Abstract
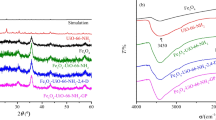
In wastewater, heavy metal Hg causes serious harm to ecology, so it needs to be removed. In this paper, a novel MOF adsorbent (UiO-66-QU) was prepared by modifying UiO-66-NH2 with 4-quinolinecarboxaldehyde, which was used to selectively remove Hg(II) from water. The adsorbent was characterized using Fourier transform infrared spectroscopy (FT-IR), field emission scanning electron microscopy (SEM), Brunauer–Emmett–Teller (BET), zeta potentiometer, and X-ray photoelectron spectroscopy (XPS). In order to investigate the Hg(II) adsorption performance of UiO-66-QU, the effect of time, initial concentration, pH, and temperature were carried out. Langmuir model fitting shows that the maximum adsorption capacity of UiO-66-QU for Hg(II) is 556 mg/g at 298 K. The experimental results show that UiO-66-QU has better Hg(II) adsorption capacity than UiO-66-NH2. The isotherm is in accordance with pseudo-second-order models. It is indicated that the adsorption process is mainly monolayer chemical adsorption. The thermodynamic parameters also indicate that the adsorption process is spontaneous and endothermic. It has excellent reusability and selectivity. XPS and the zeta potential showed that the adsorption mechanism was the complex reaction of Hg(II) with nitrogenous group. Therefore, the adsorbent has potential application prospects in removal of Hg(II) from wastewater.











Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Ahmadijokani F, Tajahmadi S, Haris MH, Bahi A, Rezakazemi M, Molavi H, Ko F, Arjmand M (2021) Fe3O4@PAA@UiO-66-NH2 magnetic nanocomposite for selective adsorption of Quercetin. Chemosphere 275:130087. https://doi.org/10.1016/j.chemosphere.2021.130087
Aika K, Kumasaka M, Oma T, Kato O, Matsuda H, Watanabe N, Yamazaki K, Ozaki A, Onishi T (1986) Support and promoter effect of ruthenium catalyst. III. Kinetics of ammonia synthesis over various Ru catalysts. Appl Catal 28:57–68. https://doi.org/10.1016/S0166-9834(00)82492-6
Awad FS, AbouZied KM, Bakry AM, Abou El-Maaty WM, El-Wakil AM, El-Shall MS (2021) Polyacrylonitrile modified partially reduced graphene oxide composites for the extraction of Hg(II) ions from polluted water. J Mater Sci 56(13):7982–7999. https://doi.org/10.1007/s10853-021-05797-2
Caicedo Salcedo OD, Vargas DP, Giraldo L, Moreno-Piraján JC (2021) Study of mercury [Hg(II)] adsorption from aqueous solution on functionalized activated carbon. ACS Omega 6(18):11849–11856. https://doi.org/10.1021/acsomega.0c06084
Chang Z, Li F, Qi X, Jiang B, Kou J, Sun C (2020) Selective and efficient adsorption of Au (III) in aqueous solution by Zr-based metal-organic frameworks (MOFs): an unconventional way for gold recycling. J Hazard Mater 391:122175. https://doi.org/10.1016/j.jhazmat.2020.122175
Chen Q, He Q, Lv M, Xu Y, Yang H, Liu X, Wei F (2015) Selective adsorption of cationic dyes by UiO-66-NH2. Appl Surf Sci 327:77–85. https://doi.org/10.1016/j.apsusc.2014.11.103
Duan L, Hu X, Sun D, Liu Y, Guo Q, Zhang T, Zhang B (2020) Rapid removal of low concentrations of mercury from wastewater using coal gasification slag. Korean J Chem Eng 37(7):1166–1173. https://doi.org/10.1007/s11814-020-0546-x
Fast CD, Woods J, Lentchner J, Makal TA (2019) Stabilizing defects in metal–organic frameworks: pendant Lewis basic sites as capping agents in UiO-66-type MOFs toward highly stable and defective porous materials. Dalton Trans 48(39):14696–14704. https://doi.org/10.1039/C9DT03004B
Foo KY, Hameed BH (2010) Insights into the modeling of adsorption isotherm systems. Chem Eng J 156(1):2–10. https://doi.org/10.1016/j.cej.2009.09.013
Fu L, Wang S, Lin G, Zhang L, Liu Q, Fang J, Wei C, Liu G (2019) Post-functionalization of UiO-66-NH2 by 2,5-Dimercapto-1,3,4-thiadiazole for the high efficient removal of Hg(II) in water. J Hazard Mater 368:42–51. https://doi.org/10.1016/j.jhazmat.2019.01.025
Fu Y, Sun Y, Zheng Y, Jiang J, Yang C, Wang J, Hu J (2021) New network polymer functionalized magnetic-mesoporous nanoparticle for rapid adsorption of Hg(II) and sequential efficient reutilization as a catalyst. Sep Purif Technol 259:118112. https://doi.org/10.1016/j.seppur.2020.118112
Gayathri S, Dev VV, Shiny Raj R, Krishnakumar A, Vishnu Maya TM, Anoop Krishnan K (2021) Spatiotemporal evaluation of hydrochemical facies and pesticide residues in the cardamom plantations of Southern Western Ghats, India. Environ Nanotechnol, Monitor Manage 16:100599. https://doi.org/10.1016/j.enmm.2021.100599
Ge H, Du J (2020) Selective adsorption of Pb(II) and Hg(II) on melamine-grafted chitosan. Int J Biol Macromol 162:1880–1887. https://doi.org/10.1016/j.ijbiomac.2020.08.070
Geravand E, Farzaneh F, Ghiasi M (2019) Metalation and DFT studies of metal organic frameworks UiO-66(Zr) with vanadium chloride as allyl alcohol epoxidation catalyst. J Mol Struct 1198:126940
Gunay Gurer A, Aktas K, Ozkaleli Akcetin M, Erdem Unsar A, Asilturk M (2021) Adsorption isotherms, thermodynamics, and kinetic modeling of methylene blue onto novel carbonaceous adsorbent derived from bitter orange peels. Water Air Soil Pollut 232(4):138. https://doi.org/10.1007/s11270-021-05090-7
Hashem A, Sanousy MA, Mohamed LA, Okoye PU, Hameed BH (2021) Natural and low-cost P turgidum for efficient adsorption of Hg(II) ions from contaminated solution: isotherms and kinetics studies. J Polymers Environ 29(1):304–312. https://doi.org/10.1007/s10924-020-01879-5
Henny CJ, Hill EF, Hoffman DJ, Spalding MG, Grove RA (2002) Nineteenth century mercury hazard to wading birds and cormorants of the carson river nevada. Ecotoxicology 11(4):213–231
Hu P, Zhao Z, Sun X, Muhammad Y, Li J, Chen S, Pang C, Liao T, Zhao Z (2019) Construction of crystal defect sites in N-coordinated UiO-66 via mechanochemical in-situ N-doping strategy for highly selective adsorption of cationic dyes. Chem Eng J 356:329–340. https://doi.org/10.1016/j.cej.2018.09.060
Hu X, Chen C, Zhang D, Xue Y (2021) Kinetics, isotherm and chemical speciation analysis of Hg(II) adsorption over oxygen-containing MXene adsorbent. Chemosphere 278:130206. https://doi.org/10.1016/j.chemosphere.2021.130206
Huang L, He M, Chen B, Hu B (2016) Mercapto functionalized magnetic Zr-MOF by solvent-assisted ligand exchange for Hg2+ removal from water. J Mater Chem A 4(14):5159–5166. https://doi.org/10.1039/C6TA00343E
Huang S, Liang Q, Geng J, Luo H, Wei Q (2019) Sulfurized biochar prepared by simplified technic with superior adsorption property towards aqueous Hg(II) and adsorption mechanisms. Mater Chem Phys 238:121919. https://doi.org/10.1016/j.matchemphys.2019.121919
Kandiah M, Nilsen MH, Usseglio S, Jakobsen S, Olsbye U, Tilset M, Larabi C, Quadrelli EA, Bonino F, Lillerud KP (2010) Synthesis and stability of tagged UiO-66 Zr-MOFs. Chem Mater 22(24):6632–6640. https://doi.org/10.1021/cm102601v
Kang C, Gao L, Zhu H, Lang C, Jiang J, Wei J (2021) Adsorption of Hg(II) in solution by mercaptofunctionalized palygorskite. Environ Sci Pollut Res 28(46):66287–66302. https://doi.org/10.1007/s11356-021-15637-0
Kurniawan A, Sutiono H, Indraswati N, Ismadji S (2012) Removal of basic dyes in binary system by adsorption using rarasaponin–bentonite: revisited of extended Langmuir model. Chem Eng J 189–190:264–274. https://doi.org/10.1016/j.cej.2012.02.070
Leus K, Perez JPH, Folens K, Meledina M, Van Tendeloo G, Du Laing G, Van Der Voort P (2017) UiO-66-(SH)2 as stable, selective and regenerable adsorbent for the removal of mercury from water under environmentally-relevant conditions. Faraday Discuss 201:145–161. https://doi.org/10.1039/C7FD00012J
Li Z, Li L, Hu D, Gao C, Xiong J, Jiang H, Li W (2019) Efficient removal of heavy metal ions and organic dyes with cucurbit[8]uril-functionalized chitosan. J Colloid Interface Sci 539:400–413. https://doi.org/10.1016/j.jcis.2018.12.078
Li H, Liu H, Zhang J, Cheng Y, Zhang C, Fei X, Xian Y (2017) Platinum nanoparticle encapsulated metal–organic frameworks for colorimetric measurement and facile removal of mercury(II). ACS Appl Mater Interfaces 9(46):40716–40725. https://doi.org/10.1021/acsami.7b13695
Lin S, Bediako JK, Cho C-W, Song M-H, Zhao Y, Kim J-A, Choi J-W, Yun Y-S (2018) Selective adsorption of Pd(II) over interfering metal ions (Co(II), Ni(II), Pt(IV)) from acidic aqueous phase by metal-organic frameworks. Chem Eng J 345:337–344. https://doi.org/10.1016/j.cej.2018.03.173
Lin S, Kumar Reddy DH, Bediako JK, Song M-H, Wei W, Kim J-A, Yun Y-S (2017) Effective adsorption of Pd(ii), Pt(iv) and Au(iii) by Zr(iv)-based metal–organic frameworks from strongly acidic solutions. J Mater Chem A 5(26):13557–13564. https://doi.org/10.1039/C7TA02518A
Liu F, Xiong W, Liu J, Cheng Q, Cheng G, Shi L, Zhang Y (2018) Novel amino-functionalized carbon material derived from metal organic framework: a characteristic adsorbent for U(VI) removal from aqueous environment. Colloids Surf, A 556:72–80. https://doi.org/10.1016/j.colsurfa.2018.08.009
Liu H, Wang J, Jin H, Wei M, Ren W, Zhang Y, Wu L, He B (2021a) Electrochemical biosensor for sensitive detection of Hg2+ baesd on clustered peonylike copper-based metal-organic frameworks and DNAzyme-driven DNA Walker dual amplification signal strategy. Sens Actuators, B Chem 329:129215. https://doi.org/10.1016/j.snb.2020.129215
Liu J, Cao H, Shi Y, Jiang P (2021b) Enhanced methane delivery in MIL-101(Cr) by means of subambient cooling. Energy Fuels 35(8):6898–6908. https://doi.org/10.1021/acs.energyfuels.1c00617
Lu M, Deng Y, Luo Y, Lv J, Li T, Xu J, Chen S-W, Wang J (2019) Graphene aerogel–metal–organic framework-based electrochemical method for simultaneous detection of multiple heavy-metal ions. Anal Chem 91(1):888–895. https://doi.org/10.1021/acs.analchem.8b03764
Luu CL, Nguyen TTV, Nguyen T, Hoang TC (2015) Synthesis, characterization and adsorption ability of UiO-66-NH2. Adv Nat Sci-Nanosci Nanotechnol 6(2):025004. https://doi.org/10.1088/2043-6262/6/2/025004
Navaneeth A, Sreeda P, Vishnu Maya TM, Sanusree PS, Harikumar PS (2021) Evaluation of the challenges in water governance through citizen’s perception and Water Quality Index: a case study of a fast-growing city in India. H2 Open J 4(1):336–351. https://doi.org/10.2166/h2oj.2021.106
Moghimi A, Abdouss M (2012) Determination and preconcentration of mercury(II) from natural water and milk sample by Modified nano polyacrylonitrile fiber. Afr J Pure Appl Chem 6(14):173–182. https://doi.org/10.5897/AJPAC12.057
Parida K, Mishra KG, Dash SK (2012) Adsorption of toxic metal ion Cr(VI) from aqueous state by TiO2-MCM-41: equilibrium and kinetic studies. J Hazard Mater 241:395–403. https://doi.org/10.1016/j.jhazmat.2012.09.052
Qin W, Qian G, Tao H, Wang J, Sun J, Cui X, Zhang Y, Zhang X (2019) Adsorption of Hg(II) ions by PAMAM dendrimers modified attapulgite composites. React Funct Polym 136:75–85. https://doi.org/10.1016/j.reactfunctpolym.2019.01.005
Sadegh H, Ali GAM, Makhlouf ASH, Chong KF, Alharbi NS, Agarwal S, Gupta VK (2018) MWCNTs-Fe3O4 nanocomposite for Hg(II) high adsorption efficiency. J Mol Liq 258:345–353. https://doi.org/10.1016/j.molliq.2018.03.012
Saleem H, Rafique U, Davies RP (2016) Investigations on post-synthetically modified UiO-66-NH2 for the adsorptive removal of heavy metal ions from aqueous solution. Microporous Mesoporous Mater 221:238–244. https://doi.org/10.1016/j.micromeso.2015.09.043
Samanta P, Desa AV, Sharma S, Chandra P, Ghosh SK (2018) Selective recognition of Hg2+ ion in water by a functionalized metal–organic framework (MOF) based chemodosimeter. Inorganic Chem 57(5):2360–2364. https://doi.org/10.1021/acs.inorgchem.7b02426
Shahat A, Hassan HMA, Azzazy HME (2013) Optical metal-organic framework sensor for selective discrimination of some toxic metal ions in water. Anal Chim Acta 793:90–98. https://doi.org/10.1016/j.aca.2013.07.012
Sompornpailin D, Ratanatawanate C, Sattayanon C, Namuangruk S, Punyapalakul P (2020) Selective adsorption mechanisms of pharmaceuticals on benzene-1,4-dicarboxylic acid-based MOFs: effects of a flexible framework, adsorptive interactions and the DFT study. Sci Total Environ 720:137449. https://doi.org/10.1016/j.scitotenv.2020.137449
Stock N, Biswas S (2012) Synthesis of metal-organic frameworks (MOFs): routes to various MOF topologies, morphologies, and composites. Chem Rev 112(2):933–969. https://doi.org/10.1021/cr200304e
Upadhyay U, Sreedhar I, Singh SA, Patel CM, Anitha KL (2021) Recent advances in heavy metal removal by chitosan based adsorbents. Carbohydrate Polymers 251:117000. https://doi.org/10.1016/j.carbpol.2020.117000
Wang C, Xiong C, He Y, Yang C, Li X, Zheng J, Wang S (2021a) Facile preparation of magnetic Zr-MOF for adsorption of Pb(II) and Cr(VI) from water: Adsorption characteristics and mechanisms. Chem Eng J 415:128923. https://doi.org/10.1016/j.cej.2021.128923
Wang J, Dai L, Hu S, Yin H, Yang M, Reheman A, Yan G (2021b) Corn bracts loading copper sulfide for rapid adsorption of Hg(II) and sequential efficient reutilization as a photocatalyst. Water Sci Technol 83(12):2921–2930. https://doi.org/10.2166/wst.2021.181
Wang J, Guo X (2020) Adsorption kinetic models: physical meanings, applications, and solving methods. J Hazard Mater 390:122156. https://doi.org/10.1016/j.jhazmat.2020.122156
Wang K, Gu J, Yin N (2017) Efficient removal of Pb(II) and Cd(II) using NH2-functionalized Zr-MOFs via rapid microwave-promoted synthesis. Ind Eng Chem Res 56(7):1880–1887. https://doi.org/10.1021/acs.iecr.6b04997
Weber TW, Chakravorti RK (1974) Pore and solid diffusion models for fixed-bed adsorbers. AIChE J 20(2):228–238. https://doi.org/10.1002/aic.690200204
Wu R, Zhao K, Lv W, Xu J, Hu J, Liu H, Wang H (2021) Solid-phase synthesis of bi-functionalized porous organic polymer for simultaneous removal of Hg(II) and Pb(II). Microporous Mesoporous Mater 316:110942. https://doi.org/10.1016/j.micromeso.2021.110942
Zabihi M, Asl AH, Ahmadpour A (2010) Studies on adsorption of mercury from aqueous solution on activated carbons prepared from walnut shell. J Hazard Mater 174(1–3):251–256. https://doi.org/10.1016/j.jhazmat.2009.09.044
Zhang X, Xia T, Jiang K, Cui Y, Yang Y, Qian G (2017) Highly sensitive and selective detection of mercury (II) based on a zirconium metal-organic framework in aqueous media. J Solid State Chem 253:277–281. https://doi.org/10.1016/j.jssc.2017.06.008
Zhao M, Huang Z, Wang S, Zhang L, Zhou Y (2019) Design of l-cysteine functionalized UiO-66 MOFs for selective adsorption of Hg(II) in aqueous medium. ACS Appl Mater Interfaces 11(50):46973–46983. https://doi.org/10.1021/acsami.9b17508
Zhao S, Mei J, Xu H, Liu W, Qu Z, Cui Y, Yan N (2018) Research of mercury removal from sintering flue gas of iron and steel by the open metal site of Mil-101(Cr). J Hazard Mater 351:301–307. https://doi.org/10.1016/j.jhazmat.2017.12.016
Zhao Y, Wang D, Wei W, Cui L, Cho C-W, Wu G (2021) Effective adsorption of mercury by Zr(IV)-based metal-organic frameworks of UiO-66-NH2 from aqueous solution. Environ Sci Pollut Res 28(6):7068–7075. https://doi.org/10.1007/s11356-020-11080-9
Zheng C, Wu Q, Hu X, Wang Y, Chen Y, Zhang S, Zheng H (2021) Adsorption behavior of heavy metal ions on a polymer-immobilized amphoteric biosorbent: Surface interaction assessment. J Hazard Mater 403:123801. https://doi.org/10.1016/j.jhazmat.2020.123801
Funding
The work was supported by the National Natural Science Foundation of China (No. U2002214) and the Natural Science Foundation of Yunnan Province (2019FA022).
Author information
Authors and Affiliations
Contributions
Yuting Hu: methodology, data curation, writing—original draft preparation. Shixing Wang: writing—reviewing and editing. Libo Zhang: supervision, conceptualization, supervision. Fan Yang: editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
The facts and views in the manuscript are solely ours, and we are totally responsible for authenticity, validity, and originality. We also declare that this manuscript is our original work, and we have not copied from anywhere else. There is no plagiarism in our manuscript.
Consent for publication
We undertake and agree that the manuscript submitted to your journal has not been published elsewhere and has not been simultaneously submitted to other journals.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, Y., Wang, S., Zhang, L. et al. Selective removal of Hg(II) by UiO-66-NH2 modified by 4-quinolinecarboxaldehyde: from experiment to mechanism. Environ Sci Pollut Res 30, 2283–2297 (2023). https://doi.org/10.1007/s11356-022-22276-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-22276-6