Arun G, Balamurugan S (2010) Monitoring of total heterotrophic bacteria and Vibrio spp. in an aquaculture pond. Curr Res J Biol Sci 2(1):48–52
Geraldine AR, Herawati H, Bishina I (2020) Isolation and identification of potential pathogenic bacteria in living carp (Cyprinus carpio Linnaeus, 1758) sold in supermarkets in Cimahi City, Java. World News Nat Sci 32:21–35
Hallam JC, Sandalli S, Floria I, Turner NC, Tang-Fichaux M, Oswald E, O’Boyle N, Roe AJ (2023) D-Serine reduces the expression of the cytopathic genotoxin colibactin. Microb Cell 10(3):63. https://doi.org/10.15698/mic2023.03.793
Handing JW, Ragland SA, Bharathan UV, Criss AK (2018) The MtrCDE efflux pump contributes to survival of Neisseria gonorrhoeae from human neutrophils and their antimicrobial components. Front Microbiol 9:2688. https://doi.org/10.3389/fmicb.2018.02688
Hanoi VN (2013) Report of the FAO/MARD technical workshop on early mortality syndrome (EMS) or acute hepatopancreatic necrosis syndrome (AHPNS) of cultured shrimp (under TCP/VIE/3304). FAO Rome. https://www.fao.org/3/i3422e/i3422e.pdf. Accessed 25–27 June 2013
Hau T (1990) Bacteria, toxins, and the peritoneum. World J Surg 14(2):167–175. https://doi.org/10.1007/BF01664869
Heiniger RW, Winther-Larsen HC, Pickles RJ, Koomey M, Wolfgang MC (2010) Infection of human mucosal tissue by Pseudomonas aeruginosa requires sequential and mutually dependent virulence factors and a novel pilus-associated adhesin. Cell Microbiol 12(8):1158–1173. https://doi.org/10.1111/j.1462-5822.2010.01461.x
Holmer M, Wildish D, Hargrave B (2005) Organic enrichment from marine finfish aquaculture and effects on sediment biogeochemical processes. Environ Effects Mar Finfish Aquac 181–206. https://doi.org/10.1007/b136010
Hu X, Zhang R, Ye J, Wu X, Zhang Y, Wu C (2018) Monitoring and research of microcystins and environmental factors in a typical artificial freshwater aquaculture pond. Environ Sci Pollut Res 25:5921–5933. https://doi.org/10.1007/s11356-017-0956-4
Huang F, Zhao Y (2022) Global control of tuberculosis: current status and future prospects. Zoonoses 2(1). https://doi.org/10.15212/ZOONOSES-2021-0021
Hui X, Chen Z, Zhang J, Lu M, Cai X, Deng Y, Hu Y, Wang Y (2021) Computational prediction of secreted proteins in gram-negative bacteria. Comput Struct Biotechnol J 19:1806–1828. https://doi.org/10.1016/j.csbj.2021.03.019
Hyams C, Yuste J, Bax K, Camberlein E, Weiser JN, Brown JS (2010) Streptococcus pneumoniae resistance to complement-mediated immunity is dependent on the capsular serotype. Infect Immun 78(2):716–725. https://doi.org/10.1128/IAI.01056-09
Igbinosa EO (2016) Detection and antimicrobial resistance of Vibrio isolates in aquaculture environments: implications for public health. Microb Drug Resist 22(3):238–245. https://doi.org/10.1089/mdr.2015.0169
Jia X, Yang L, Dong M, Chen S, Lv L, Cao D, Fu J, Yang T, Zhang J, Zhang X (2017) The bioinformatics analysis of comparative genomics of Mycobacterium tuberculosis complex (MTBC) provides insight into dissimilarities between intraspecific groups differing in host association, virulence, and epitope diversity. Front Cell Infect Microbiol 7:88. https://doi.org/10.3389/fcimb.2017.00088
Johnson CM, Grossman AD (2015) Integrative and conjugative elements (ICEs): what they do and how they work. Annu Rev Genet 49:577–601. https://doi.org/10.1146/annurev-genet-112414-055018
Lairson L, Henrissat B, Davies G, Withers S (2008) Glycosyltransferases: structures, functions, and mechanisms. Annu Rev Biochem 77:521–555. https://doi.org/10.1146/annurev.biochem.76.061005.092322
Lam MM, Wick RR, Wyres KL, Gorrie CL, Judd LM, Jenney AW, Brisse S, Holt KE (2018) Genetic diversity, mobilisation and spread of the yersiniabactin-encoding mobile element ICEKp in Klebsiella pneumoniae populations. Microb Genom 4(9). https://doi.org/10.1099/mgen.0.000196
Lammens W, Le Roy K, Schroeven L, Van Laere A, Rabijns A, Van den Ende W (2009) Structural insights into glycoside hydrolase family 32 and 68 enzymes: functional implications. J Exp Bot 60(3):727–740. https://doi.org/10.1093/jxb/ern333
Levasseur A, Drula E, Lombard V, Coutinho PM, Henrissat B (2013) Expansion of the enzymatic repertoire of the CAZy database to integrate auxiliary redox enzymes. Biotechnol Biofuels 6(1):1–14. https://doi.org/10.1186/1754-6834-6-41
Li F, Feng J, Zhou X, Xu C, Haissam Jijakli M, Zhang W, Fang F (2019a) Impact of rice-fish/shrimp co-culture on the N2O emission and NH3 volatilization in intensive aquaculture ponds. Sci Total Environ 655:284–291. https://doi.org/10.1016/j.scitotenv.2018.10.440
Li Y, Gao Y, Zhang W, Wang C, Wang P, Niu L, Wu H (2019b) Homogeneous selection dominates the microbial community assembly in the sediment of the Three Gorges Reservoir. Sci Total Environ 690:50–60. https://doi.org/10.1016/j.scitotenv.2019.07.014
Li R, Zhu L, Wang Y, Zhu Y-G (2022) Metagenomic insights into environmental risk of field microplastics in an urban river. Water Res 223:119018. https://doi.org/10.1016/j.watres.2022.119018
Li Q, Wang J, Yuan H, Chen B, Zhuang S (2023) Climatic effects on soil organic nitrogen fractions and amino acid chirality in paddy soils. Pedosphere 33(4):579–588. https://doi.org/10.1016/j.pedsph.2022.06.062
Lieke T, Meinelt T, Hoseinifar SH, Pan B, Straus DL, Steinberg CE (2020) Sustainable aquaculture requires environmental-friendly treatment strategies for fish diseases. Rev Aquac 12(2):943–965. https://doi.org/10.1111/raq.12365
Lin J, Wang Y, Lin C, Li R, Wang G (2023) High prevalence of group III-like mutations among BLPACR and first report of Haemophilus influenzae ST95 isolated from blood in China. Infect Drug Resist 999–1008. https://doi.org/10.2147/IDR.S400207
Linhardt R, Galliher P, Cooney C (1987) Polysaccharide lyases. Appl Biochem Biotechnol 12:135–176. https://doi.org/10.1007/BF02798420
Liu L, Shen P, Zheng B, Yu W, Ji J, Xiao Y (2020) Comparative genomic analysis of 19 clinical isolates of tigecycline-resistant Acinetobacter baumannii. Front Microbiol 11:1321. https://doi.org/10.3389/fmicb.2020.01321
Liu X, Shao Z, Cheng G, Lu S, Gu Z, Zhu H, Shen H, Wang J, Chen X (2021) Ecological engineering in pond aquaculture: a review from the whole-process perspective in China. Rev Aquac 13(2):1060–1076. https://doi.org/10.1111/raq.12512
Liu B, Zheng D, Zhou S, Chen L, Yang J (2022a) VFDB 2022: a general classification scheme for bacterial virulence factors. Nucleic Acids Res 50(D1):D912–D917. https://doi.org/10.1093/nar/gkab1107
Liu T, Li C, Tan W, Wang J, Feng J, Hu Q, Cao C (2022b) Rice-crayfish co-culture reduces ammonia volatilization and increases rice nitrogen uptake in central China. Agr Ecosyst Environ 330:107869. https://doi.org/10.1016/j.agee.2022.107869
Lu H, Weng Z, Wei H, Zhou J, Wang J, Liu G, Guo W (2017) Simultaneous bisphenol F degradation, heterotrophic nitrification and aerobic denitrification by a bacterial consortium. J Chem Technol Biotechnol 92(4):854–860. https://doi.org/10.1002/jctb.5069
Lu J, Zhang X, Wang C, Li M, Chen J, Xiong J (2021) Responses of sediment resistome, virulence factors and potential pathogens to decades of antibiotics pollution in a shrimp aquafarm. Sci Total Environ 794:148760. https://doi.org/10.1016/j.scitotenv.2021.148760
Majumdar T, Ghosh S, Pal J, Mazumder S (2006) Possible role of a plasmid in the pathogenesis of a fish disease caused by Aeromonas hydrophila. Aquaculture 256(1):95–104. https://doi.org/10.1016/j.aquaculture.2006.02.042
Martin RM, Bachman MA (2018) Colonization, infection, and the accessory genome of Klebsiella pneumoniae. Front Cell Infect Microbiol 8:4. https://doi.org/10.3389/fcimb.2018.00004
Martínez-Córdova LR, Emerenciano M, Miranda-Baeza A, Martínez-Porchas M (2015) Microbial-based systems for aquaculture of fish and shrimp: an updated review. Rev Aquac 7(2):131–148. https://doi.org/10.1111/raq.12058
Martínez-Lara P, Martínez-Porchas M, Gollas-Galván T, Hernández-López J, Robles-Porchas GR (2021) Granulomatosis in fish aquaculture: a mini review. Rev Aquac 13(1):259–268. https://doi.org/10.1111/raq.12472
Meibom KL, Charbit A (2010) Francisella tularensis metabolism and its relation to virulence. Front Microbiol 1:140. https://doi.org/10.3389/fmicb.2010.00140
Mohan SB, Schmid M, Jetten M, Cole J (2004) Detection and widespread distribution of the nrfA gene encoding nitrite reduction to ammonia, a short circuit in the biological nitrogen cycle that competes with denitrification. FEMS Microbiol Ecol 49(3):433–443. https://doi.org/10.1016/j.femsec.2004.04.012
Molofsky AB, Swanson MS (2004) Differentiate to thrive: lessons from the Legionella pneumophila life cycle. Mol Microbiol 53(1):29–40. https://doi.org/10.1111/j.1365-2958.2004.04129.x
Monguió-Tortajada M, Franquesa M, Sarrias M-R, Borràs FE (2018) Low doses of LPS exacerbate the inflammatory response and trigger death on TLR3-primed human monocytes. Cell Death Dis 9(5):499. https://doi.org/10.1038/s41419-018-0520-2
Müller SI, Valdebenito M, Hantke K (2009) Salmochelin, the long-overlooked catecholate siderophore of Salmonella. Biometals 22:691–695. https://doi.org/10.1007/s10534-009-9217-4
Naylor RL, Hardy RW, Buschmann AH, Bush SR, Cao L, Klinger DH, Little DC, Lubchenco J, Shumway SE, Troell M (2021) A 20-year retrospective review of global aquaculture. Nature 591(7851):551–563. https://doi.org/10.1038/s41586-021-03308-6
Nel HA, Dalu T, Wasserman RJ (2018) Sinks and sources: assessing microplastic abundance in river sediment and deposit feeders in an Austral temperate urban river system. Sci Total Environ 612:950–956. https://doi.org/10.1016/j.scitotenv.2017.08.298
Nimrat S, Suksawat S, Maleeweach P, Vuthiphandchai V (2008) Effect of different shrimp pond bottom soil treatments on the change of physical characteristics and pathogenic bacteria in pond bottom soil. Aquaculture 285(1):123–129. https://doi.org/10.1016/j.aquaculture.2008.08.020
Nisar S, Kirkpatrick LD, Shupp JW (2021) Bacterial virulence factors and their contribution to pathophysiology after thermal injury. Surg Infect 22(1):69–76. https://doi.org/10.1089/sur.2020.188
Niu C, Yu D, Wang Y, Ren H, Jin Y, Zhou W, Li B, Cheng Y, Yue J, Gao Z (2013) Common and pathogen-specific virulence factors are different in function and structure. Virulence 4(6):473–482. https://doi.org/10.4161/viru.25730
Osman KM, Ali MN, Radwan I, ElHofy F, Abed AH, Orabi A, Fawzy NM (2016) Dispersion of the vancomycin resistance genes vanA and vanC of Enterococcus isolated from Nile tilapia on retail sale: a public health hazard. Front Microbiol 7:1354. https://doi.org/10.3389/fmicb.2016.01354
Pan Y, Zeng J, Li L, Yang J, Tang Z, Xiong W, Li Y, Chen S, Zeng Z (2020) Coexistence of antibiotic resistance genes and virulence factors deciphered by large-scale complete genome analysis. Msystems 5(3):e00821-e919. https://doi.org/10.1128/mSystems.00821-19
Pang M, Jiang J, Xie X, Wu Y, Dong Y, Kwok AH, Zhang W, Yao H, Lu C, Leung FC (2015) Novel insights into the pathogenicity of epidemic Aeromonas hydrophila ST251 clones from comparative genomics. Sci Rep 5(1):9833. https://doi.org/10.1038/srep09833
Peeler EJ, Oidtmann BC, Midtlyng PJ, Miossec L, Gozlan RE (2011) Non-native aquatic animals introductions have driven disease emergence in Europe. Biol Invasions 13:1291–1303. https://doi.org/10.1007/s10530-010-9890-9
Pickard JM, Zeng MY, Caruso R, Núñez G (2017) Gut microbiota: role in pathogen colonization, immune responses, and inflammatory disease. Immunol Rev 279(1):70–89. https://doi.org/10.1111/imr.12567
Qin S, Xiao W, Zhou C, Pu Q, Deng X, Lan L, Liang H, Song X, Wu M (2022) Pseudomonas aeruginosa: pathogenesis, virulence factors, antibiotic resistance, interaction with host, technology advances and emerging therapeutics. Signal Transduct Target Ther 7(1):199. https://doi.org/10.1038/s41392-022-01056-1
Ragab W, Kawato S, Nozaki R, Kondo H, Hirono I (2022) Comparative genome analyses of five Vibrio penaeicida strains provide insights into their virulence-related factors. Microb Genom 8(2). https://doi.org/10.1099/mgen.0.000766
Ramirez Maria S, Traglia German M, Lin David L, Tran T, Tolmasky Marcelo E (2014) Plasmid-mediated antibiotic resistance and virulence in gram-negatives: the Klebsiella pneumoniae paradigm. Microbiol Spectr 2(5):https://doi.org/10.1128/microbiolspec.plas-0016-2013
Ramsay JP, Firth N (2017) Diverse mobilization strategies facilitate transfer of non-conjugative mobile genetic elements. Curr Opin Microbiol 38:1–9. https://doi.org/10.1016/j.mib.2017.03.003
Ratledge C, Dover LG (2000) Iron metabolism in pathogenic bacteria. Ann Rev Microbiol 54(1):881–941. https://doi.org/10.1146/annurev.micro.54.1.881
Riedel K, Grunau A (2011) Exoenzymes. In: Reitner J, Thiel V (eds) Encyclopedia of geobiology. Springer, Netherlands, pp 355–359
Rohmer L, Hocquet D, Miller SI (2011) Are pathogenic bacteria just looking for food? Metabolism and microbial pathogenesis. Trends Microbiol 19(7):341–348. https://doi.org/10.1016/j.tim.2011.04.003
Sarkar P, Issac PK, Raju SV, Elumalai P, Arshad A, Arockiaraj J (2021) Pathogenic bacterial toxins and virulence influences in cultivable fish. Aquac Res 52(6):2361–2376. https://doi.org/10.1111/are.15089
Sarkar P, Raju VS, Kuppusamy G, Rahman MA, Elumalai P, Harikrishnan R, Arshad A, Arockiaraj J (2022) Pathogenic fungi affecting fishes through their virulence molecules. Aquaculture 548:737553. https://doi.org/10.1016/j.aquaculture.2021.737553
Schar D, Klein EY, Laxminarayan R, Gilbert M, Van Boeckel TP (2020) Global trends in antimicrobial use in aquaculture. Sci Rep 10(1):21878. https://doi.org/10.1038/s41598-020-78849-3
Schniederberend M, Williams JF, Shine E, Shen C, Jain R, Emonet T, Kazmierczak BI (2019) Modulation of flagellar rotation in surface-attached bacteria: a pathway for rapid surface-sensing after flagellar attachment. PLoS Pathog 15(11):e1008149. https://doi.org/10.1371/journal.ppat.1008149
Schroeder GN, Petty NK, Mousnier A, Harding CR, Vogrin AJ, Wee B, Fry NK, Harrison TG, Newton HJ, Thomson NR (2010) Legionella pneumophila strain 130b possesses a unique combination of type IV secretion systems and novel Dot/Icm secretion system effector proteins. J Bacteriol 192(22):6001–6016. https://doi.org/10.1128/JB.00778-10
Shahbaaz M, ImtaiyazHassan M, Ahmad F (2013) Functional annotation of conserved hypothetical proteins from Haemophilus influenzae Rd KW20. PLoS One 8(12):e84263. https://doi.org/10.1371/journal.pone.0084263
Sharma AK, Dhasmana N, Dubey N, Kumar N, Gangwal A, Gupta M, Singh Y (2017) Bacterial virulence factors: secreted for survival. Indian J Microbiol 57:1–10. https://doi.org/10.1007/s12088-016-0625-1
Siguier P, Filée J, Chandler M (2006a) Insertion sequences in prokaryotic genomes. Curr Opin Microbiol 9(5):526–531. https://doi.org/10.1016/j.mib.2006.08.005
Siguier P, Perochon J, Lestrade L, Mahillon J, Chandler M (2006) ISfinder: the reference centre for bacterial insertion sequences. Nucleic Acids Res 34(Database issue):D32-6. https://doi.org/10.1093/nar/gkj014
Song H-C, Yang B-T, Zhao T, Sun Y-F, Zhou J-H, Shan X-F, Qian A-D, Sun W-C, Kang Y-H (2021) Comparative genomics analysis of strains from diverse sources reveals the evolutionary relationship of Aeromonas veronii. Microb Pathog 159:105134. https://doi.org/10.1016/j.micpath.2021.105134
Sonika S, Singh S, Mishra S, Verma S (2023) Toxin-antitoxin systems in bacterial pathogenesis. Heliyon 9(4):e14220. https://doi.org/10.1016/j.heliyon.2023.e14220
Steinert M, Hentschel U, Hacker J (2002) Legionella pneumophila: an aquatic microbe goes astray. FEMS Microbiol Rev 26(2):149–162. https://doi.org/10.1111/j.1574-6976.2002.tb00607.x
Stephens RS, Kalman S, Lammel C, Fan J, Marathe R, Aravind L, Mitchell W, Olinger L, Tatusov RL, Zhao Q (1998) Genome sequence of an obligate intracellular pathogen of humans: Chlamydia trachomatis. Science 282(5389):754–759. https://doi.org/10.1126/science.282.5389.75
Su H, Hu X, Xu W, Xu Y, Wen G, Cao Y (2022) Diversity, abundances and distribution of antibiotic resistance genes and virulence factors in the South China Sea revealed by metagenomic sequencing. Sci Total Environ 814:152803. https://doi.org/10.1016/j.scitotenv.2021.152803
Sun X, Wang H, Wang F, Zhao Y, Wang H, Zhu J, Wei S, Chen H (2023) Effects of different fertilization patterns on the dietary composition of Procambarus clarkii in a rice-crayfish coculture system. Aquac Rep 33:101801. https://doi.org/10.1016/j.aqrep.2023.101801
van de Bund WJ, Goedkoop W, Johnson RK (1994) Effects of deposit-feeder activity on bacterial production and abundance in profundal lake sediment. J N Am Benthol Soc 13(4):532–539. https://doi.org/10.2307/1467849
Wardman JF, Bains RK, Rahfeld P, Withers SG (2022) Carbohydrate-active enzymes (CAZymes) in the gut microbiome. Nat Rev Microbiol 20(9):542–556. https://doi.org/10.1038/s41579-022-00712-1
Woolhouse ME, Taylor LH, Haydon DT (2001) Population biology of multihost pathogens. Science 292(5519):1109–1112. https://doi.org/10.1126/science.1059026
Xu J, Wang J, Lin S, Hou L, Ma S, Lv Y, Chen R, He X, Hou J (2023) The effect of novel aquaculture mode on phosphorus sorption-release in pond sediment. Sci Total Environ 905:167019. https://doi.org/10.1016/j.scitotenv.2023.167019
Yan L, Pei X, Zhang X, Guan W, Chui H, Jia H, Ma G, Yang S, Li Y, Li N (2019) Occurrence of four pathogenic Vibrios in Chinese freshwater fish farms in 2016. Food Control 95:85–89. https://doi.org/10.1016/j.foodcont.2018.07.043
Yue GH, Tay YX, Wong J, Shen Y, Xia J (2023) Aquaculture species diversification in China. Aquac Fish. https://doi.org/10.1016/j.aaf.2022.12.001
Zadeh Hosseingholi E, Zarrini G, Pashazadeh M, Gheibi Hayat SM, Molavi G (2020) In silico identification of probable drug and vaccine candidates against antibiotic-resistant Acinetobacter baumannii. Microb Drug Resist 26(5):456–467. https://doi.org/10.1089/mdr.2019.0236
Ze X, Le Mougen F, Duncan SH, Louis P, Flint HJ (2013) Some are more equal than others: the role of “keystone” species in the degradation of recalcitrant substrates. Gut Microbes 4(3):236–240. https://doi.org/10.1038/ismej.2012.4
Zhang C, Mi W, Xu Y, Zhou W, Bi Y (2023a) Long-term integrated rice-crayfish culture disrupts the microbial communities in paddy soil. Aquac Rep 29:101515. https://doi.org/10.1016/j.aqrep.2023.101515
Zhang X, Ren Y, Zhu X, Pan H, Yao H, Wang J, Liu M, He M (2023b) Driving factors for black-odor-related microorganisms and potential self-remediation strategies. Sustainability 15(1):521. https://doi.org/10.3390/su15010521
Zhou R, Zeng S, Hou D, Liu J, Weng S, He J, Huang Z (2019) Occurrence of human pathogenic bacteria carrying antibiotic resistance genes revealed by metagenomic approach: a case study from an aquatic environment. J Environ Sci 80:248–256. https://doi.org/10.1016/j.jes.2019.01.001
Zhou A, Xie S, Tang H, Zhang L, Zhang Y, Zuo Z, Li X, Zhao W, Xu G, Zou J (2022) The dynamic of the potential pathogenic bacteria, antibiotic-resistant bacteria, and antibiotic resistance genes in the water at different growth stages of grass carp pond. Environ Sci Pollut Res 29(16):23806–23822. https://doi.org/10.1007/s11356-021-17578-0
Zhu J, Wang T, Chen L, Du H (2021) Virulence factors in hypervirulent Klebsiella pneumoniae. Front Microbiol 12. https://doi.org/10.3389/fmicb.2021.642484
Zipfel C, Kunze G, Chinchilla D, Caniard A, Jones JD, Boller T, Felix G (2006) Perception of the bacterial PAMP EF-Tu by the receptor EFR restricts Agrobacterium-mediated transformation. Cell 125(4):749–760. https://doi.org/10.1016/j.cell.2006.03.037
Funding
This work is supported by the National Natural Science Foundation of China (Grant Number 41902257), the Hubei Key Laboratory of Intelligent Yangtze and Hydroelectric Science (No. ZH2102000113), and the Natural Science Foundation of Hubei Province of China (No. 2020CFB750).
Author information
Authors and Affiliations
Contributions
HP and XZ designed research; XZ and XZ performed research; WX, JW, and HX analyzed data; WX wrote the manuscript. XZ revised and reviewed the drafts.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the author.
Consent to participate
All participants gave informed consent at enrollment.
Consent for publication
All participants gave final approval for publication.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Diane Purchase
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xi, W., Zhang, X., Zhu, X. et al. Distribution patterns and influential factors of pathogenic bacteria in freshwater aquaculture sediments. Environ Sci Pollut Res 31, 16028–16047 (2024). https://doi.org/10.1007/s11356-024-31897-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-31897-y