Abstract
Titanium nanoparticles and pyrethroid pesticides are now being widely used in industrial, agriculture, and biomedical applications. In recent years, their health safety profiles have aroused concerns among health scientists. This study mainly explored the combined effects of titanium dioxide nanoparticles (TiO2NPs) and cypermethrin (CYP) on reproductive toxicity in male rats by gavage for 90 days. Thirty-two male Sprague–Dawley rats were assigned to four groups: the control group, the TiO2NPs group, the CYP group, and the combined titanium dioxide nanoparticles with cypermethrin (TiO2NPs + CYP) group. The results of biochemical analysis on testicular tissue homogenate showed that TiO2NPs and CYP mixtures decreased the activities of glutathione peroxidase (GSH-Px) and catalase (CAT) while increasing the activity of malondialdehyde (MDA) and lactate dehydrogenase (LDH). Meanwhile, the results of two-way factorial analysis of variance (ANOVA) showed a significant effect on GSH-Px, CAT, LDH, testicular cell apoptosis, and sperm quality in rats after exposure. Furthermore, the combined exposure group exhibited apoptosis of testicular cells and DNA damage. The results indicated that exposure to a mixture of TiO2NPs and CYP had adverse effects on the reproductive status of male rats.








Similar content being viewed by others
Data availability
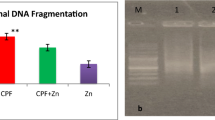
The datasets generated and/or analyzed during the current study are not publicly available due to that the data is original data, but are available from the corresponding author on reasonable request. The DNA was not extracted and analyzed for the study. This study only examined whether titanium dioxide nanoparticles and cypermethrin caused DNA damage in the rats.
References
Abdel-Razik RK, Mosallam EM, Hamed NA, Badawy MEI, Abo-El-Saad MM (2021) Testicular deficiency associated with exposure to cypermethrin, imidacloprid, and chlorpyrifos in adult rats. Environ Toxicol Pharmacol 87:103724. https://doi.org/10.1016/j.etap.2021.103724
Afolabi OK, Aderibigbe FA, Folarin DT, Arinola A, Wusu AD (2019) Oxidative stress and inflammation following sub-lethal oral exposure of cypermethrin in rats: mitigating potential of epicatechin. Heliyon 5(8):e02274. https://doi.org/10.1016/j.heliyon.2019.e02274
Ansari AS, Hussain M, Khan SR, Badar A, Khilwani B, Lohiya NK (2022) Studies on biochemical, oxidative and genotoxicity alterations following vas blockage with reversible inhibition of sperm under guidance and reversal in rats. Indian J Pharmacol 54(1):33–40. https://doi.org/10.4103/ijp.IJP_557_20
Bahadar H, Maqbool F, Niaz K, Abdollahi M (2016) Toxicity of nanoparticles and an overview of current experimental models. Iran Biomed J 20(1):1–11. https://doi.org/10.7508/ibj.2016.01.001
Brohi RD, Wang L, Talpur HS, Wu D, Khan FA, Bhattarai D, Rehman ZU et al (2017) Toxicity of nanoparticles on the reproductive system in animal models: a review. Front Pharmacol 8:606. https://doi.org/10.3389/fphar.2017.00606
Chen Z, Han S, Zheng P, Zhou S, Jia G (2020) Combined effect of titanium dioxide nanoparticles and glucose on the blood glucose homeostasis in young rats after oral administration. J Appl Toxicol 40(9):1284–1296. https://doi.org/10.1002/jat.3985
Chen Z, Shi, J, Zhang, Y, Han S, Zhang J, & Jia, G. (2022). DNA oxidative damage as a sensitive genetic endpoint to detect the genotoxicity induced by titanium dioxide nanoparticles. Nanomaterials (Basel) 12(15). https://doi.org/10.3390/nano12152616
Chrustek A, Hołyńska-Iwan I, Dziembowska I, Bogusiewicz J, Wróblewski M, Cwynar A, & Olszewska-Słonina D (2018). Current research on the safety of pyrethroids used as insecticides. Medicina (Kaunas) 54(4). https://doi.org/10.3390/medicina54040061
de Jager TL, Cockrell AE, Du Plessis SS (2017) Ultraviolet light induced generation of reactive oxygen species. Adv Exp Med Biol 996:15–23. https://doi.org/10.1007/978-3-319-56017-5_2
Dréno B, Alexis A, Chuberre B, Marinovich M (2019) Safety of titanium dioxide nanoparticles in cosmetics. J Eur Acad Dermatol Venereol 33(Suppl 7):34–46. https://doi.org/10.1111/jdv.15943
Fang Q, Shi Q, Guo Y, Hua J, Wang X, Zhou B (2016) Enhanced bioconcentration of bisphenol A in the presence of Nano-TiO2 can lead to adverse reproductive outcomes in zebrafish. Environ Sci Technol 50(2):1005–1013. https://doi.org/10.1021/acs.est.5b05024
Hardy A, Benford D, Halldorsson T, Jeger MJ, Knutsen HK, More S, Naegeli H et al (2018) Guidance on risk assessment of the application of nanoscience and nanotechnologies in the food and feed chain: Part 1, human and animal health. Efsa J 16(7):e05327. https://doi.org/10.2903/j.efsa.2018.5327
Hong F, Zhou Y (2020) Spermatogenic apoptosis and the involvement of the Nrf2 pathway in male mice following exposure to nano titanium dioxide. J Biomed Nanotechnol 16(3):373–381. https://doi.org/10.1166/jbn.2020.2895
Hua N, & Lin Y (2020). New technology in pesticide field-nanopesticide preparation. Chinese Pesticide (3), 19–35. (in Chinese)
Hussein MMA, Gad E, Ahmed MM, Arisha AH, Mahdy HF, Swelum AA, Tukur HA et al (2019) Amelioration of titanium dioxide nanoparticle reprotoxicity by the antioxidants morin and rutin. Environ Sci Pollut Res Int 26(28):29074–29084. https://doi.org/10.1007/s11356-019-06091-0
Jelodar G, Nazifi S, Akbari A (2013) The prophylactic effect of vitamin C on induced oxidative stress in rat testis following exposure to 900 MHz radio frequency wave generated by a BTS antenna model. Electromagn Biol Med 32(3):409–416. https://doi.org/10.3109/15368378.2012.735208
Kazimirova A, Baranokova M, Staruchova M, Drlickova M, Volkovova K, Dusinska M (2019) Titanium dioxide nanoparticles tested for genotoxicity with the comet and micronucleus assays in vitro, ex vivo and in vivo. Mutat Res Genet Toxicol Environ Mutagen 843:57–65. https://doi.org/10.1016/j.mrgentox.2019.05.001
Khosravi-Katuli K, Prato E, Lofrano G, Guida M, Vale G, Libralato G (2017) Effects of nanoparticles in species of aquaculture interest. Environ Sci Pollut Res Int 24(21):17326–17346. https://doi.org/10.1007/s11356-017-9360-3
Li M, Wu Q, Wang Q, Xiang D, Zhu G (2018) Effect of titanium dioxide nanoparticles on the bioavailability and neurotoxicity of cypermethrin in zebrafish larvae. Aquat Toxicol 199:212–219. https://doi.org/10.1016/j.aquatox.2018.03.022
Liu M, Liu P (2006) Effects of reproductive devel opment in male offspring of wister rats after prenatal exposure to several pestisides. Inspection Quarantine Sci 16(05):24–27. https://doi.org/10.3969/j.issn.1674-5354.2006.05.011 (in Chinese)
Liu Z, Ma Q (2015) Effect of cortinarius rufo -livceus polysaccharide combined with 5-Fu on antioridase system in H22 tumor bearingmouse. Ningxia Med J 37(03):193–196+186. https://doi.org/10.13621/j.1001-5949.2015.03.0193 (in Chinese)
Ma LY, Zhao J, Li XY, Xie YX, Guo X, Yang HF (2022) The function of STING in cyfluthrin-induced testicular injury in rats. J Ningxia Med Univ 44(02):122-127 146. https://doi.org/10.16050/j.cnki.issn1674-6309.2022.02.003 (in Chinese)
Meena R, Kajal K, R, P. (2015) Cytotoxic and genotoxic effects of titanium dioxide nanoparticles in testicular cells of male wistar rat. Appl Biochem Biotechnol 175(2):825–840. https://doi.org/10.1007/s12010-014-1299-y
Minetto D, Volpi Ghirardini A, Libralato G (2016) Saltwater ecotoxicology of Ag, Au, CuO, TiO2, ZnO and C60 engineered nanoparticles: an overview. Environ Int 92–93:189–201. https://doi.org/10.1016/j.envint.2016.03.041
Pandey RK, Prajapati VK (2018) Molecular and immunological toxic effects of nanoparticles. Int J Biol Macromol 107(Pt A):1278–1293. https://doi.org/10.1016/j.ijbiomac.2017.09.110
Saratale RG, Saratale GD, Shin HS, Jacob JM, Pugazhendhi A, Bhaisare M, Kumar G (2018) New insights on the green synthesis of metallic nanoparticles using plant and waste biomaterials: current knowledge, their agricultural and environmental applications. Environ Sci Pollut Res Int 25(11):10164–10183. https://doi.org/10.1007/s11356-017-9912-6
Seven B, Kültiğin Ç, Yalçin E, Acar A (2022) Investigation of cypermethrin toxicity in Swiss albino mice with physiological, genetic and biochemical approaches. Sci Rep 12(1):11439. https://doi.org/10.1038/s41598-022-15800-8
Shi Y, Zhang JH, Jiang M, Zhu LH, Tan HQ, Lu B (2010) Synergistic genotoxicity caused by low concentration of titanium dioxide nanoparticles and p, p’-DDT in human hepatocytes. Environ Mol Mutagen 51(3):192–204. https://doi.org/10.1002/em.20527
Simon U, David O, Peter R, Joseph R, Ijeoma C, Celestine N (2018) Pathological effects of cypermethrin on the testes and accessory sexual glands of Yankasa rams. Arch Pathol Clin Res 2:6–12. https://doi.org/10.29328/journal.apcr.1001006
Sun CJ, Wang Y, Zhao X (2020) Progress on categories and synergistic mechanism of nanopesticides. Chin J Pestic Sci 22(2):205–213. https://doi.org/10.16801/j.issn.1008-7303.2020.0058 (in Chinese)
Wang D, Pei Z, Wang J, Wei L, Zhang X, Wang Y, Xin Z (2022) Application of nanopesticides in pest control. Chin J Vector Biol Control 33(03):442–445+452. https://doi.org/10.11853/j.issn.1003.8280.2022.03.025 (in Chinese)
Wang HX, Zhang R, Li Z, Wang LS, Yu Y, Wang Q, Ding Z et al (2021) Cypermethrin induces Sertoli cell apoptosis through mitochondrial pathway associated with calcium. Toxicol Res (camb) 10(4):742–750. https://doi.org/10.1093/toxres/tfab056
WHO (2016) Pesticide Evaluation Scheme, Vector Ecology and Management. World Health Organization, Geneva
Wu X, Zhang L, Yang C, Zong M, Huang Q, Tao L (2016) Detection on emamectin benzoate-induced apoptosis and DNA damage in Spodoptera frugiperda Sf-9 cell line. Pestic Biochem Physiol 126:6–12. https://doi.org/10.1016/j.pestbp.2015.06.009
Xiao Y, Peijnenburg WJ, Chen G, Vijver MG (2016) Toxicity of copper nanoparticles to Daphnia magna under different exposure conditions. Sci Total Environ 563–564:81–88. https://doi.org/10.1016/j.scitotenv.2016.04.104
Yang C, Wang S (2019) Effects of mixed exposure to low-dose chlorpyrifos and cypermethrin on reproductive function in female rats. Pract Prev Med 26(02):173–176. https://doi.org/10.3969/j.issn.1006-3110.2019.02.013 (in Chinese)
Yang S, Li G, Fu Z, Zhang Y, Zhang J (2016) Detection of single cell DNA damage after subacute exposure to acrylamide in rats by single cell gel electrophoresis. J Shanxi Med Univ 47(12):1071–1075. https://doi.org/10.13753/j.issn.1007-6611.2016.12.005 (in Chinese)
Yang Y, Liu W, Wang J, Zhang Y, Xu W, Tao L (2018) The different effects of natural pyrethrins and beta-cypermethrin on human hepatocyte QSG7701 cells by ROS-mediated oxidative damage. Environ Sci Pollut Res Int 25(24):24230–24240. https://doi.org/10.1007/s11356-018-2503-3
Yao CJ, Zhao C, Liu SM (2018) Mechanisms of the three pathways regulating the apoptosis of testicular germ cells. Zhonghua Nan Ke Xue 24(9):844–850. https://doi.org/10.13263/j.cnki.nja.2018.09.014 (in Chinese)
Zhang Y, Ding H, Xu L, Zhao S, Hu S, Ma A, & Ma Y (2022). Lutein can alleviate oxidative stress, inflammation, and apoptosis induced by excessive alcohol to ameliorate reproductive damage in male rats. Nutrients, 14(12) https://doi.org/10.3390/nu14122385
Zhang Y, Kong C, Chi H, Li J, Xing J, Wang F, Shao L et al (2020) Effect of a beta-cypermethrin and emamectin benzoate pesticide mixture on reproductive toxicity in male mice in a greenhouse environment. Toxicol Mech Methods 30(2):100–106. https://doi.org/10.1080/15376516.2019.1669241
Zhang Y, Luo M, Xu W, Yang M, Wang B, Gao J, Li Y et al (2016) Avermectin confers its cytotoxic effects by inducing DNA damage and mitochondria-associated apoptosis. J Agric Food Chem 64(36):6895–6902. https://doi.org/10.1021/acs.jafc.6b02812
Zhang Y, Zhou Y, Tang Q, Hu F, Feng L, Shen J, Huang B (2018) The protective effects of selenium-enriched spirulina on the reproductive system of male zebrafish (Danio rerio) exposed to beta-cypermethrin. Food Funct 9(11):5791–5804. https://doi.org/10.1039/c8fo01527a
Zhang Z, Gao Y, Deng Y, Liang Q (2017) Experinental study of nano-SiO2 and nano-TiO2 on reproductive toxicity of male rats. Occup Health 33(05):609–612. https://doi.org/10.13329/j.cnki.zyyjk.2017.0185 (in Chinese)
Funding
This work was supported by Weifang Medical University “Healthy Shandong” Major Social Risk Prediction and Governance Collaborative Innovation Center, the Shandong Medical and Health Technology Development Plan Project (202102080346).
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design. The experimental design and implementation were performed by Mingqing Zhong, Xianzhi He, and Yu Fu; the literature search, data collection, and analysis were performed by Yuxin Li, Ruoyu Zhang, Ruolan You, Feiyan Tao, and Lei Fang. The first draft of the manuscript was written by Yuxin Li, Qingfeng Zhai, and Yuanyuan Li drafted and critically revised the work. All the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate.
All experiments were approved by the Animal Experimental Center of Weifang Medical College Committee. All experiments were performed in accordance with relevant guidelines and regulations. All methods are reported in accordance with ARRIVE guidelines for the reporting of animal experiments.
Consent for publication.
Not applicable.
Competing interests
The authors declare no competing interests to disclose.
Additional information
Responsible Editor: Ludek Blaha
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yuxin Li, Mingqing Zhong, and Xianzhi He contributed equally to this work.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Y., Zhong, M., He, X. et al. The combined effect of titanium dioxide nanoparticles and cypermethrin on male reproductive toxicity in rats. Environ Sci Pollut Res 30, 22176–22187 (2023). https://doi.org/10.1007/s11356-022-23796-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-23796-x