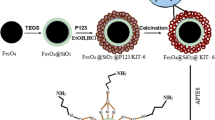
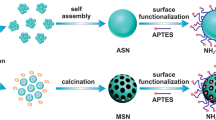
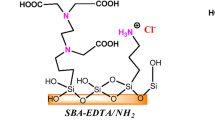
Abstract
Presence of heavy metals in wastewater is a critical environmental issue, and efficient extraction of the metals remains a challenging task. In this study, the adsorption behavior of Ce(III), Hg(II), and Cu(II) metal ions using MCM-48 material modified with acid and base functional groups was examined. The modified materials were characterized using various techniques, including XRD, BET, FT-IR, NMR, and SEM, which revealed that the materials’ properties remained unchanged after modification. The adsorption capacity of the modified materials for metal ions was then evaluated and was found that the amine-modified MCM-48 material exhibited the highest adsorption efficiency. Precisely, the amine-modified material achieved an adsorption capacity of 97% for Ce(III), 98% for Hg(II), and 90% for Cu(II) after 180 min of adsorption. These results highlight the effectiveness of amine functionalization in enhancing the adsorption capacity of silica material for heavy metals.













Similar content being viewed by others
Data availability
Due to ethical reason data were not provided in the manuscript but will be available on request.
References
Abbott AP, Harris RC, Holyoak F, Frisch G, Hartley J, Jenkin GR (2015) Electrocatalytic recovery of elements from complex mixtures using deep eutectic solvents. Green Chem 17:2172–2179. https://doi.org/10.1039/C4GC02246G
Adebayo MA, Jabar JM, Amoko JS, Openiyi EO, Shodiya OO (2022) Coconut husk-raw clay-Fe composite: preparation, characteristics and mechanisms of Congo red adsorption. Scient Rep 12:14370. https://doi.org/10.1038/s41598-022-18763-y
Aguado J, Arsuaga JM, Arencibia A, Lindo M, Gascón V (2009) Aqueous heavy metals removal by adsorption on amine-functionalized mesoporous silica. J Hazard Mater 163:213–221. https://doi.org/10.1016/j.jhazmat.2008.06.080
Ali H, Khan E, Ilahi I (2019) Environmental chemistry and ecotoxicology of hazardous heavy metals: environmental persistence, toxicity, and bioaccumulation. J Chem. https://doi.org/10.1155/2019/6730305
Anbia M, Kargosha K, Khoshbooei S (2015) Heavy metal ions removal from aqueous media by modified magnetic mesoporous silica MCM-48. Chem Eng Res Des 93:779–788. https://doi.org/10.1016/j.cherd.2014.07.018
Awual MR, Yaita T, Shiwaku H (2013a) Design a novel optical adsorbent for simultaneous ultra-trace cerium (III) detection, sorption and recovery. Chem Eng J 228:327–335. https://doi.org/10.1016/j.cej.2013.05.010
Awual MR, Kobayashi T, Miyazaki Y, Motokawa R, Shiwaku H, Suzuki S, Okamoto Y, Yaita T (2013b) Selective lanthanide sorption and mechanism using novel hybrid Lewis base (N-methyl-N-phenyl-1, 10-phenanthroline-2-carboxamide) ligand modified adsorbent. J Hazard Mater 252:313–320. https://doi.org/10.1016/j.jhazmat.2013.03.020
Bandyopadhyay M, Shiju NR, Brown DR (2010) MCM-48 as a support for sulfonic acid catalysts. Catal Commun 11:660–664. https://doi.org/10.1016/j.catcom.2010.01.018
Bandyopadhyay M, Tsunoji N, Sano T (2017) Nano-porous MCM-48 immobilized with aminopropyltriethoxysilane: a potential catalyst for transesterification of triacetin. Catal Lett 147:1040–1050. https://doi.org/10.1007/s10562-017-1997-5
Bandyopadhyay M, Tsunoji N, Bandyopadhyay R, Sano T (2019) Comparison of sulfonic acid loaded mesoporous silica in transesterification of triacetin. React Kinet Mech Catal 126:167–179. https://doi.org/10.1007/s11144-018-1447-4
Barik B, Kumar A, Nayak PS, Achary LS, Rout L, Dash P (2020) Ionic liquid assisted mesoporous silica-graphene oxide nanocomposite synthesis and its application for removal of heavy metal ions from water. Mater Chem Phys 239:122028. https://doi.org/10.1016/j.matchemphys.2019.122028
Chien SW, Ng DQ, Kumar D, Lam SM, Jaffari ZH (2022) Investigating the effects of various synthesis routes on morphological, optical, photoelectrochemical and photocatalytic properties of single-phase perovskite BiFeO3. J Phys Chem Solids 160:110342. https://doi.org/10.1016/j.jpcs.2021.110342
Da’na E, Sayari A (2012) Adsorption of heavy metals on amine-functionalized SBA-15 prepared by co-condensation: applications to real water samples. Desalination 285:62–67. https://doi.org/10.1016/j.desal.2011.09.034
Deng Y, Wei J, Sun Z, Zhao D (2013) Large-pore ordered mesoporous materials templated from non-pluronic amphiphilic block copolymers. Chem Soc Rev 42:4054–4070. https://doi.org/10.1039/C2CS35426H
Du Z, Deng S, Bei Y, Huang Q, Wang B, Huang J, Yu G (2014) Adsorption behavior and mechanism of perfluorinated compounds on various adsorbents—a review. J Hazard Mater 274:443–454. https://doi.org/10.1016/j.jhazmat.2014.04.038
Duan L, Wang C, Zhang W, Ma B, Deng Y, Li W, Zhao D (2021) Interfacial assembly and applications of functional mesoporous materials. Chem Rev 121:14349–14429. https://doi.org/10.1021/acs.chemrev.1c00236
Dubey SS, Rao BS, Andrews BS, Kiran BV (2011) Efficient removal of Ce (III) and Eu (III) Ions from aqueous solutions by local clay-a radiotracer study. E J Chem 8:917–923
Feini LI, Zhang G, Qin M, Zhang H (2008) Performance of nanofiltration and reverse osmosis membranes in metal effluent treatment. Chin J Chem Eng 16:441–445. https://doi.org/10.1016/S1004-9541(08)60102-0
Gautam RK, Sharma SK, Mahiya S, Chattopadhyaya MC (2014) Contamination of heavy metals in aquatic media: transport, toxicity and technologies for remediation. https://doi.org/10.1039/9781782620174-00001
Gérardin C, Reboul J, Bonne M, Lebeau B (2013) Ecodesign of ordered mesoporous silica materials. Chem Soc Rev 42:4217–4255. https://doi.org/10.1039/C3CS35451B
Ghaedi M, Shokrollahi A, Kianfar AH, Pourfarokhi A, Khanjari N, Mirsadeghi AS, Soylak M (2009) Preconcentration and separation of trace amount of heavy metal ions on bis (2-hydroxy acetophenone) ethylendiimine loaded on activated carbon. J Hazard Mater 162:1408–1414. https://doi.org/10.1016/j.jhazmat.2008.06.057
Gharabaghi M, Irannajad M, Azadmehr AR (2012) Selective sulphide precipitation of heavy metals from acidic polymetallic aqueous solution by thioacetamide. Indust Eng Chem Res 51:954–9563. https://doi.org/10.1016/j.desal.2011.08.032
Grün M, Kurganov AA, Schacht S, Schüth F, Unger KK (1996) Comparison of an ordered mesoporous aluminosilicate, silica, alumina, titania and zirconia in normal-phase high-performance liquid chromatography. J Chromatography a 740:1–9. https://doi.org/10.1016/0021-9673(96)00205-1
Hartmann M (2005) Ordered mesoporous materials for bioadsorption and biocatalysis. Chem Mater 17:4577–4593. https://doi.org/10.1021/cm0485658
He C, Ren L, Zhu W, Xu Y, Qian X (2015) Removal of mercury from aqueous solution using mesoporous silica nanoparticles modified with polyamide receptor. J Colloid Interface Sci 458:229–234. https://doi.org/10.1016/j.jcis.2015.07.054
Heidari A, Younesi H, Mehraban Z (2009) Removal of Ni (II), Cd (II), and Pb (II) from a ternary aqueous solution by amino functionalized mesoporous and nano mesoporous silica. Chem Eng J 153:70–79. https://doi.org/10.1016/j.cej.2009.06.016
Hou S, Li X, Wang H, Wang M, Zhang Y, Chi Y, Zhao Z (2017) Synthesis of core–shell structured magnetic mesoporous silica microspheres with accessible carboxyl functionalized surfaces and radially oriented large mesopores as adsorbents for the removal of heavy metal ions. RSC Adv 7:51993–52000. https://doi.org/10.1039/C7RA08937F
Hua M, Zhang S, Pan B, Zhang W, Lv L, Zhang Q (2012) Heavy metal removal from water/wastewater by nanosized metal oxides: a review. J Hazard Mater 211:317–331. https://doi.org/10.1016/j.jhazmat.2011.10.016
Jabar JM, Odusote YA (2020) Removal of cibacron blue 3G-A (CB) dye from aqueous solution using chemo-physically activated biochar from oil palm empty fruit bunch fiber. Arab J Chem 13:5417–5429. https://doi.org/10.1016/j.arabjc.2020.03.020
Jabar JM, Owokotomo IA, Ayinde YT, Alafabusuyi AM, Olagunju GO, Mobolaji VO (2021) Characterization of prepared eco-friendly biochar from almond (Terminalia catappa L) leaf for sequestration of bromophenol blue (BPB) from aqueous solution. Carbon Lett 31:1001–1014. https://doi.org/10.1007/s42823-020-00214-1
Jabar JM, Adebayo MA, Owokotomo IA, Odusote YA, Yılmaz M (2022) Synthesis of high surface area mesoporous ZnCl2–activated cocoa (Theobroma cacao L) leaves biochar derived via pyrolysis for crystal violet dye removal. Heliyon 8:e10873. https://doi.org/10.1016/j.heliyon.2022.e10873
Jadav D, Shukla P, Bandyopadhyay R, Kubota Y, Das S, Bandyopadhyay M (2020) Tetranuclear Zn complex covalently immobilized on sulfopropylsilylated nano-porous silica: an efficient catalyst for ring opening reaction of epoxide with amine. Mol Catal 497:111220. https://doi.org/10.1016/j.mcat.2020.111220
Jadav D, Shukla P, Bandyopadhyay R, Sarma PJ, Deka RC, Kumar R, Das S, Tsunoji N, Bandyopadhyay M (2022) Immobilization of a Zn4 complex on functionalized layered HUS-7: synthesis, structural investigation and catalytic activity. New J Chem 46:9418–9431. https://doi.org/10.1039/D2NJ00669C
Jaffari ZH, Lam SM, Sin JC, Zeng H, Mohamed AR (2020) Magnetically recoverable Pd-loaded BiFeO3 microcomposite with enhanced visible light photocatalytic performance for pollutant, bacterial and fungal elimination. Sep Purif Techn 236:116195. https://doi.org/10.1016/j.seppur.2019.116195
Jaffari ZH, Abbas A, Umer M, Kim ES, Cho KH (2023) Crystal graph convolution neural networks for fast and accurate prediction of adsorption ability of Nb2CT x towards Pb (ii) and Cd (ii) ions. J Mater Chem a 11:9009–9018. https://doi.org/10.1039/D3TA00019B
Jaffari ZH, Jeong H, Shin J, Kwak J, Son C, Lee YG, Kim S, Chon K, Cho KH (2023) Machine-learning-based prediction and optimization of emerging contaminants’ adsorption capacity on biochar materials. Chem Eng J 466:143073. https://doi.org/10.1016/j.cej.2023.143073
Järup L (2003) Hazards of heavy metal contamination. Br Med Bull 68:167–182. https://doi.org/10.1093/bmb/ldg032
Jia W, Xu H, Yang Q, Ren S, Wang J (2019) Synthesis of anionic Gemini surfactant-templated mesoporous silica nanoparticles and its adsorption application for Pb2+. J Dispers Sci Techno 11:1664–1674. https://doi.org/10.1080/01932691.2018.1535979
Kailasa SK, Chandel M, Mehta VN, Park TJ (2018) Influence of ligand chemistry on silver nanoparticles for colorimetric detection of Cr3+ and Hg2+ ions. Spectrochim Acta A: Mol Biomol Spectrosc 195:120–127. https://doi.org/10.1016/j.saa.2018.01.038
Karacan MS, Aslantaş N (2008) Simultaneous preconcentration and removal of iron, chromium, nickel with N, N′-etylenebis-(ethane sulfonamide) ligand on activated carbon in aqueous solution and determination by ICP-OES. J Hazard Mater 155(3):551–557. https://doi.org/10.1016/j.jhazmat.2007.11.107
Khan AA, Paquiza L (2011) Characterization and ion-exchange behavior of thermally stable nano-composite polyaniline zirconium titanium phosphate: its analytical application in separation of toxic metals. Desalination 265:242–254. https://doi.org/10.1016/j.desal.2010.07.058
Kraus U, Wiegand J (2006) Long-term effects of the Aznalcóllar mine spill—heavy metal content and mobility in soils and sediments of the Guadiamar river valley (SW Spain). Sci Total Environ 367:855–871. https://doi.org/10.1016/j.scitotenv.2005.12.027
Lai CY, Trewyn BG, Jeftinija DM, Jeftinija K, Xu S, Jeftinija S, Lin VS (2003) A mesoporous silica nanosphere-based carrier system with chemically removable CdS nanoparticle caps for stimuli-responsive controlled release of neurotransmitters and drug molecules. J Am Chem Soc 125:4451–4459. https://doi.org/10.1021/ja028650l
Lam SM, Jaffari ZH, Sin JC, Zeng H, Lin H, Li H, Mohamed AR, Ng DQ (2021) Surface decorated coral-like magnetic BiFeO3 with Au nanoparticles for effective sunlight photodegradation of 2, 4-D and E. coli inactivation. J Mol Liq 326:115372. https://doi.org/10.1016/j.molliq.2021.115372
Li G, Zhao Z, Liu J, Jiang G (2011) Effective heavy metal removal from aqueous systems by thiol functionalized magnetic mesoporous silica. J Hazard Mater 192:277–283. https://doi.org/10.1016/j.jhazmat.2011.05.015
Mallik AK, Moktadir MA, Rahman MA, Shahruzzaman M, Rahman MM (2022) Progress in surface-modified silicas for Cr (VI) adsorption: a review. J Hazard Mater 423:127041. https://doi.org/10.1016/j.jhazmat.2021.127041
Marjani A, Shirazian S (2011) Simulation of heavy metal extraction in membrane contactors using computational fluid dynamics. Desalination 281:422–428. https://doi.org/10.1016/j.desal.2011.08.032
Mousavi SJ, Parvini M, Ghorbani M (2018) Adsorption of heavy metals (Cu2+ and Zn2+) on novel bifunctional ordered mesoporous silica: optimization by response surface methodology. J Taiwan Inst Chem Eng 84:123–141. https://doi.org/10.1016/j.jtice.2018.01.010
Mureseanu M, Reiss A, Stefanescu I, David E, Parvulescu V, Renard G, Hulea V (2008) Modified SBA-15 mesoporous silica for heavy metal ions remediation. Chemosphere 73:1499–1504. https://doi.org/10.1016/j.chemosphere.2008.07.039
Murthy ZV, Choudhary A (2011) Separation of cerium from feed solution by nanofiltration. Desalination 279:428–432. https://doi.org/10.1016/j.desal.2011.06.014
Naidu G, Ryu S, Thiruvenkatachari R, Choi Y, Jeong S, Vigneswaran S (2019) A critical review on remediation, reuse, and resource recovery from acid mine drainage. Environm Pollut 247:1110–1124. https://doi.org/10.1016/j.envpol.2019.01.085
Orilall MC, Wiesner U (2011) Block copolymer based composition and morphology control in nanostructured hybrid materials for energy conversion and storage: solar cells, batteries, and fuel cells. Chem Soc Rev 40:520–535. https://doi.org/10.1039/C0CS00034E
Pan B, Pan B, Zhang W, Lv L, Zhang Q, Zheng S (2009) Development of polymeric and polymer-based hybrid adsorbents for pollutants removal from waters. Chem Eng J 151:19–29. https://doi.org/10.1016/j.cej.2009.02.036
Pandey M, Jadav D, Manhas A, Kediya S, Tsunoji N, Kumar R, Das S, Bandyopadhyay M (2022) Synthesis and characterization of mononuclear Zn complex, immobilized on ordered nano-porous silica and their tunable catalytic properties. Mol Catal 525:112365. https://doi.org/10.1016/j.mcat.2022.112365
Pandey M, Prajapati R, Shukla P, Paredi P, Tsunoji N, Kumar R, Shahabuddin S, Das S, Bandyopadhyay M (2022) Synthesis of novel tetranuclear Ni complex incorporated nano-porous silica for improved photocatalytic degradation of methylene blue in presence of visible light. Polyhedron 228:116161. https://doi.org/10.1016/j.poly.2022.116161
Pandey M, Tsunoji N, Kubota Y, Bandyopadhyay M (2022c) Amine and sulfonic acid anchored porous silica as recyclable heterogeneous catalysts for ring-opening of oxiranes. ChemistrySelect 7:e202201756. https://doi.org/10.1002/slct.202201756
Pandey M, Shukla P, Manhas A, Kediya S, Tsunoji N, Kumar R, Harmalkar SS, Das S, Bandyopadhyay M (2023) Synthesis and characterization of novel binuclear zinc complex, immobilization in nano-porous support, and its catalytic application. Mol Catal 537:112964. https://doi.org/10.1016/j.mcat.2023.112964
Panthi G, Park M (2022) Synthesis of metal nanoclusters and their application in Hg2+ ions detection: a review. J Hazard Mater 424:127565. https://doi.org/10.1016/j.jhazmat.2021.127565
Puangsamlee T, Tachapermpon Y, Kammalun P, Sukrat K, Wainiphithapong C, Sirirak J, Wanichacheva N (2018) Solvent control bifunctional fluorescence probe for selective detection of Cu2+ and Hg2+ via the excimer of pyrenylacetamide subunits. J Luminescence 196:227–235. https://doi.org/10.1016/j.jlumin.2017.11.048
Shahbazi A, Younesi H, Badiei A (2011) Functionalized SBA-15 mesoporous silica by melamine-based dendrimer amines for adsorptive characteristics of Pb (II), Cu (II) and Cd (II) heavy metal ions in batch and fixed bed column. Chem Eng J 168(2):505–518. https://doi.org/10.1016/j.cej.2010.11.053
Shan R, Shi Y, Gu J, Wang Y, Yuan H (2022) Single and competitive adsorption affinity of heavy metals toward peanut shell-derived biochar and its mechanisms in aqueous systems. Chin J Chem Eng 28:1375–1383. https://doi.org/10.1016/j.cjche.2020.02.012
Singh S, Ramakrishna S, Gupta MK (2017) Towards zero waste manufacturing: a multidisciplinary review. J Clean Product 168:1230–1243. https://doi.org/10.1016/j.jclepro.2017.09.108
Taguchi A, Schüth F (2005) Ordered mesoporous materials in catalysis. Microporous Mesoporous Mater 77:1–45. https://doi.org/10.1016/j.micromeso.2004.06.030
Vojoudi H, Badiei A, Bahar S, Ziarani GM, Faridbod F, Ganjali MR (2017) A new nano-sorbent for fast and efficient removal of heavy metals from aqueous solutions based on modification of magnetic mesoporous silica nanospheres. J Magn Magn Mater 441:193–203. https://doi.org/10.1016/j.jmmm.2017.05.065
Wang X, Pei Y, Lu M, Lu X, Du X (2015) Highly efficient adsorption of heavy metals from wastewaters by graphene oxide-ordered mesoporous silica materials. J Mater Sci 50:2113–2121. https://doi.org/10.1007/s10853-014-8773-3
Xue Y, Wang Y (2022) Green electrochemical redox mediation for valuable metal extraction and recycling from industrial waste. Green Chem 22:6288–6309. https://doi.org/10.1039/D0GC02028A
Yang W, Ding P, Zhou L, Yu J, Chen X, Jiao F (2013) Preparation of diamine modified mesoporous silica on multi-walled carbon nanotubes for the adsorption of heavy metals in aqueous solution. Appl Surf Sci 282:38–45. https://doi.org/10.1016/j.apsusc.2013.05.028
Yokoi T, Yoshitake H, Tatsumi T (2004) Synthesis of amino-functionalized MCM-41 via direct co-condensation and post-synthesis grafting methods using mono-di-and tri-amino-organoalkoxysilanes. J Chem Mater 14:951–957. https://doi.org/10.1039/B310576H
Yong ZJ, Lam SM, Sin JC, Zeng H, Mohamed AR, Jaffari ZH (2022) Boosting sunlight-powered photocatalytic fuel cell with S-scheme Bi2WO6/ZnO nanorod array composite photoanode. Inorg Chem Commun 143:109826. https://doi.org/10.1016/j.inoche.2022.109826
Yuvaraj P, Ajantha J, Karuppusamy M, Easwaramoorthi S, Rao JR (2021) Hydrogen bond-assisted colorimetric picomolar level detection of Hg2+ ions in 100% aqueous solution. ACS Sustain Chem Eng 9:10309–10317. https://doi.org/10.1021/acssuschemeng.1c03127
Zeb S, Ali N, Ali Z, Bilal M, Adalat B, Hussain S, Gul S, Ali F, Ahmad R, Iqbal HM (2020) Silica-based nanomaterials as designer adsorbents to mitigate emerging organic contaminants from water matrices. J Water Process Eng 38:101675. https://doi.org/10.1016/j.jwpe.2020.101675
Zhang L, Yang S, Han T, Zhong L, Ma C, Zhou Y, Han X (2012) Improvement of Ag (I) adsorption onto chitosan/triethanolamine composite sorbent by an ion-imprinted technology. Appl Surf Sci 263:696–703. https://doi.org/10.1016/j.apsusc.2012.09.143
Acknowledgement
Madhu Pandey acknowledged SHODH (Scheme of Developing High quality research) for financial support.
Funding
Madhu Pandey received financial support from SHODH (Scheme of Developing High quality research).
Author information
Authors and Affiliations
Contributions
Madhu Pandey:,methodology, writing-original draft preparation; Syed Shahabuddin: adsorption supervision, reviewing and editing; Nao Tsunoji: characterization; Sourav Das: reviewing, Editing; Mahuya Bandyopadhyay: conceptualization, reviewing, editing, supervision, execution.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
This article does not contain any study with human and animals performed by any of the author. Consent to participate is not applicable.
Consent for publication
The authors have consented to the submission of the study to the journal.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pandey, M., Shabuddhin, S., Tsunoji, N. et al. Extraction of heavy metals from wastewater using amine-modified mesoporous silica. Environ Sci Pollut Res 30, 113409–113423 (2023). https://doi.org/10.1007/s11356-023-30092-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-30092-9